Details of the Target
General Information of Target
Target Site Mutations in Different Cell Lines
| Cell line | Mutation details | Probe for labeling this protein in this cell | |||
|---|---|---|---|---|---|
| NB1 | SNV: p.A236V | DBIA Probe Info | |||
| YSCCC | SNV: p.R22K | DBIA Probe Info | |||
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
m-APA Probe Info |
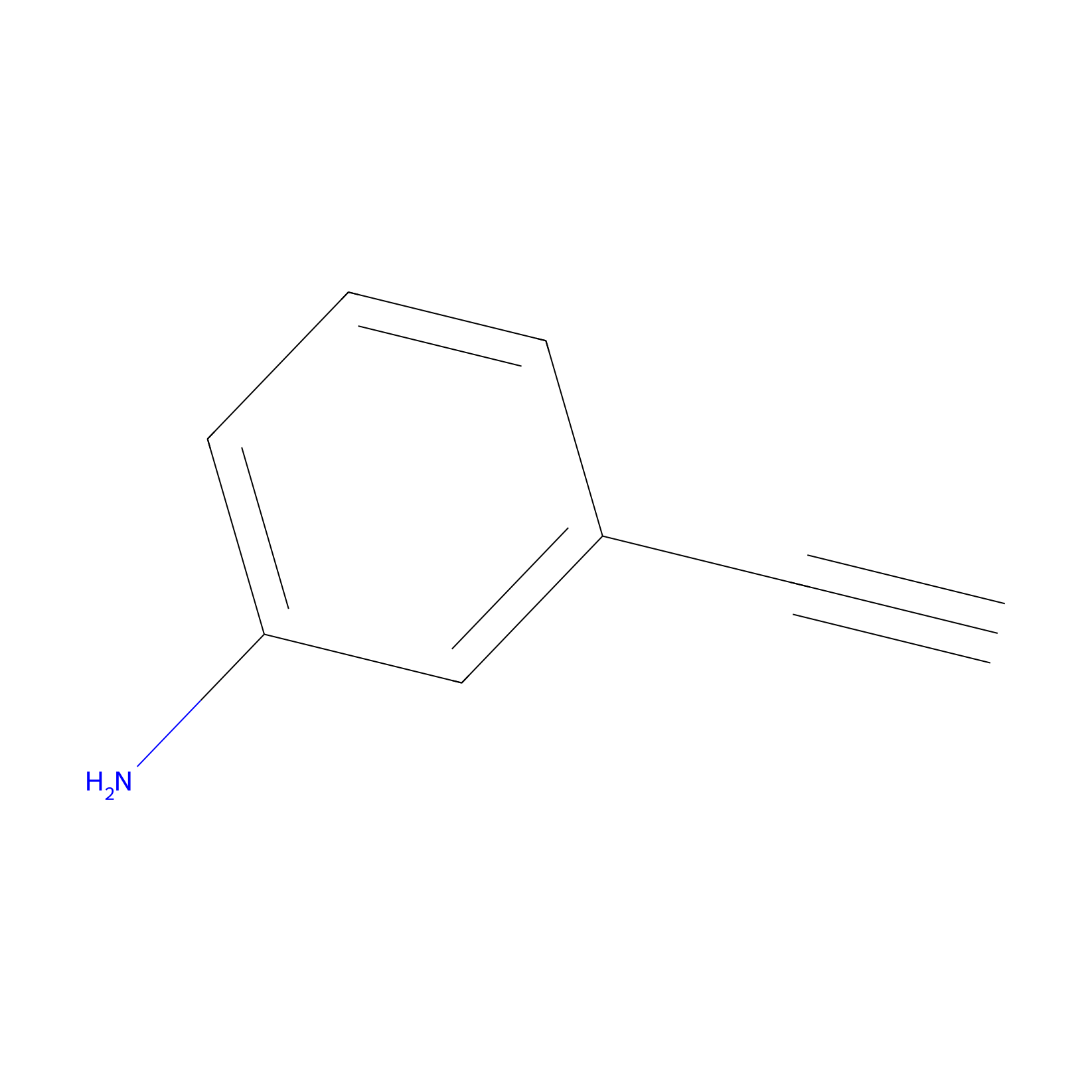 |
10.18 | LDD0402 | [1] | |
|
N1 Probe Info |
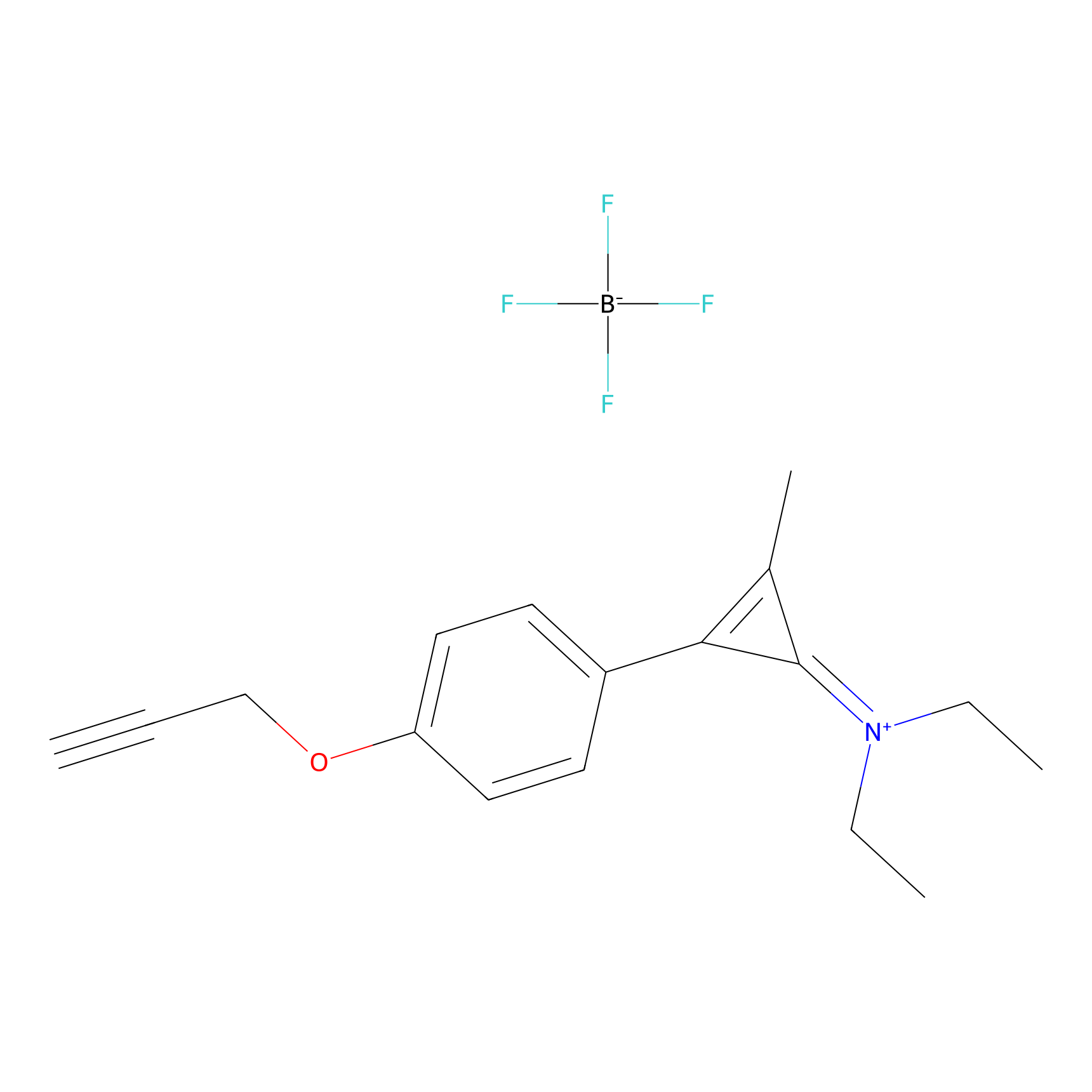 |
100.00 | LDD0242 | [2] | |
|
TH211 Probe Info |
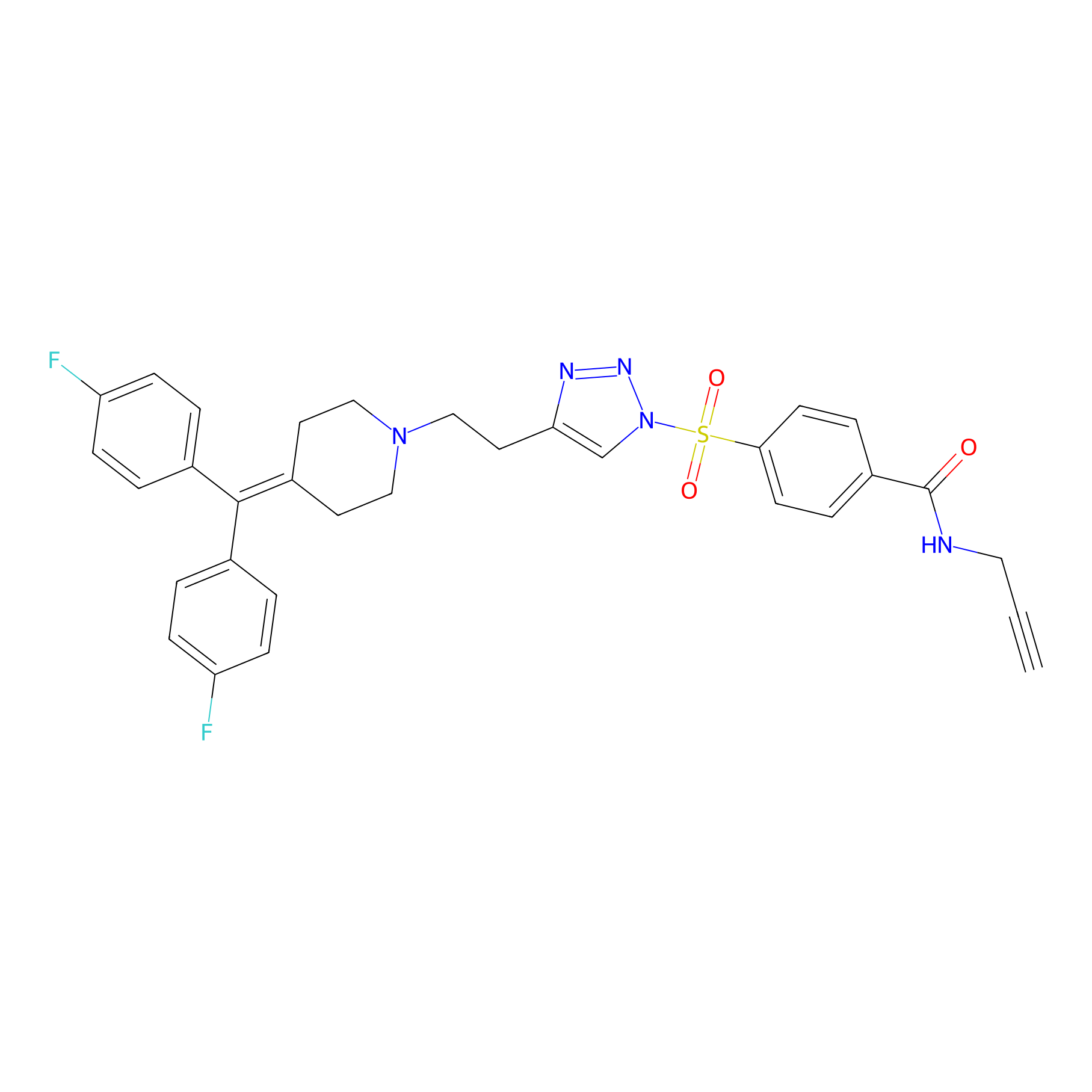 |
Y118(20.00); Y227(20.00); Y301(20.00); Y156(13.01) | LDD0257 | [3] | |
|
TH214 Probe Info |
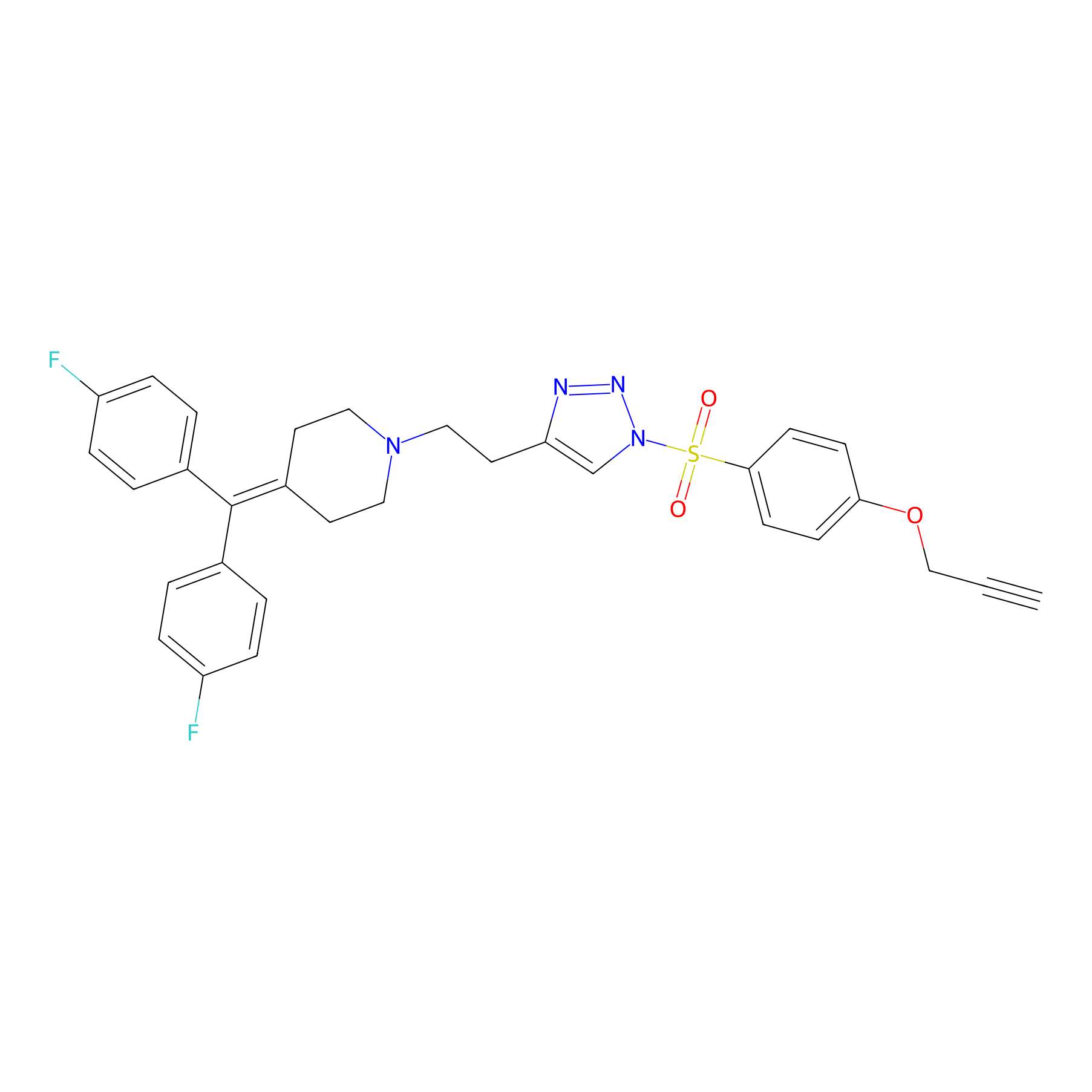 |
Y243(7.62) | LDD0258 | [3] | |
|
TH216 Probe Info |
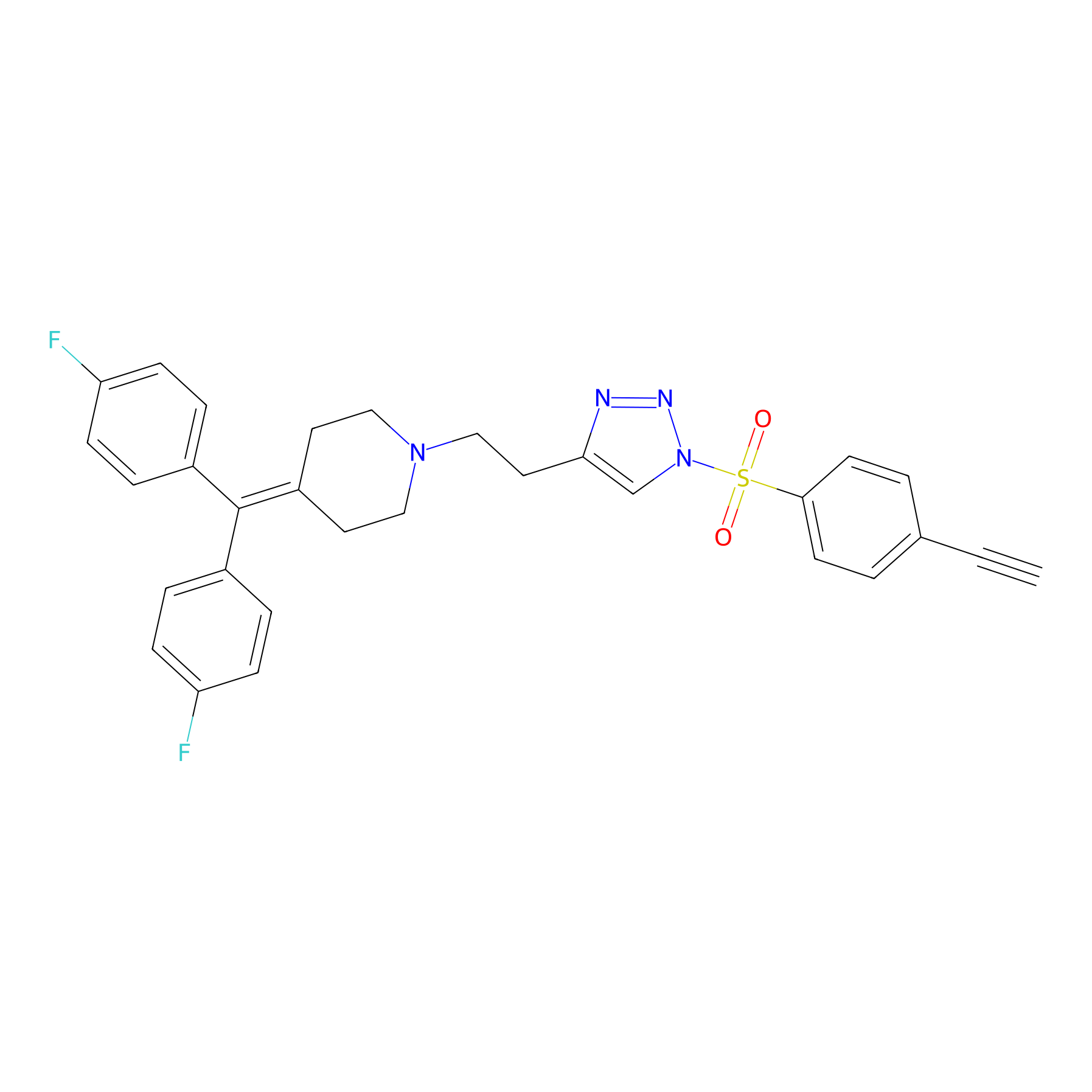 |
Y227(20.00) | LDD0259 | [3] | |
|
YN-1 Probe Info |
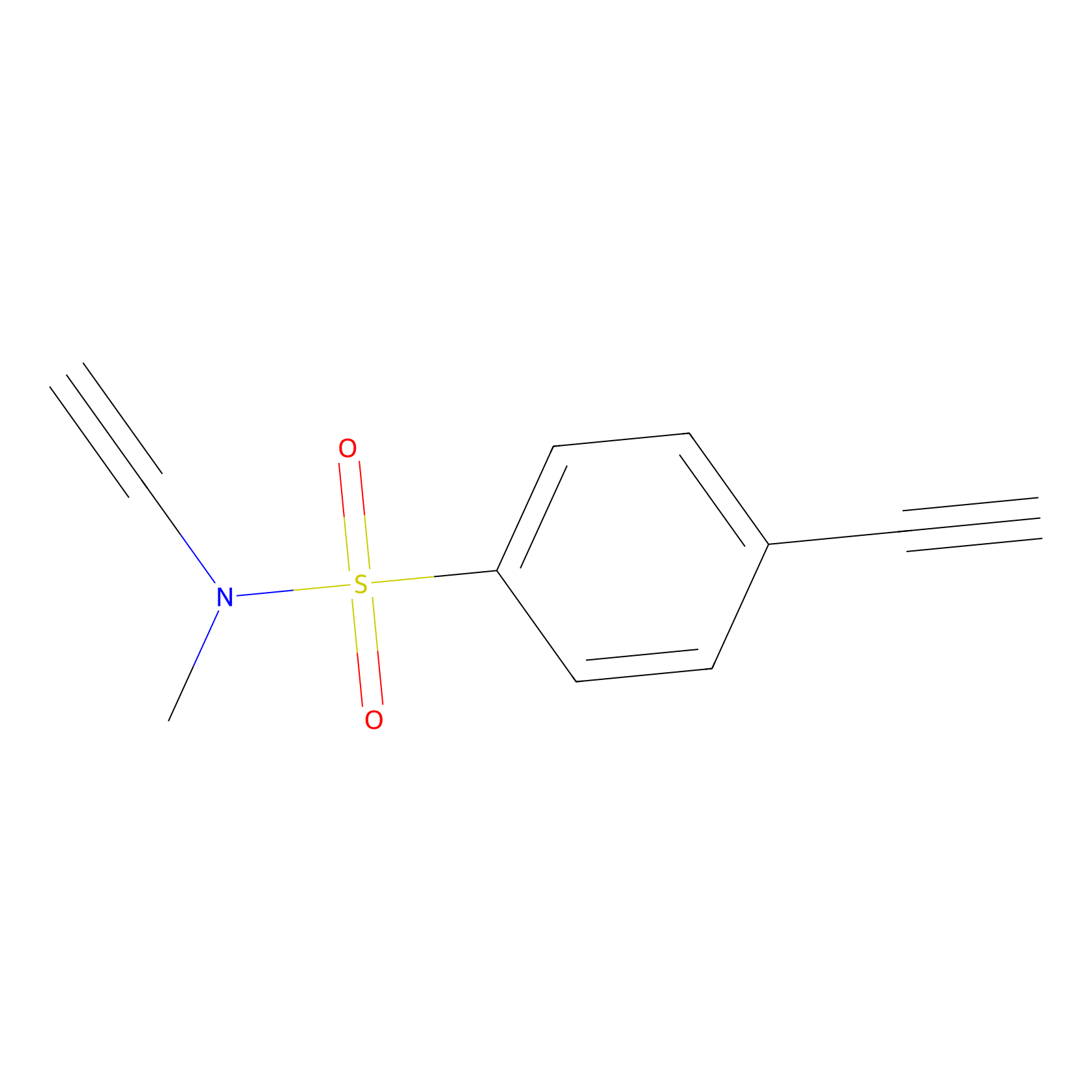 |
100.00 | LDD0444 | [4] | |
|
STPyne Probe Info |
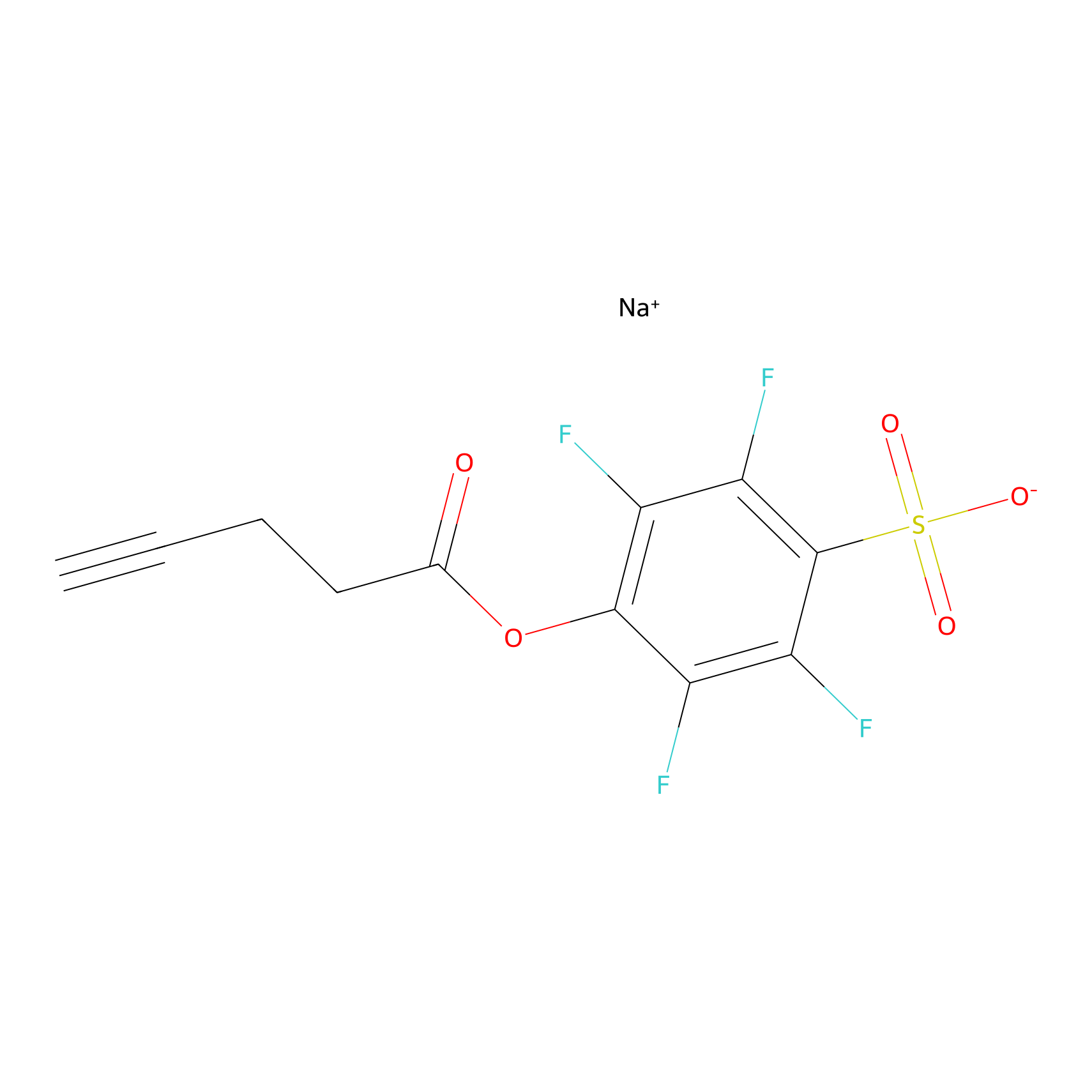 |
K18(7.14); K313(8.93); K321(9.71); K385(10.00) | LDD0277 | [5] | |
|
ONAyne Probe Info |
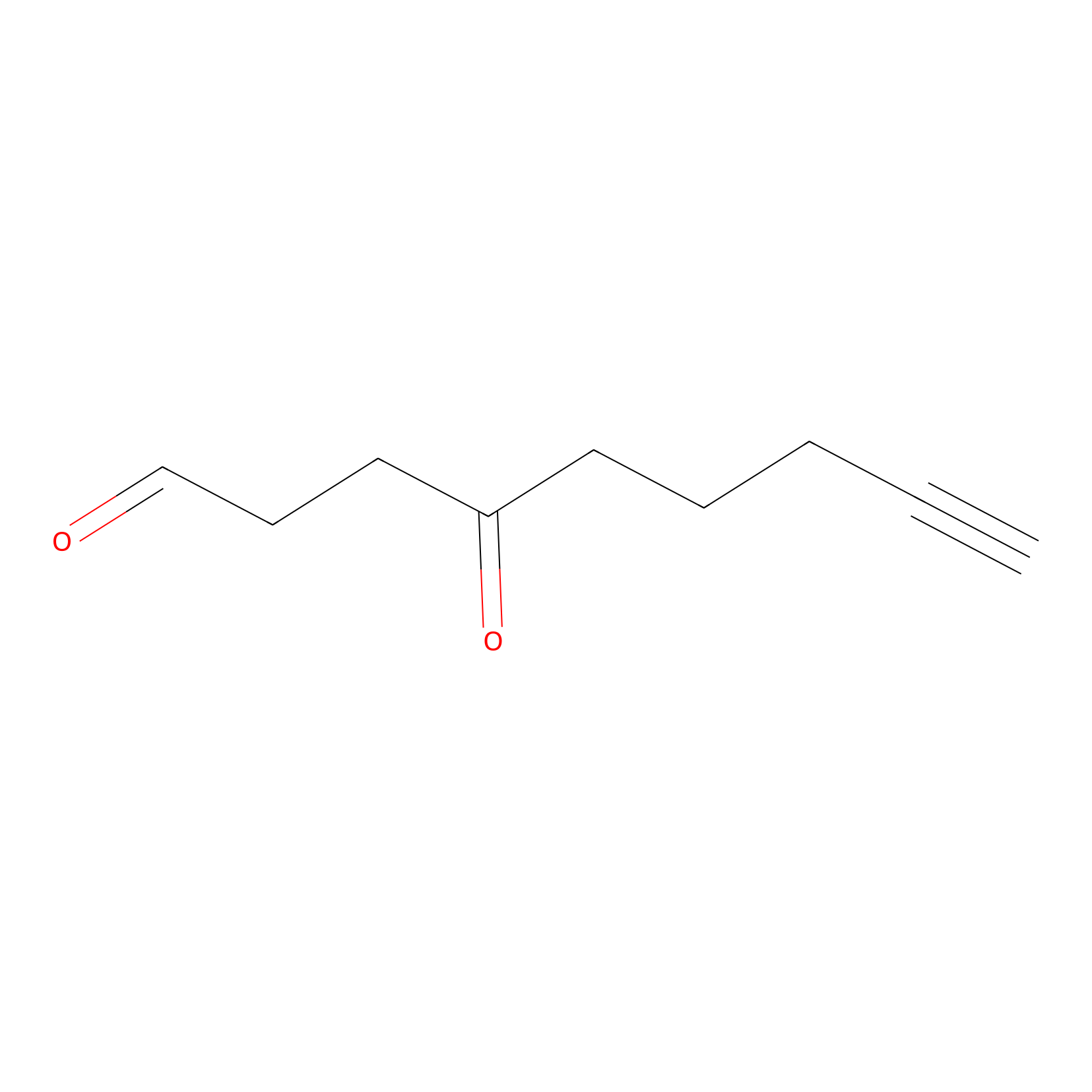 |
N.A. | LDD0273 | [5] | |
|
Probe 1 Probe Info |
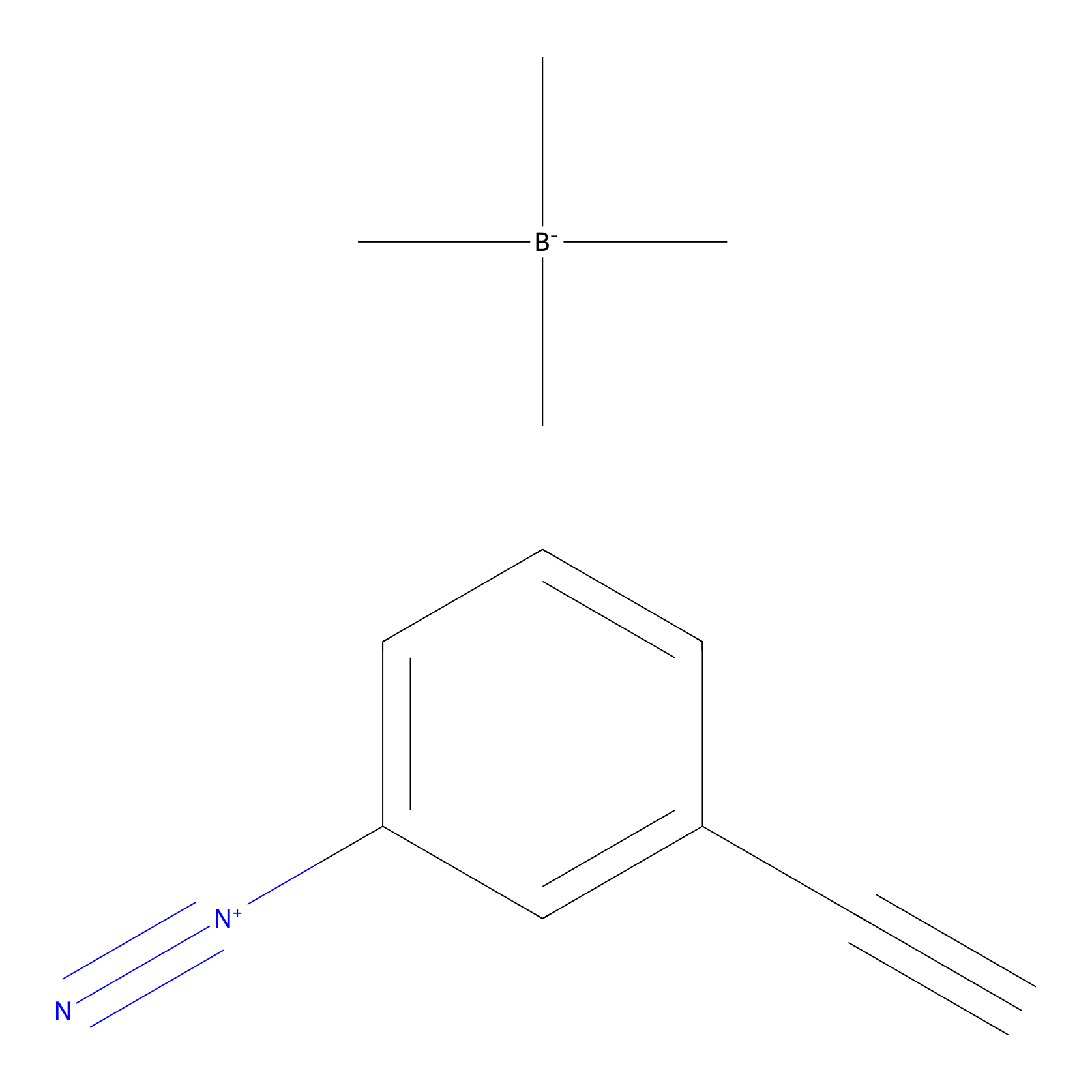 |
Y82(29.77); Y156(8.93); Y227(58.05); Y272(14.51) | LDD3495 | [6] | |
|
BTD Probe Info |
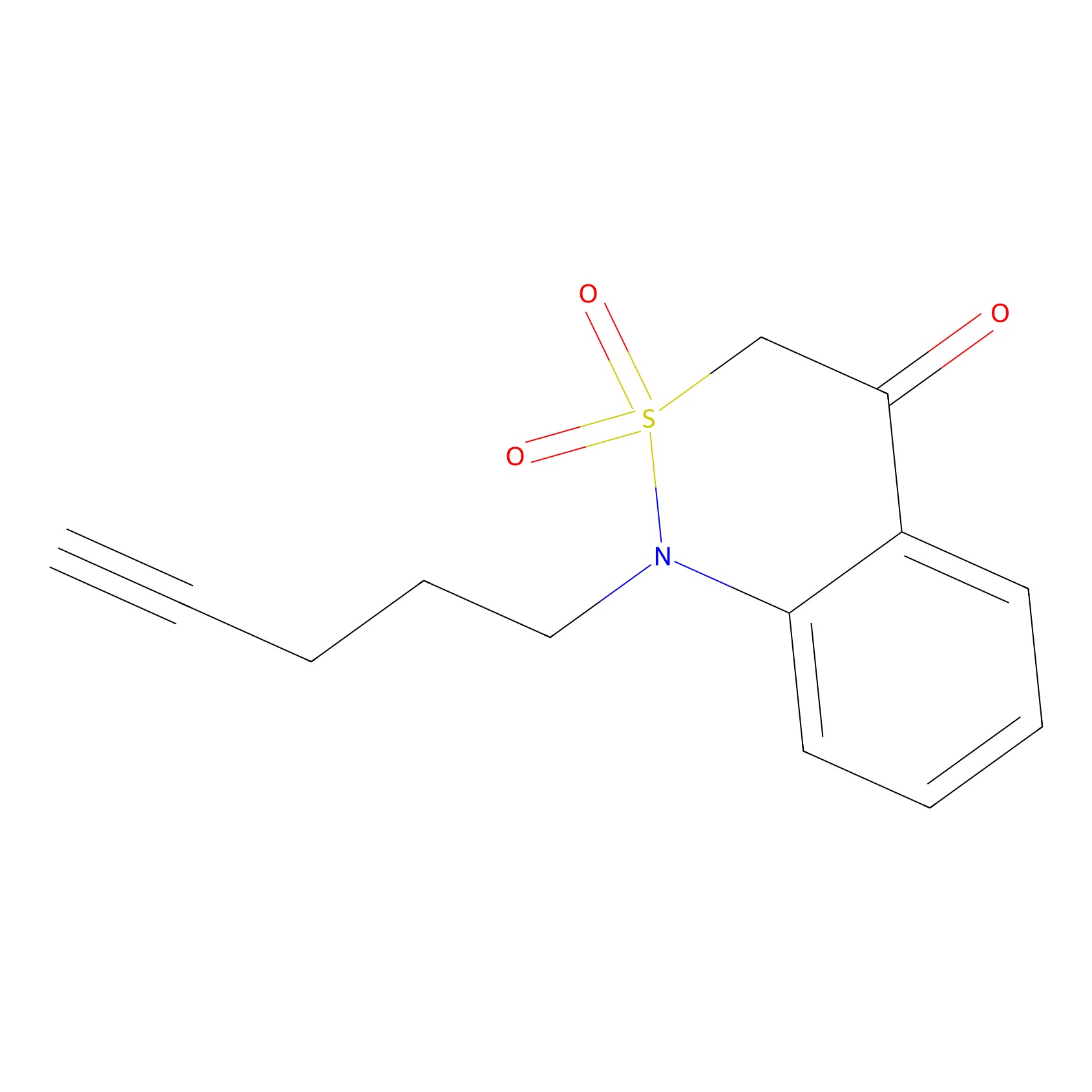 |
C273(1.80); C41(1.57) | LDD1700 | [7] | |
|
AHL-Pu-1 Probe Info |
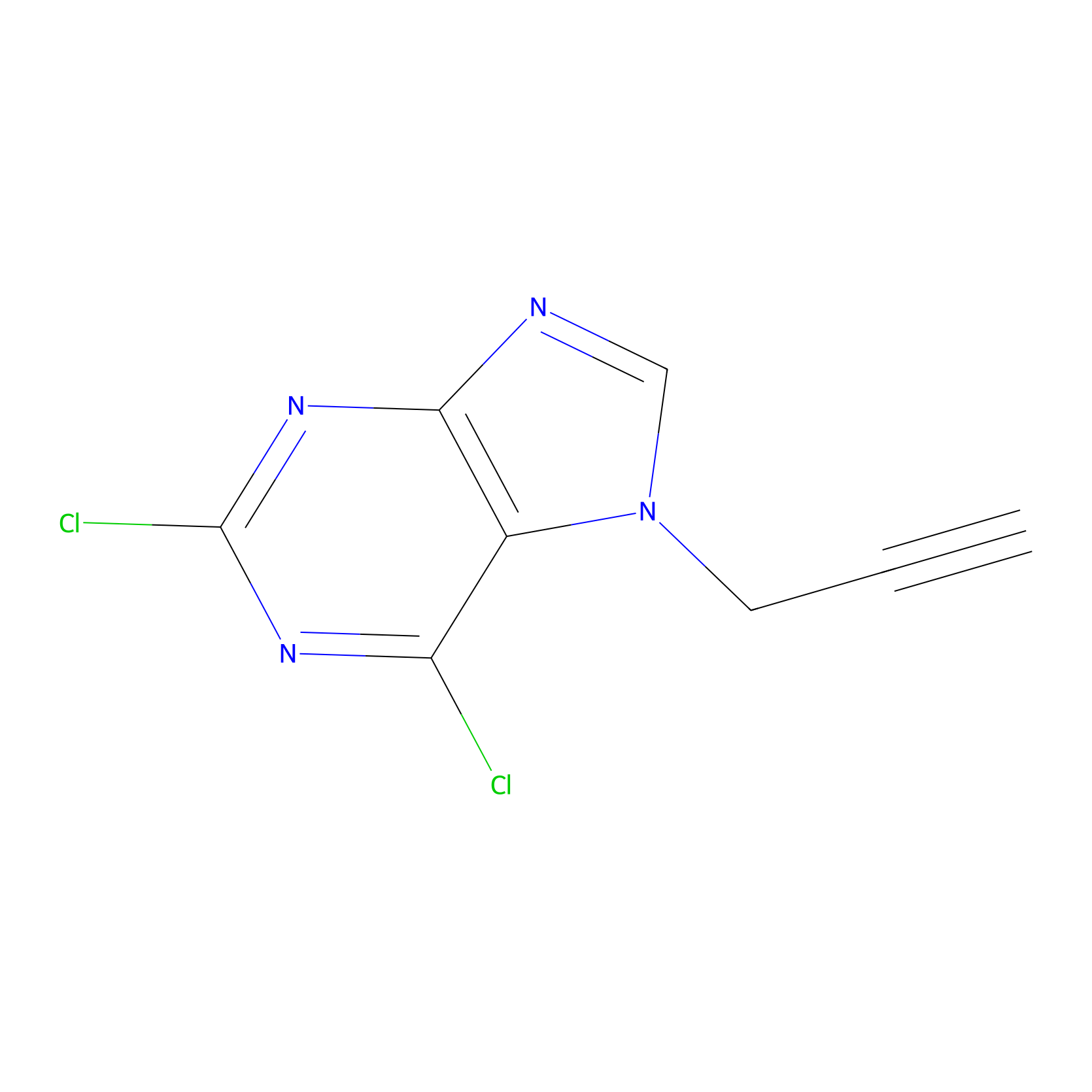 |
C273(6.24) | LDD0170 | [8] | |
|
EA-probe Probe Info |
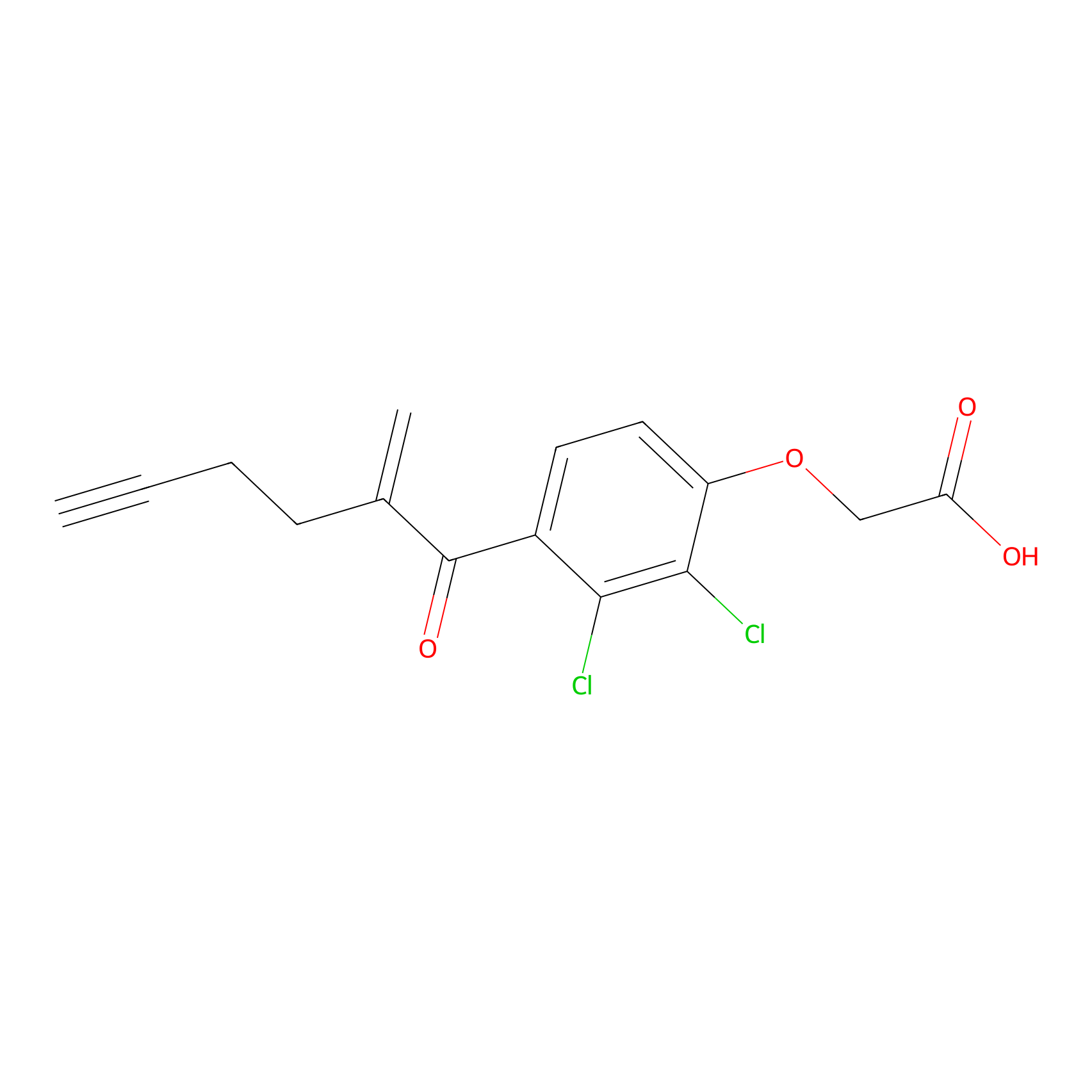 |
C181(0.88) | LDD2210 | [9] | |
|
DBIA Probe Info |
 |
C261(4.31) | LDD0209 | [10] | |
|
HHS-482 Probe Info |
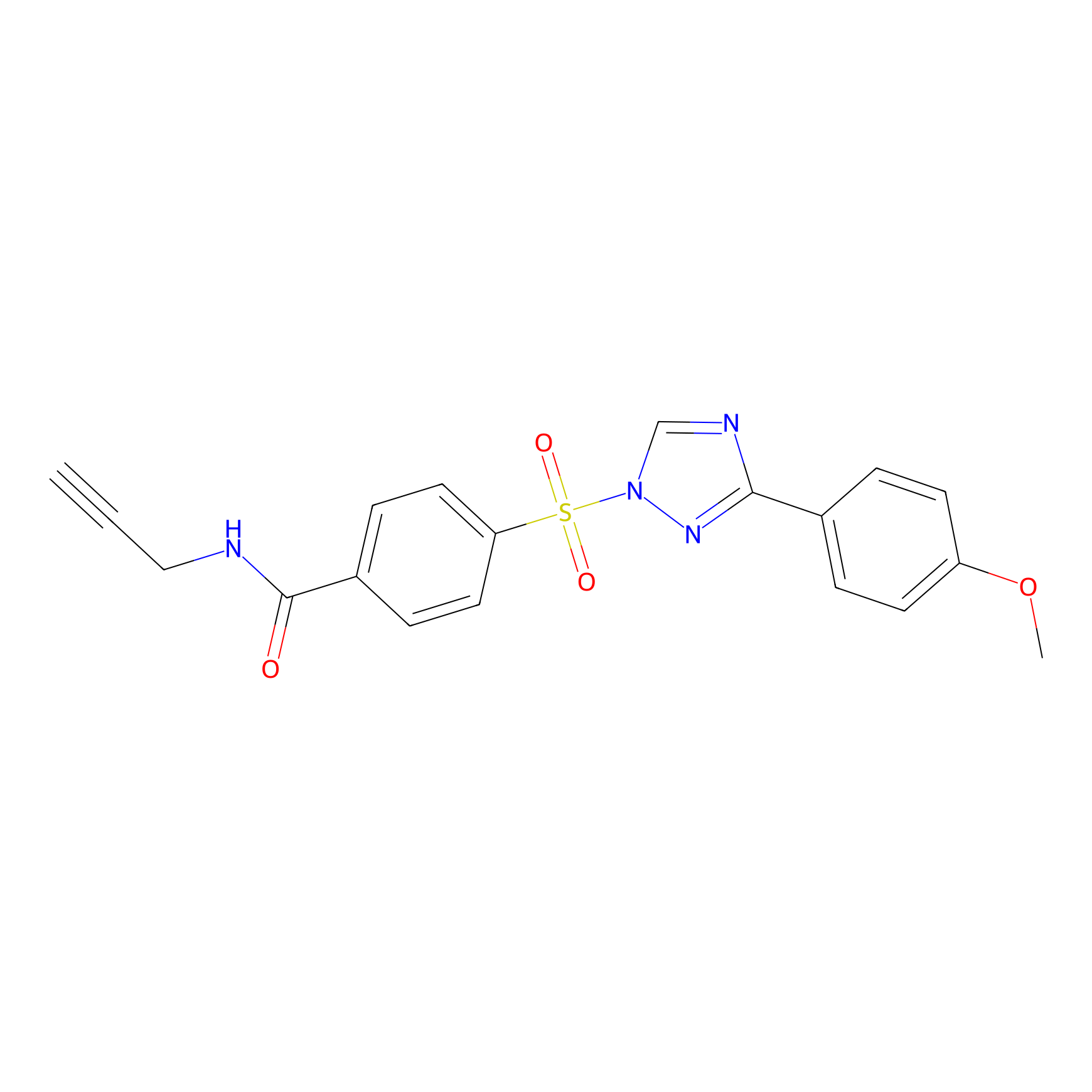 |
Y156(0.59); Y227(0.71) | LDD0285 | [11] | |
|
5E-2FA Probe Info |
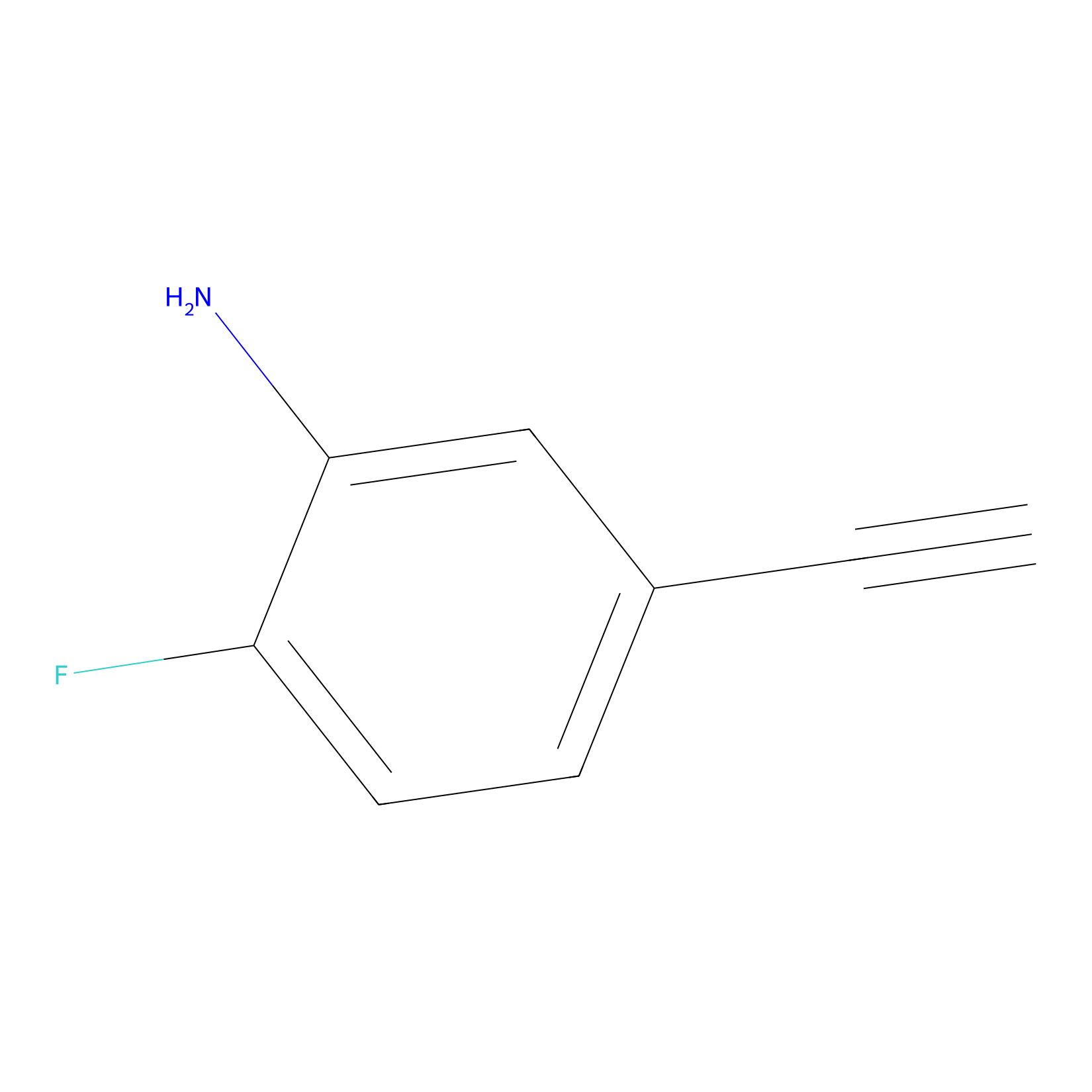 |
N.A. | LDD2235 | [12] | |
|
ATP probe Probe Info |
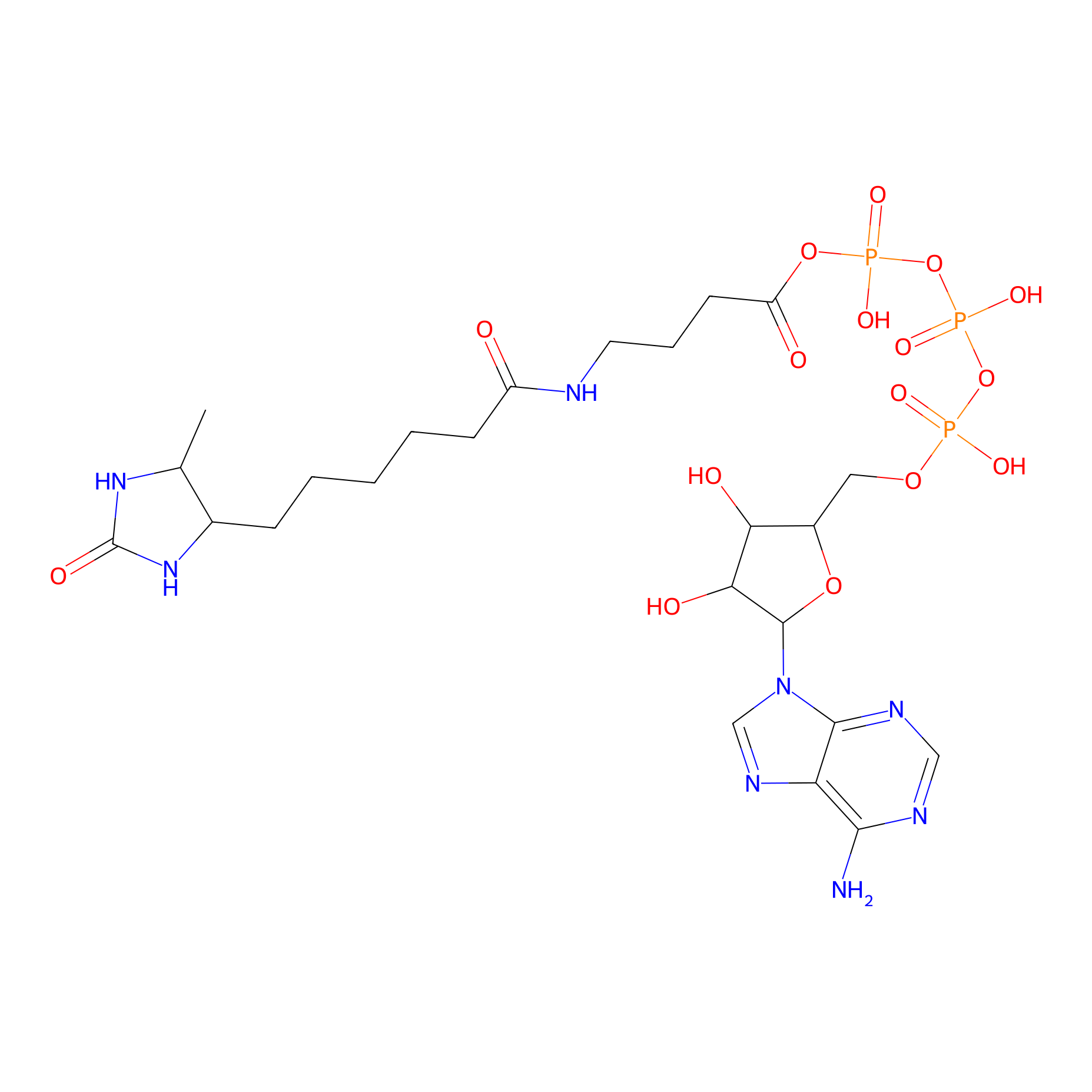 |
K149(0.00); K385(0.00); K83(0.00); K85(0.00) | LDD0199 | [13] | |
|
4-Iodoacetamidophenylacetylene Probe Info |
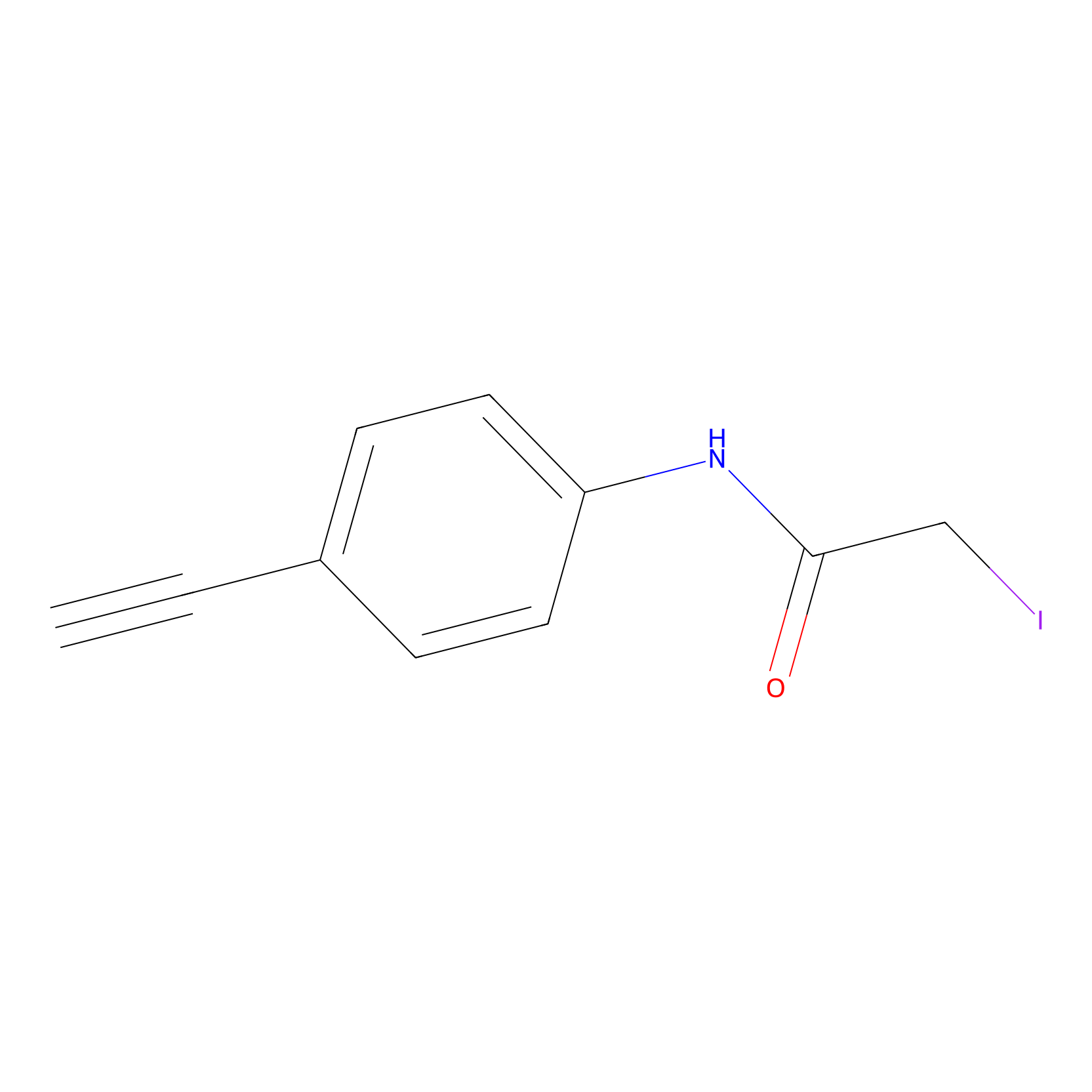 |
C273(0.00); C181(0.00); C261(0.00); C218(0.00) | LDD0038 | [14] | |
|
IA-alkyne Probe Info |
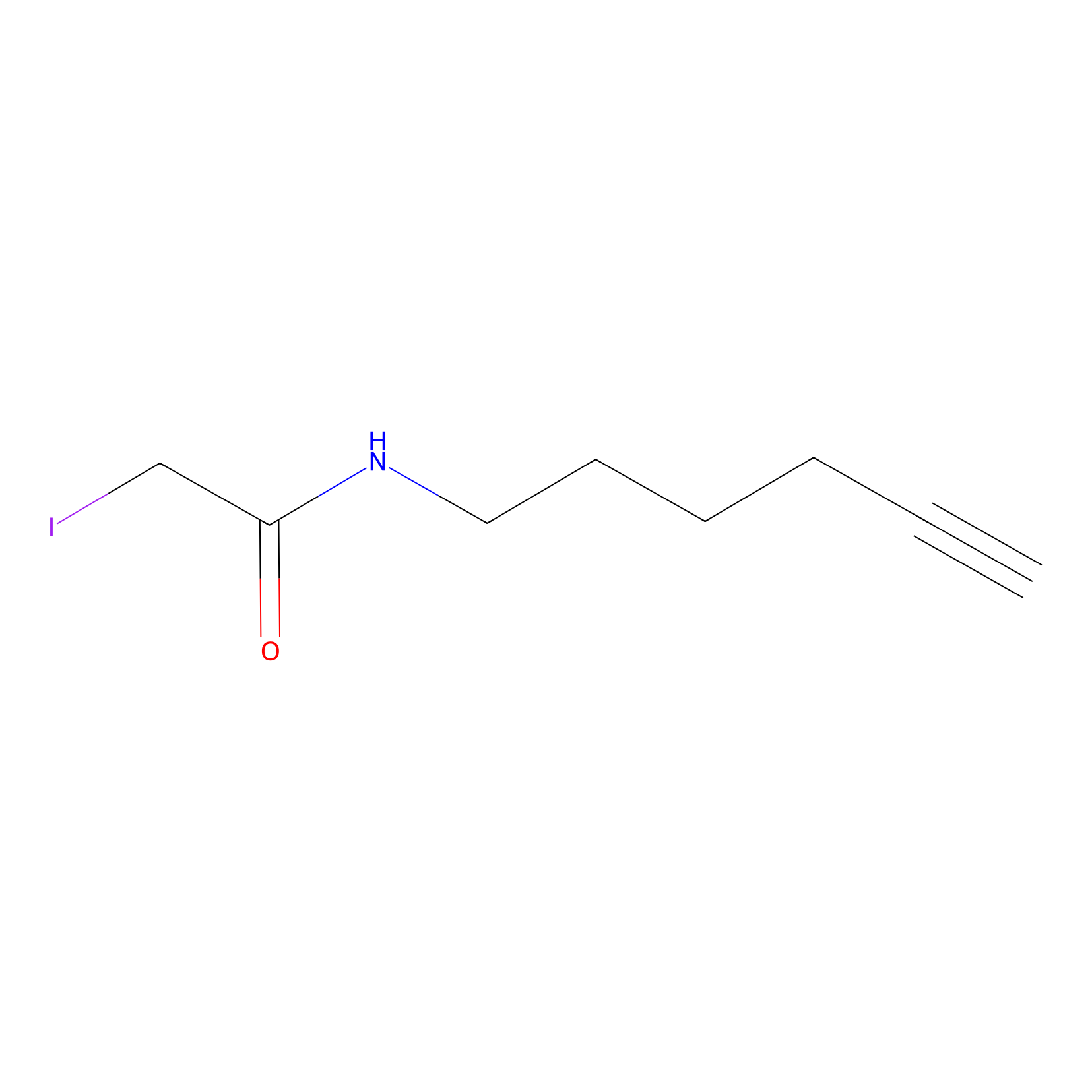 |
C261(0.00); C273(0.00); C218(0.00) | LDD0032 | [15] | |
|
Lodoacetamide azide Probe Info |
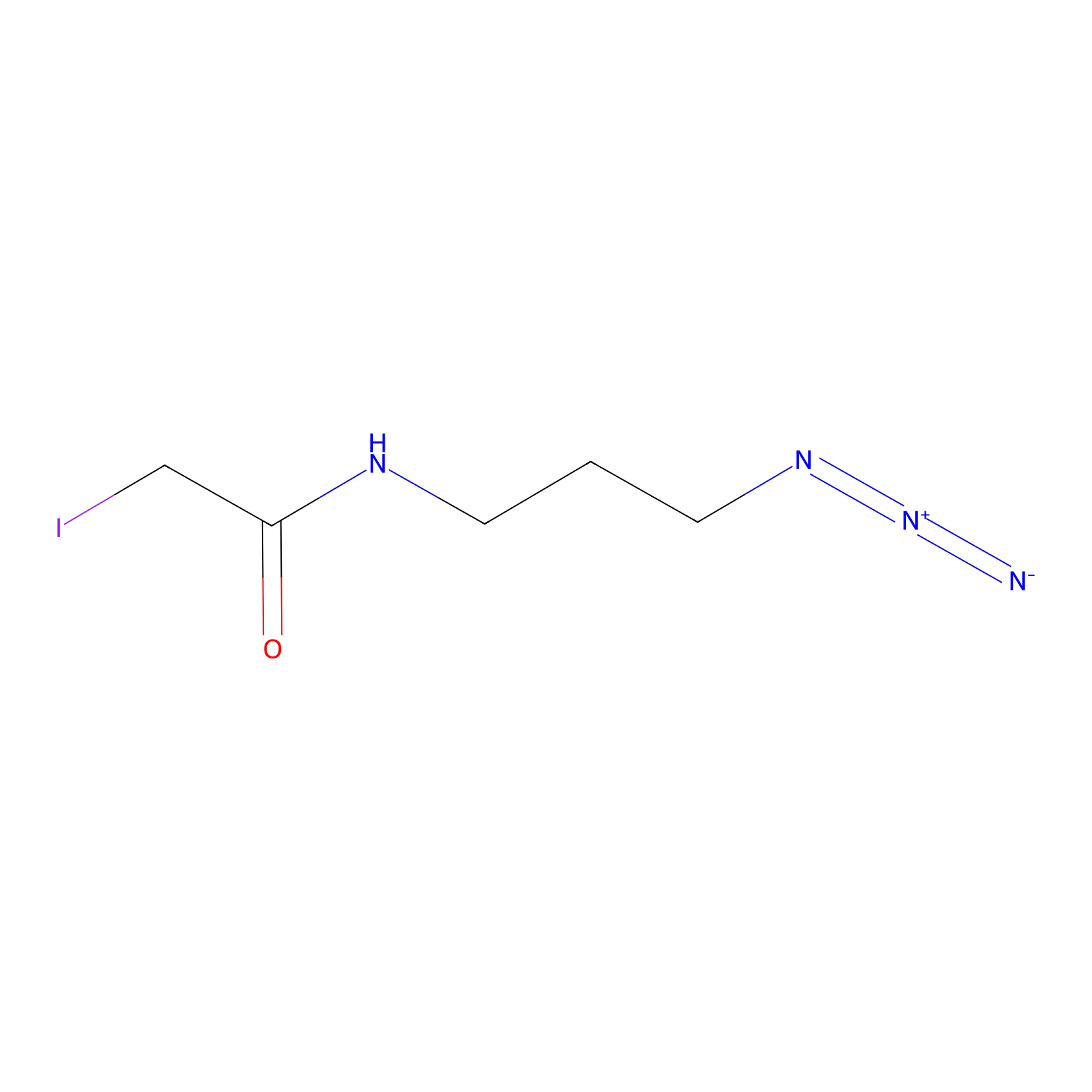 |
C273(0.00); C181(0.00); C261(0.00); C218(0.00) | LDD0037 | [14] | |
|
ATP probe Probe Info |
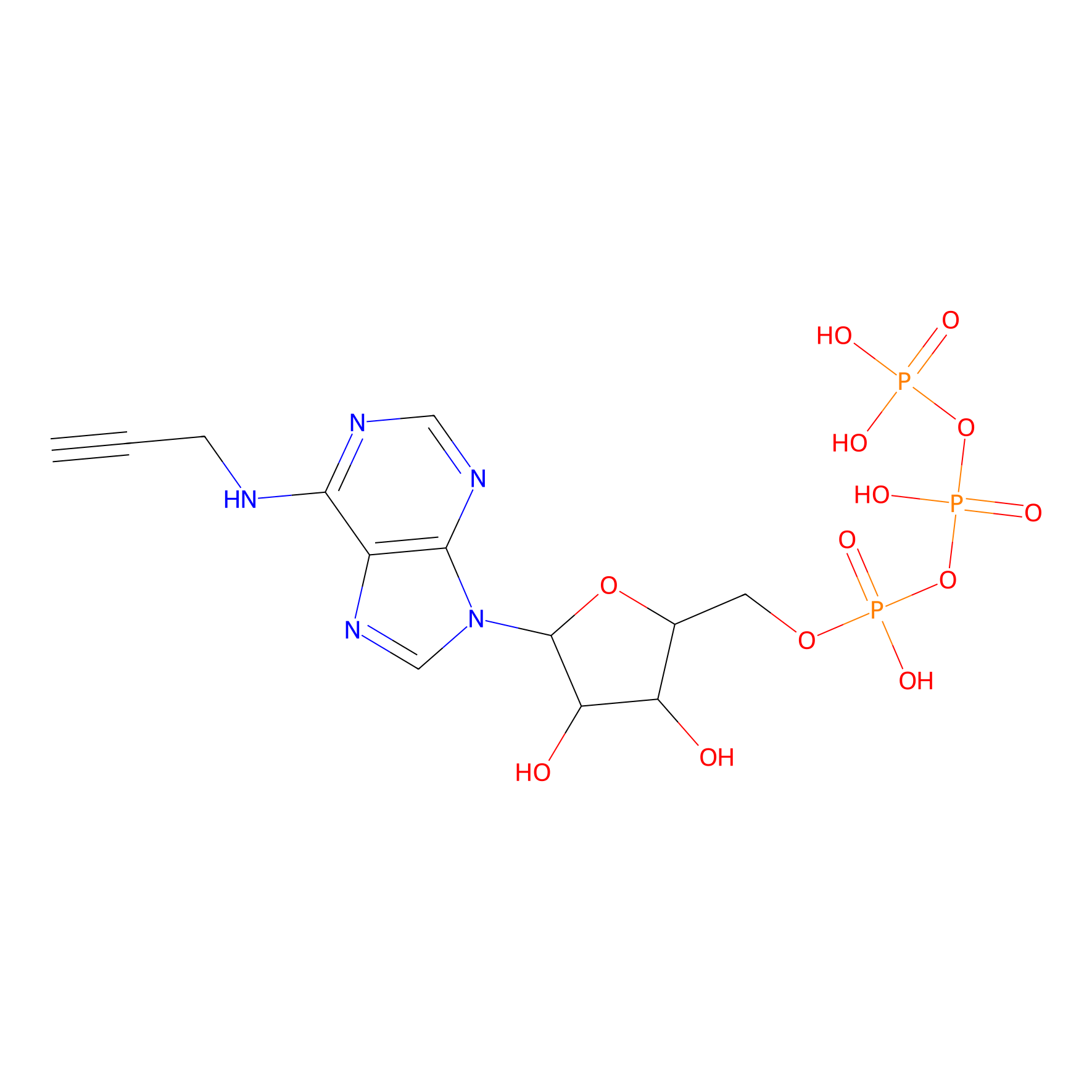 |
N.A. | LDD0035 | [16] | |
|
IPM Probe Info |
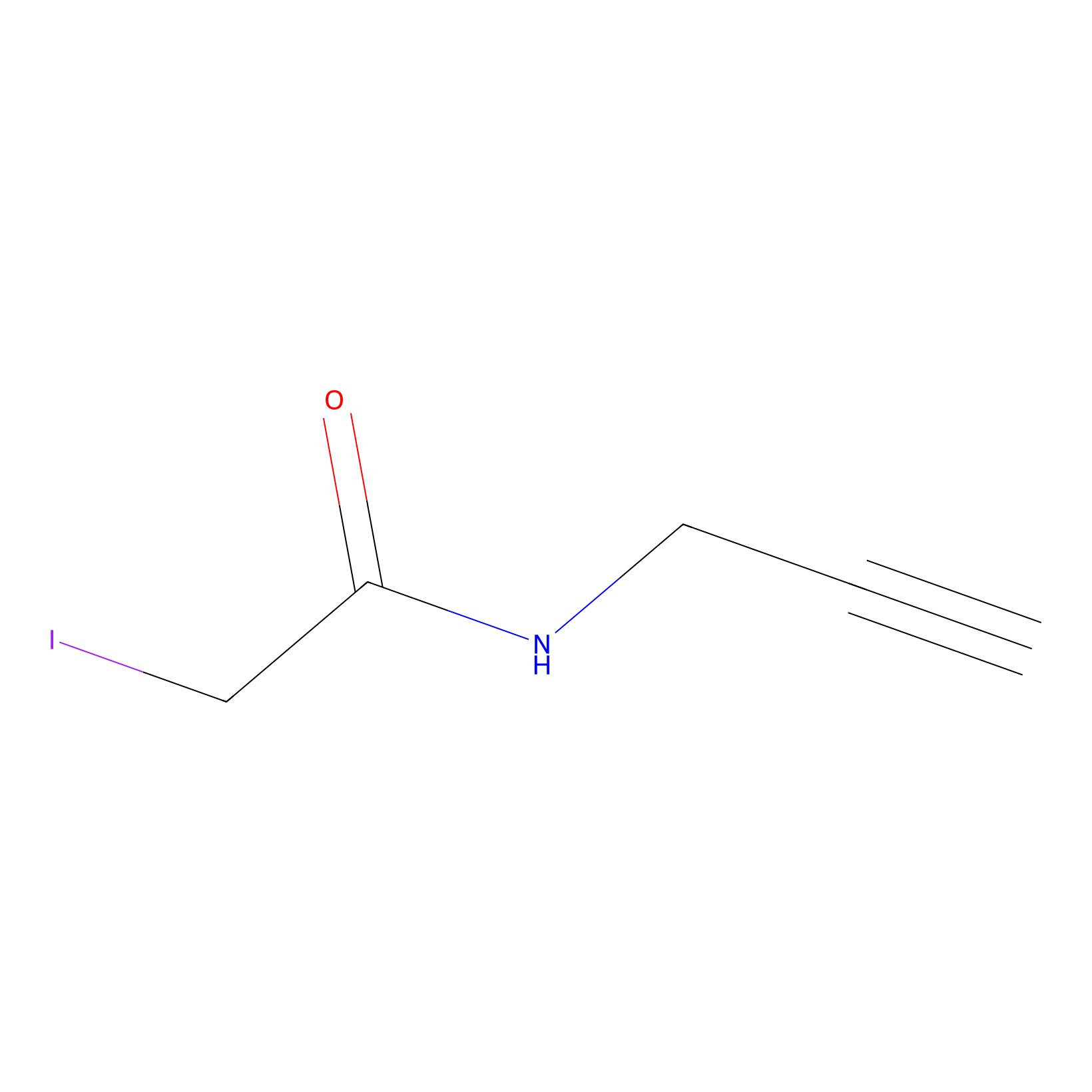 |
N.A. | LDD0025 | [17] | |
|
JW-RF-010 Probe Info |
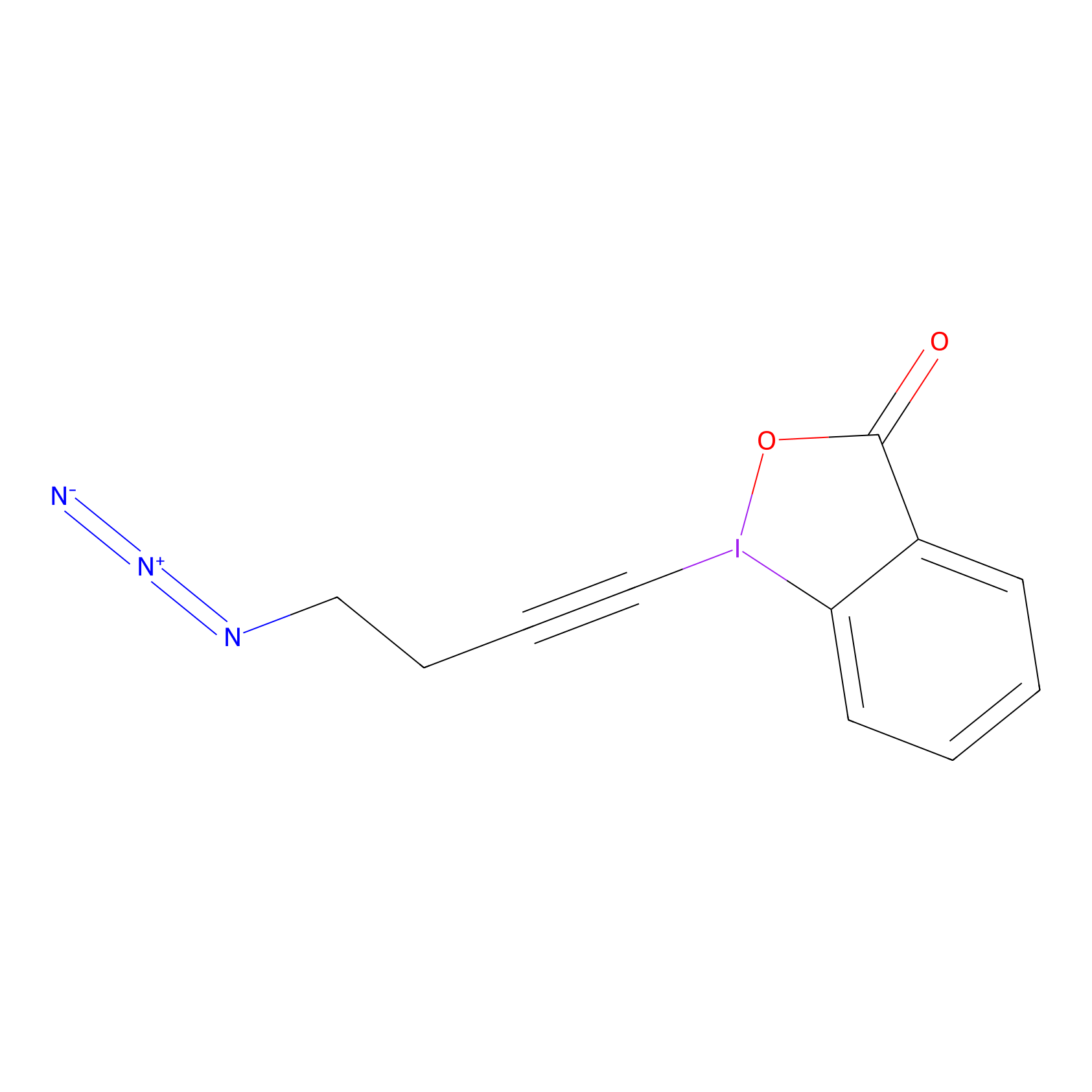 |
C41(0.00); C273(0.00) | LDD0026 | [17] | |
|
NAIA_4 Probe Info |
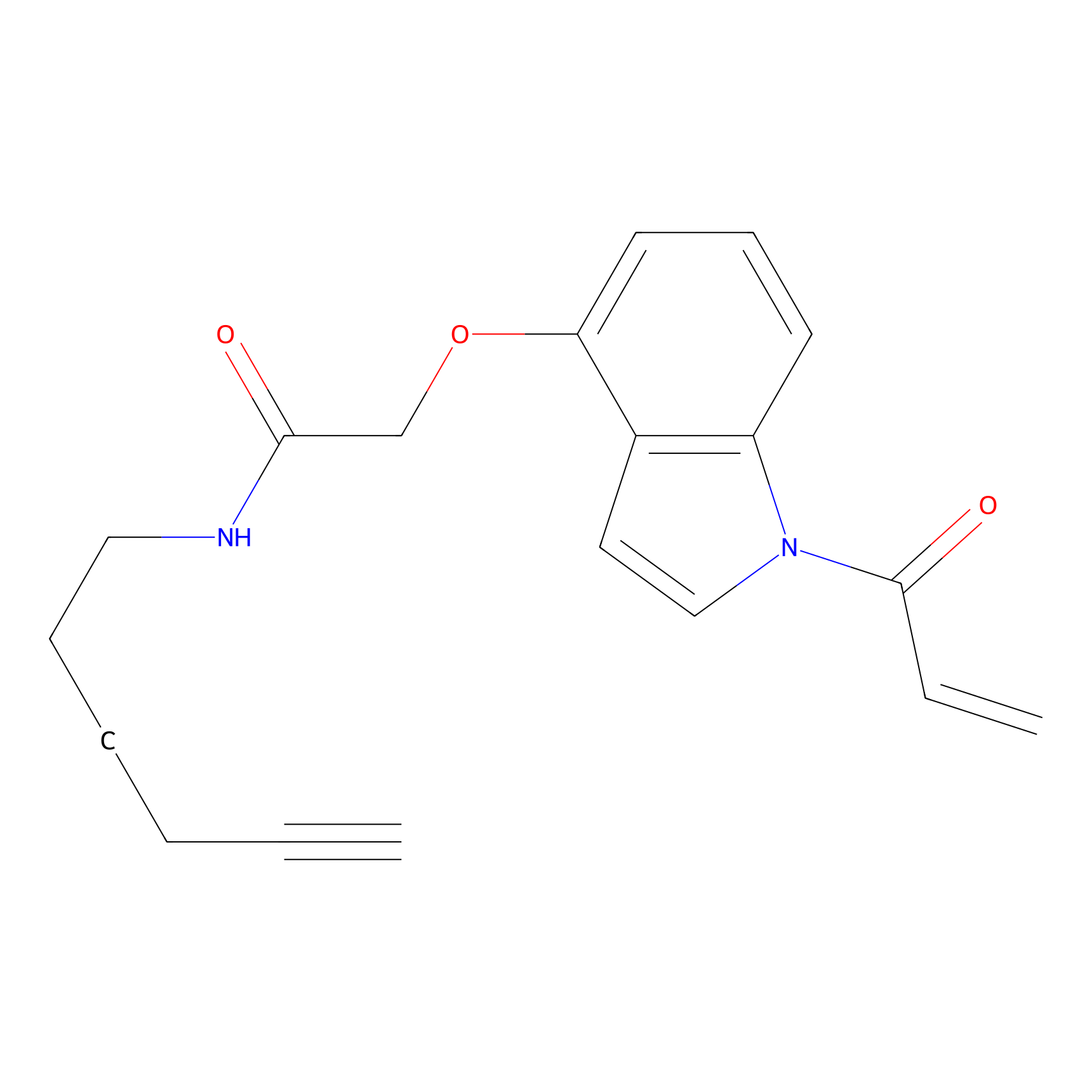 |
C181(0.00); C222(0.00) | LDD2226 | [18] | |
|
TFBX Probe Info |
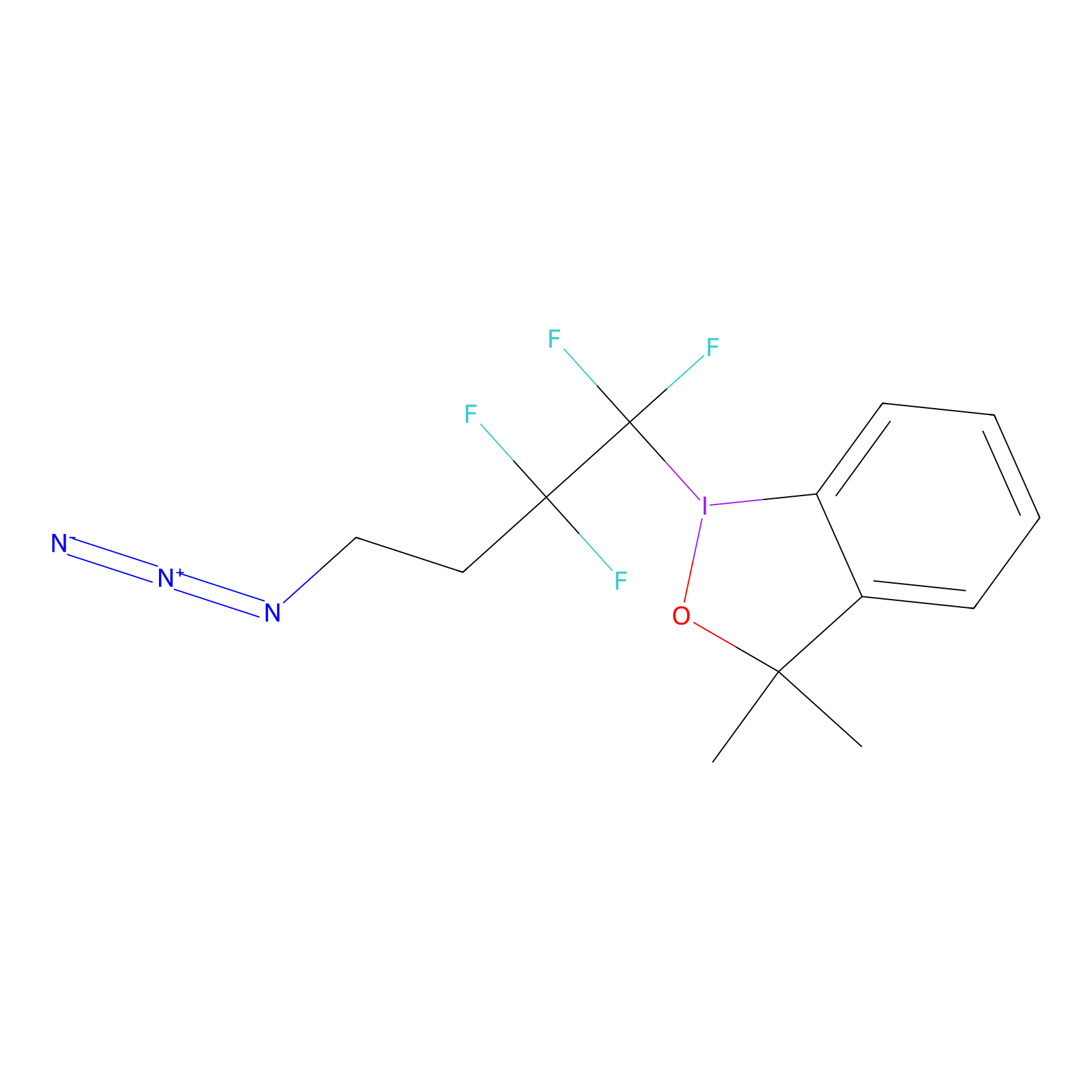 |
C273(0.00); C218(0.00) | LDD0027 | [17] | |
|
WYneN Probe Info |
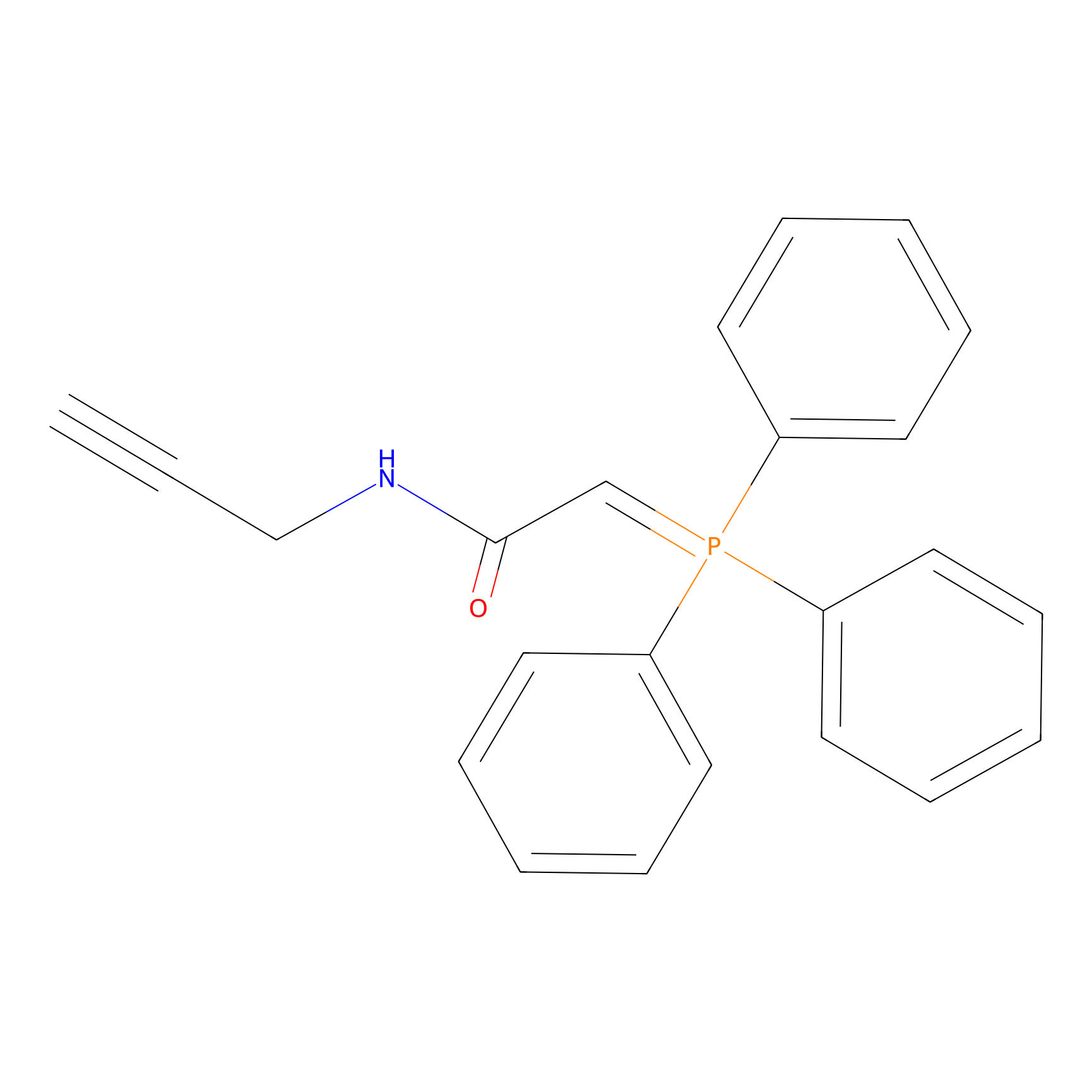 |
N.A. | LDD0021 | [19] | |
|
NHS Probe Info |
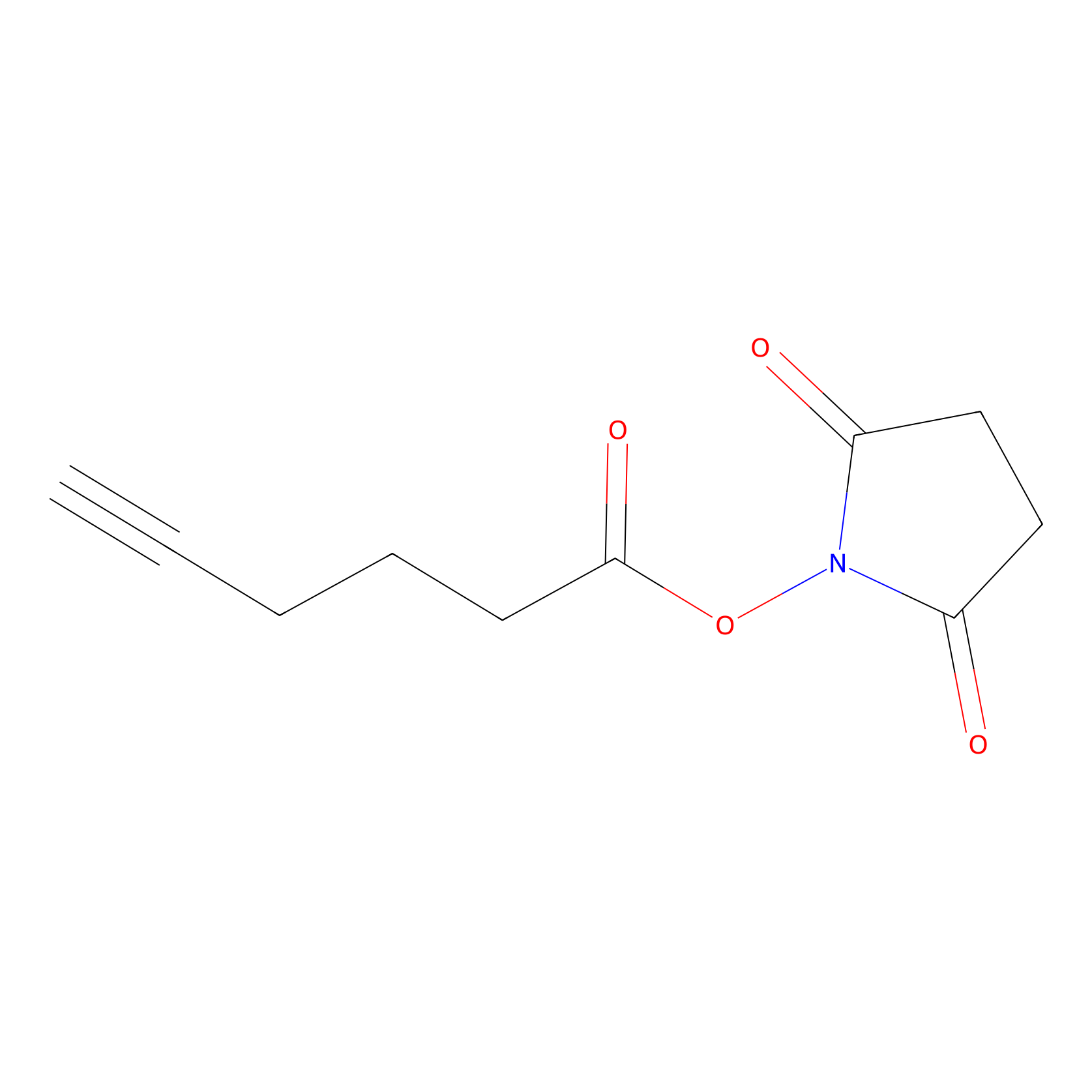 |
K321(0.00); K77(0.00); K244(0.00); K277(0.00) | LDD0010 | [19] | |
|
SF Probe Info |
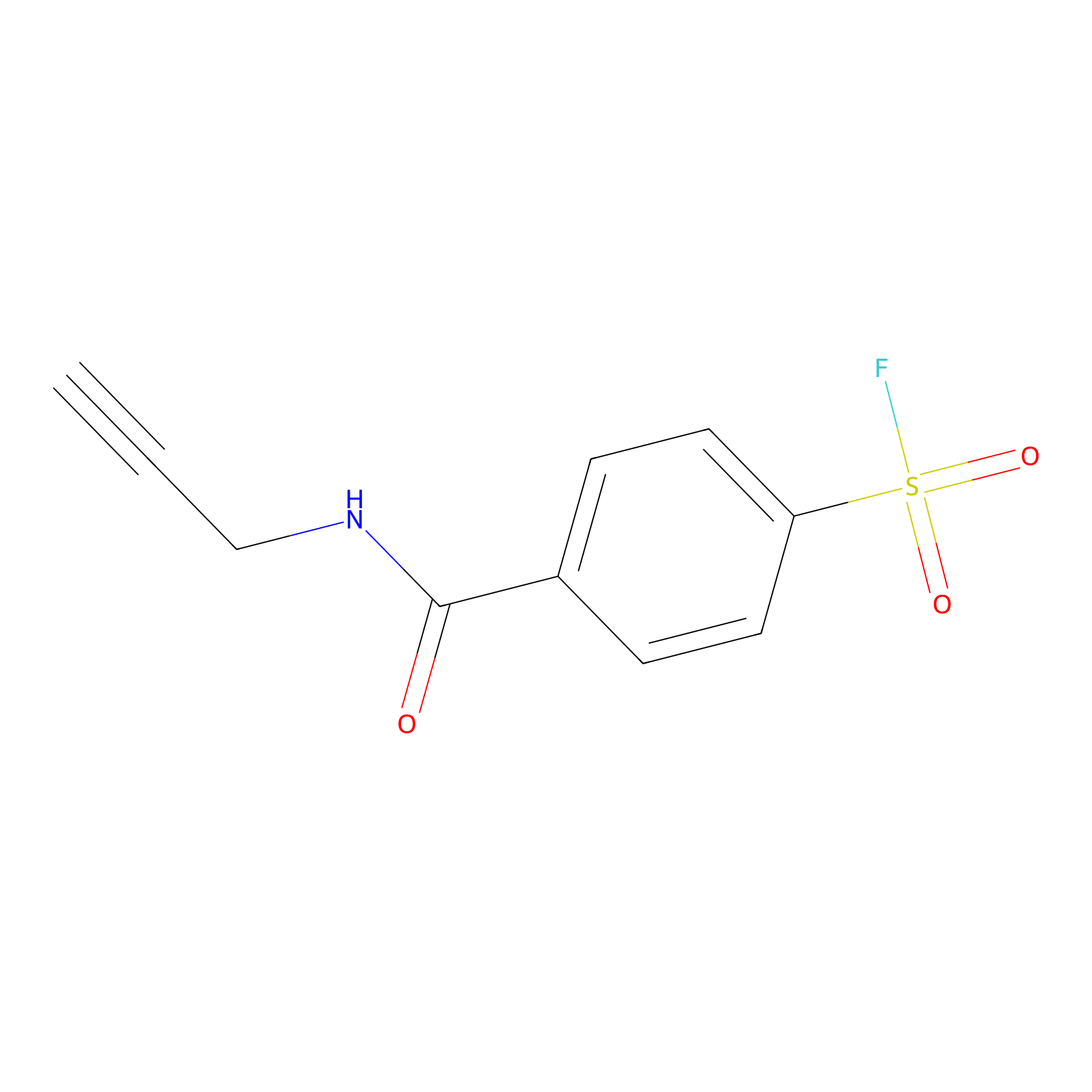 |
Y227(0.00); Y272(0.00); Y287(0.00); Y301(0.00) | LDD0028 | [20] | |
|
Phosphinate-6 Probe Info |
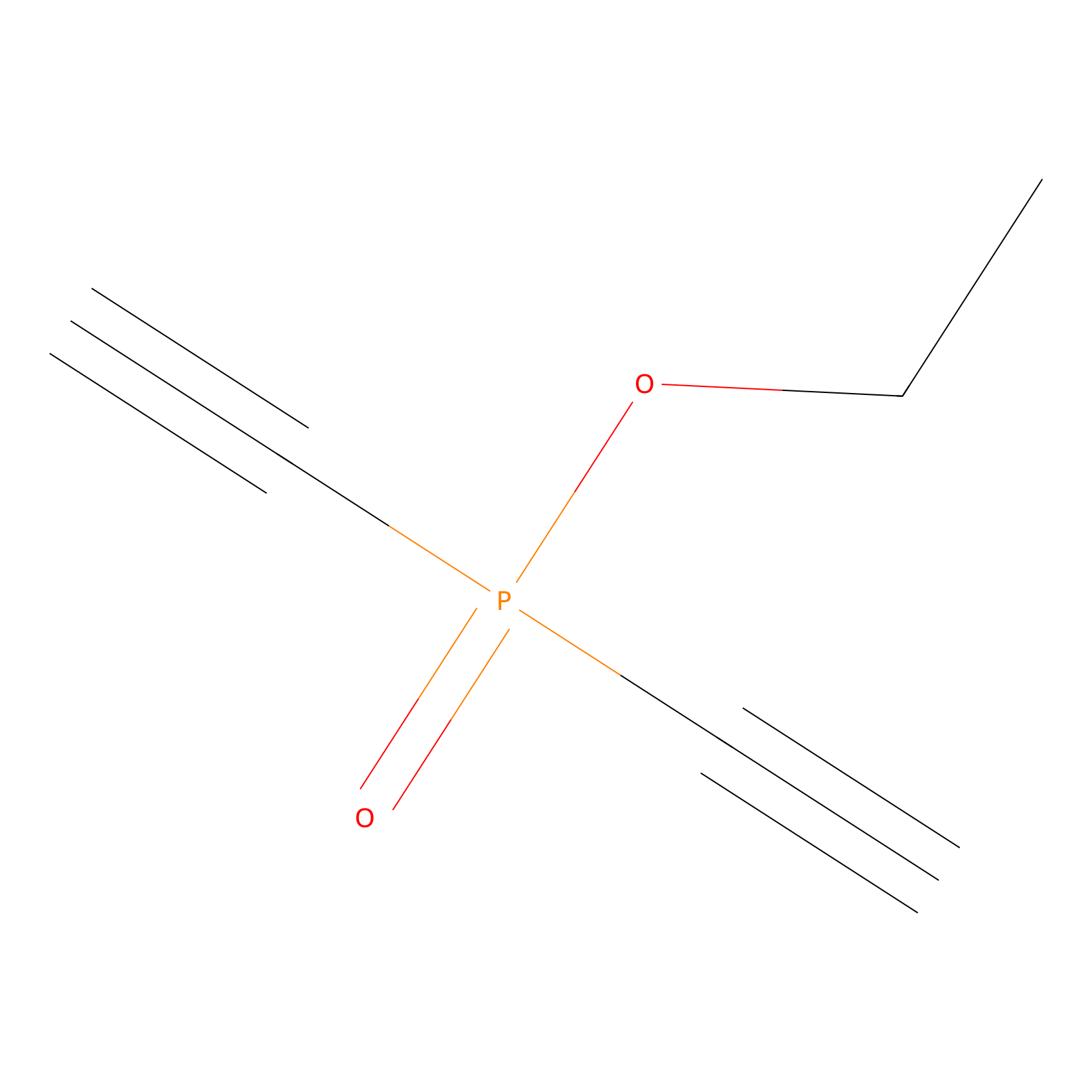 |
C91(0.00); C145(0.00) | LDD0018 | [21] | |
|
1c-yne Probe Info |
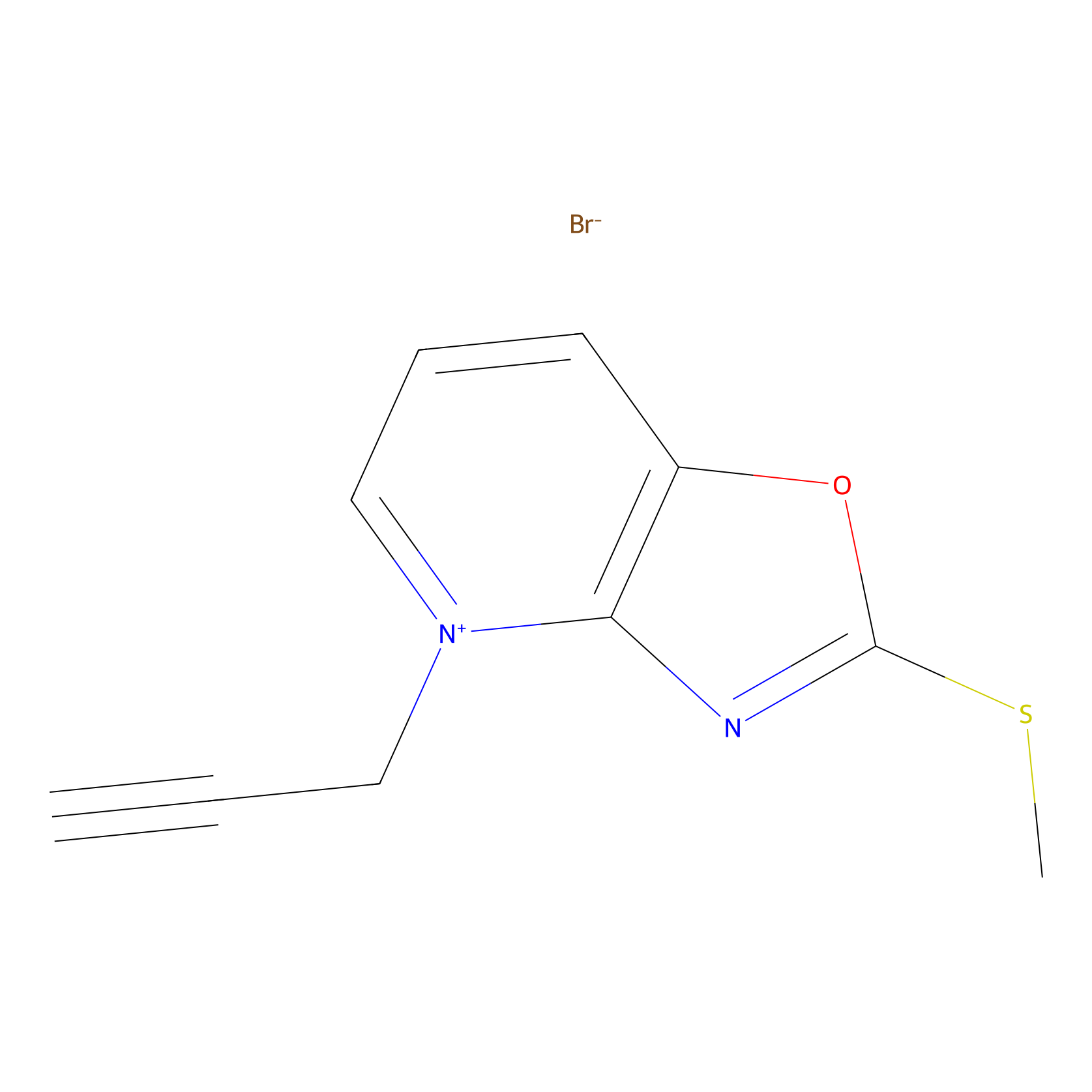 |
N.A. | LDD0228 | [22] | |
|
Acrolein Probe Info |
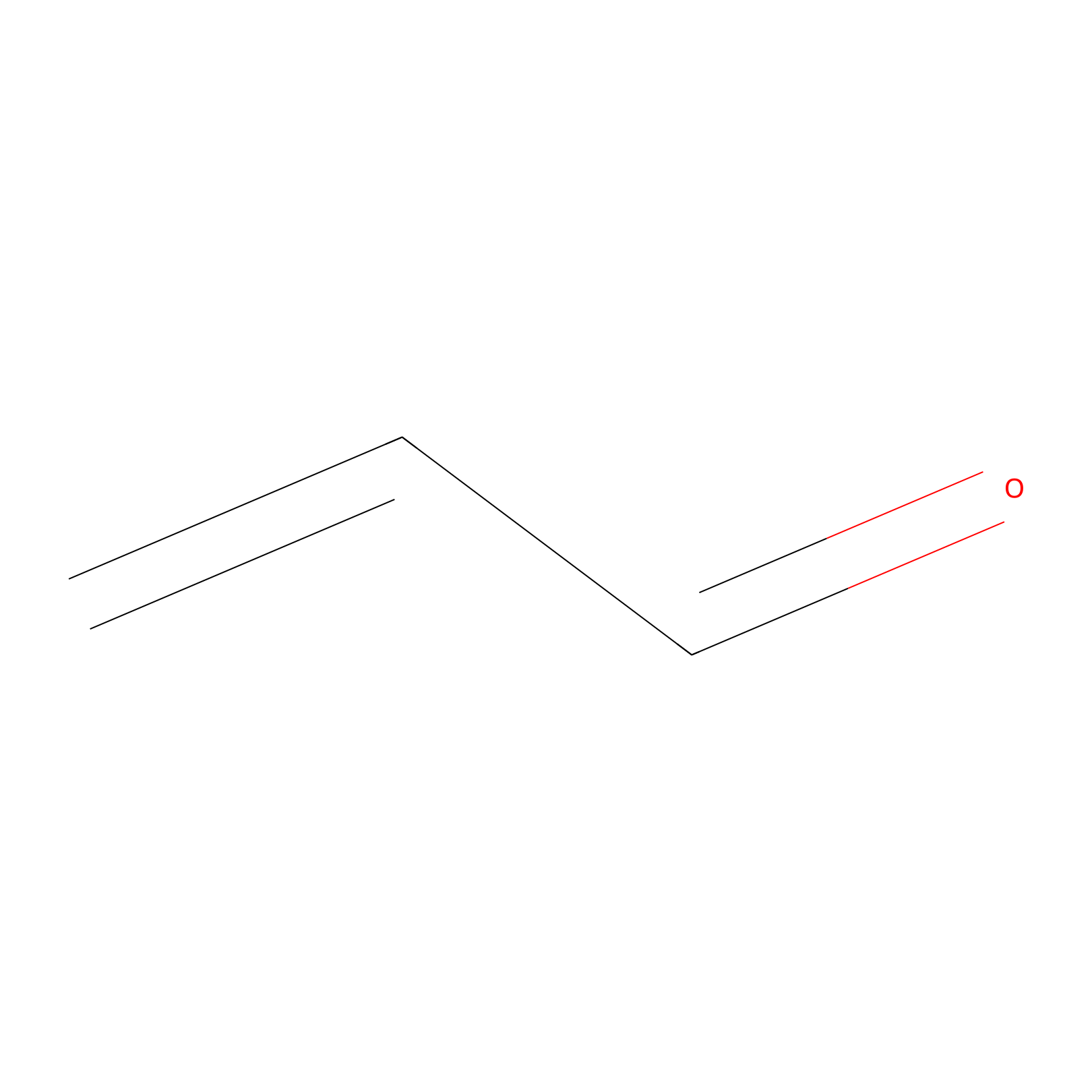 |
C261(0.00); C181(0.00); C218(0.00); C222(0.00) | LDD0217 | [23] | |
|
Cinnamaldehyde Probe Info |
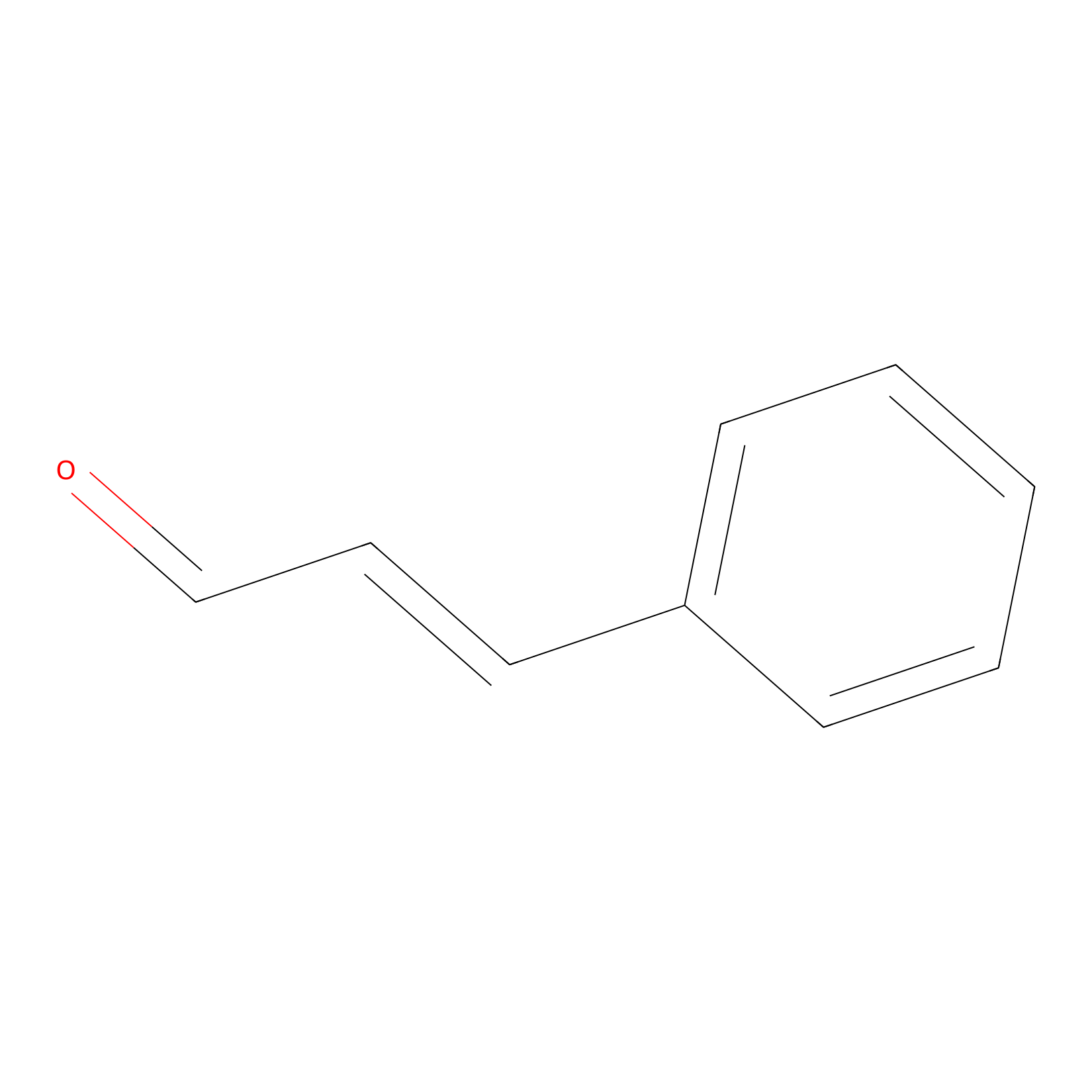 |
C261(0.00); C273(0.00) | LDD0220 | [23] | |
|
Crotonaldehyde Probe Info |
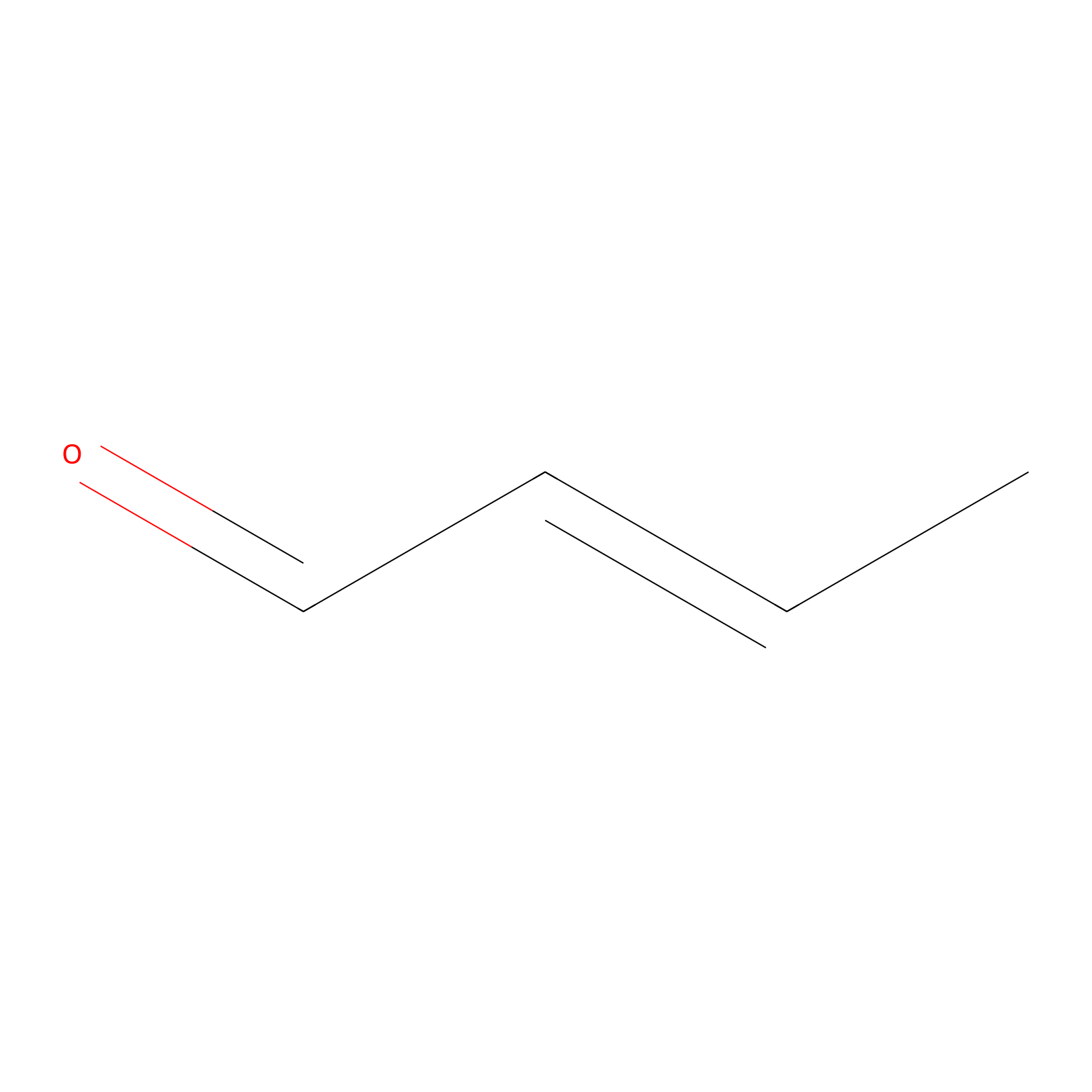 |
H292(0.00); H290(0.00); H121(0.00); C273(0.00) | LDD0219 | [23] | |
|
Methacrolein Probe Info |
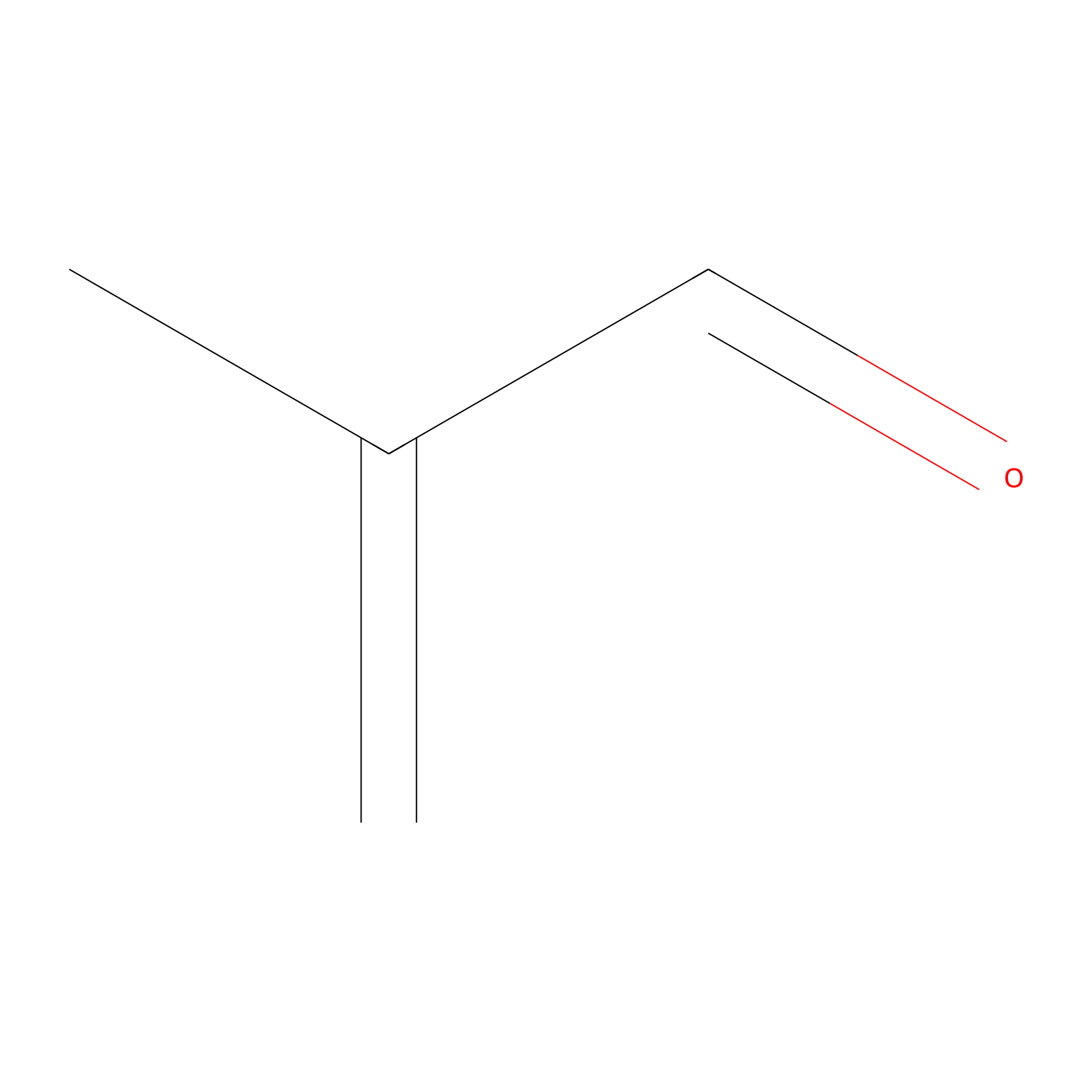 |
C261(0.00); C273(0.00); C41(0.00) | LDD0218 | [23] | |
|
W1 Probe Info |
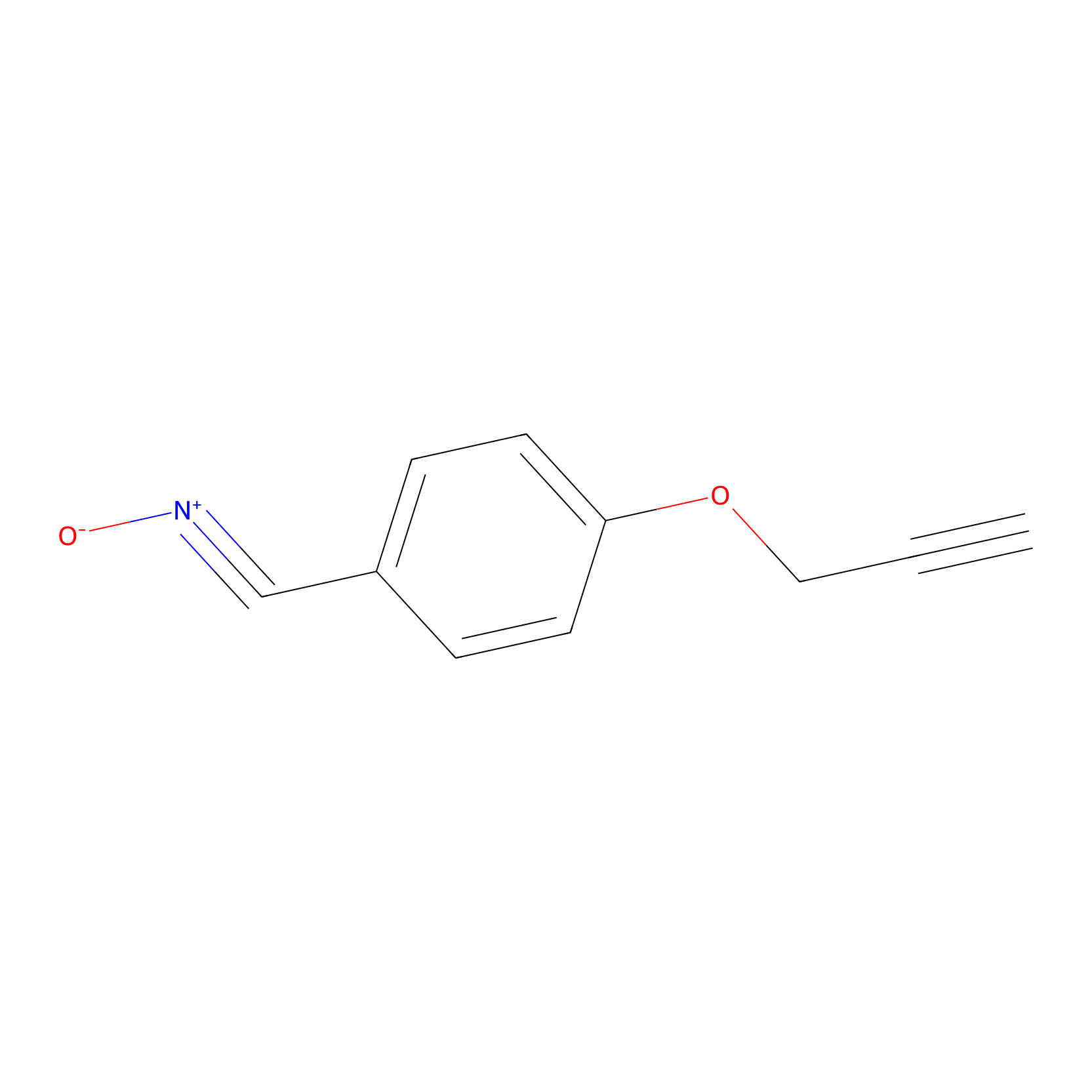 |
N.A. | LDD0236 | [24] | |
|
AOyne Probe Info |
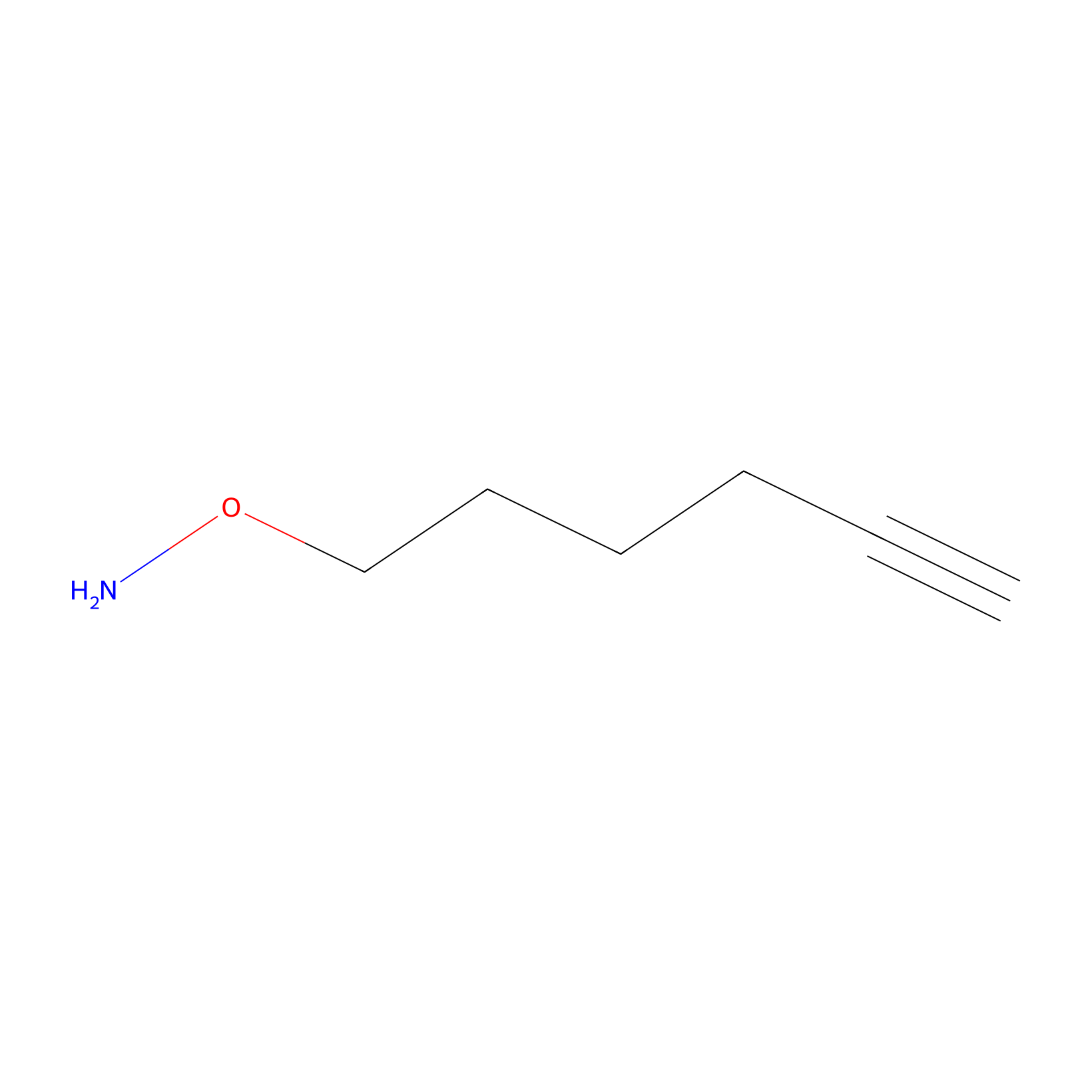 |
7.40 | LDD0443 | [25] | |
|
NAIA_5 Probe Info |
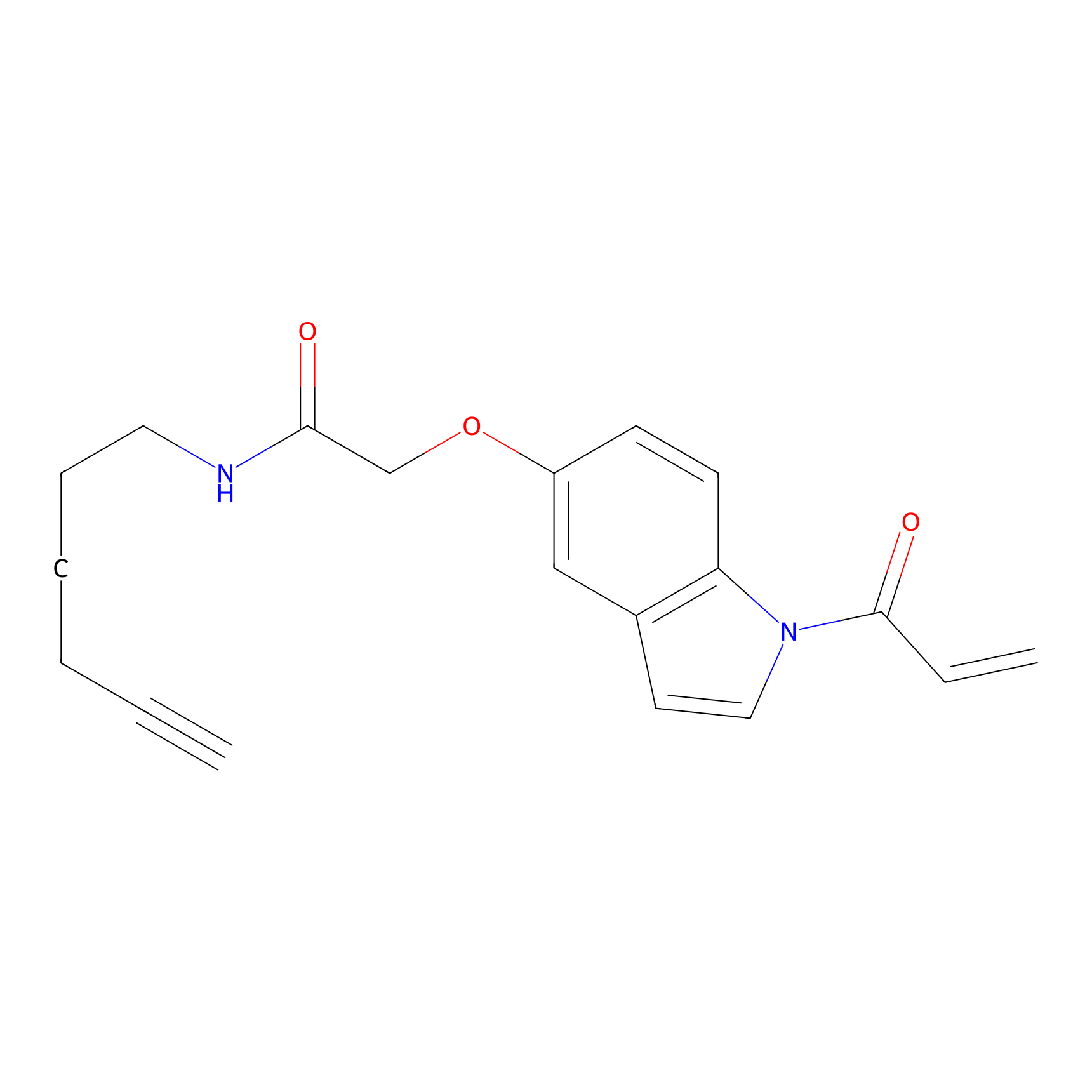 |
C41(0.00); C273(0.00); C145(0.00); C261(0.00) | LDD2223 | [18] | |
|
TER-AC Probe Info |
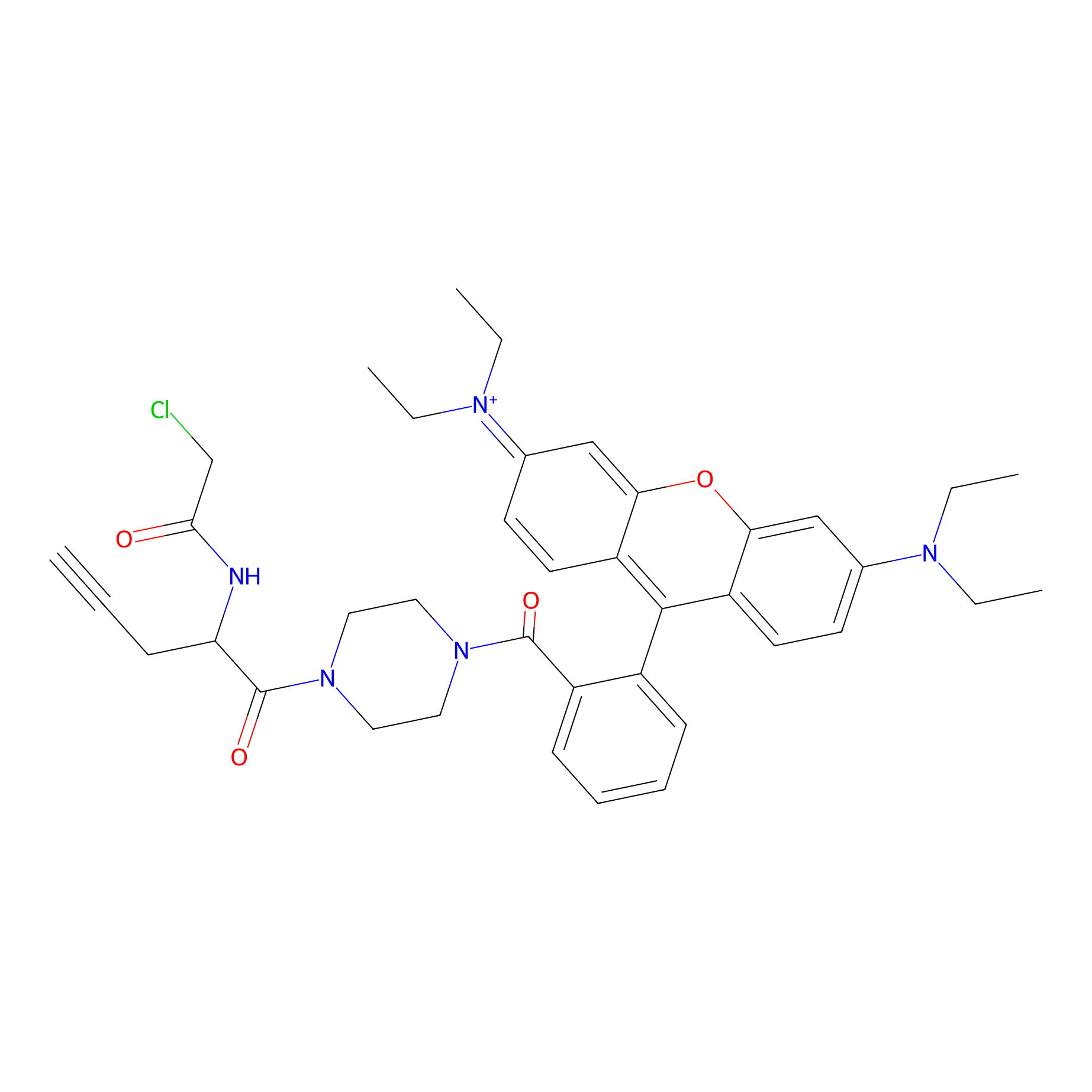 |
N.A. | LDD0426 | [26] | |
|
HHS-465 Probe Info |
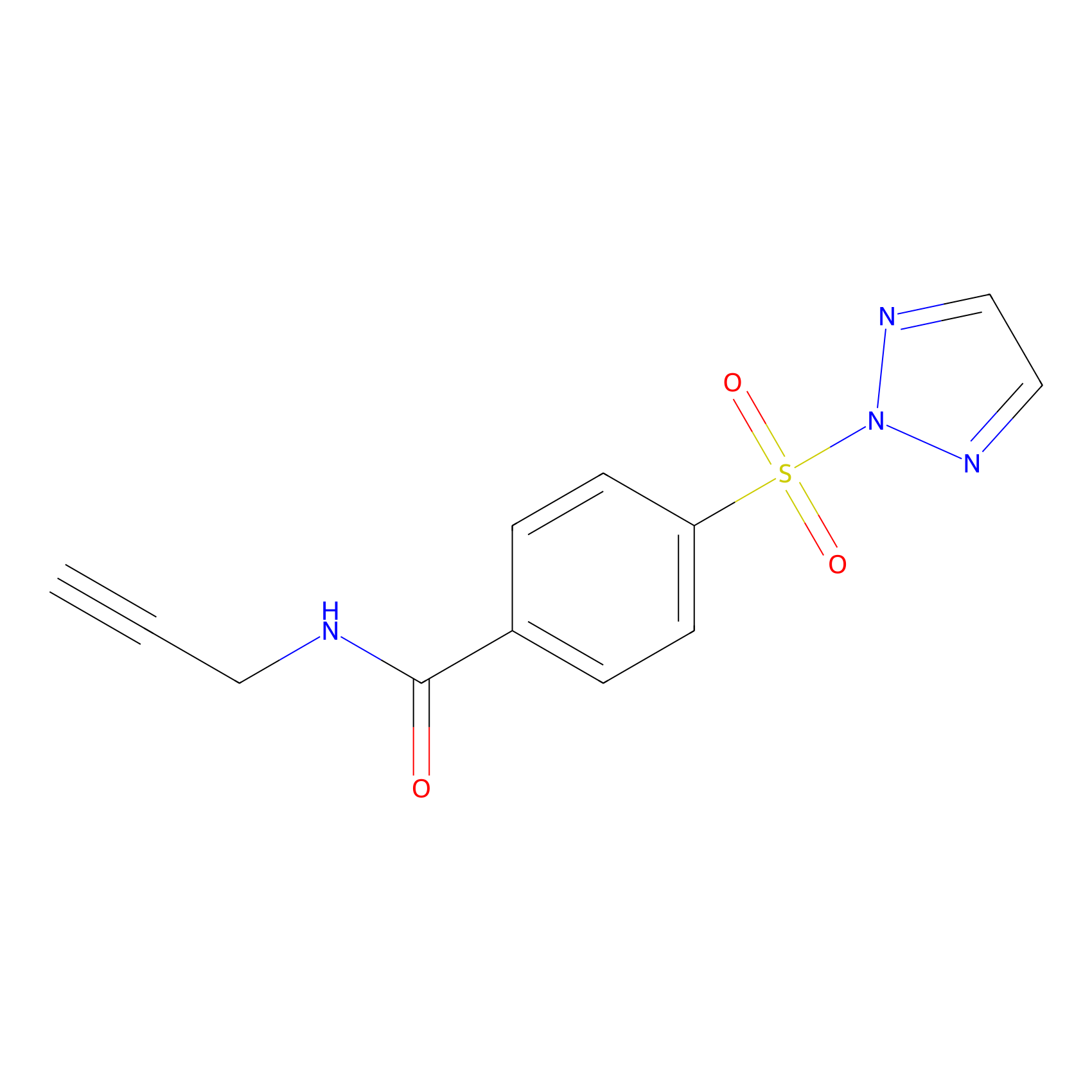 |
N.A. | LDD2240 | [27] | |
PAL-AfBPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
FFF probe13 Probe Info |
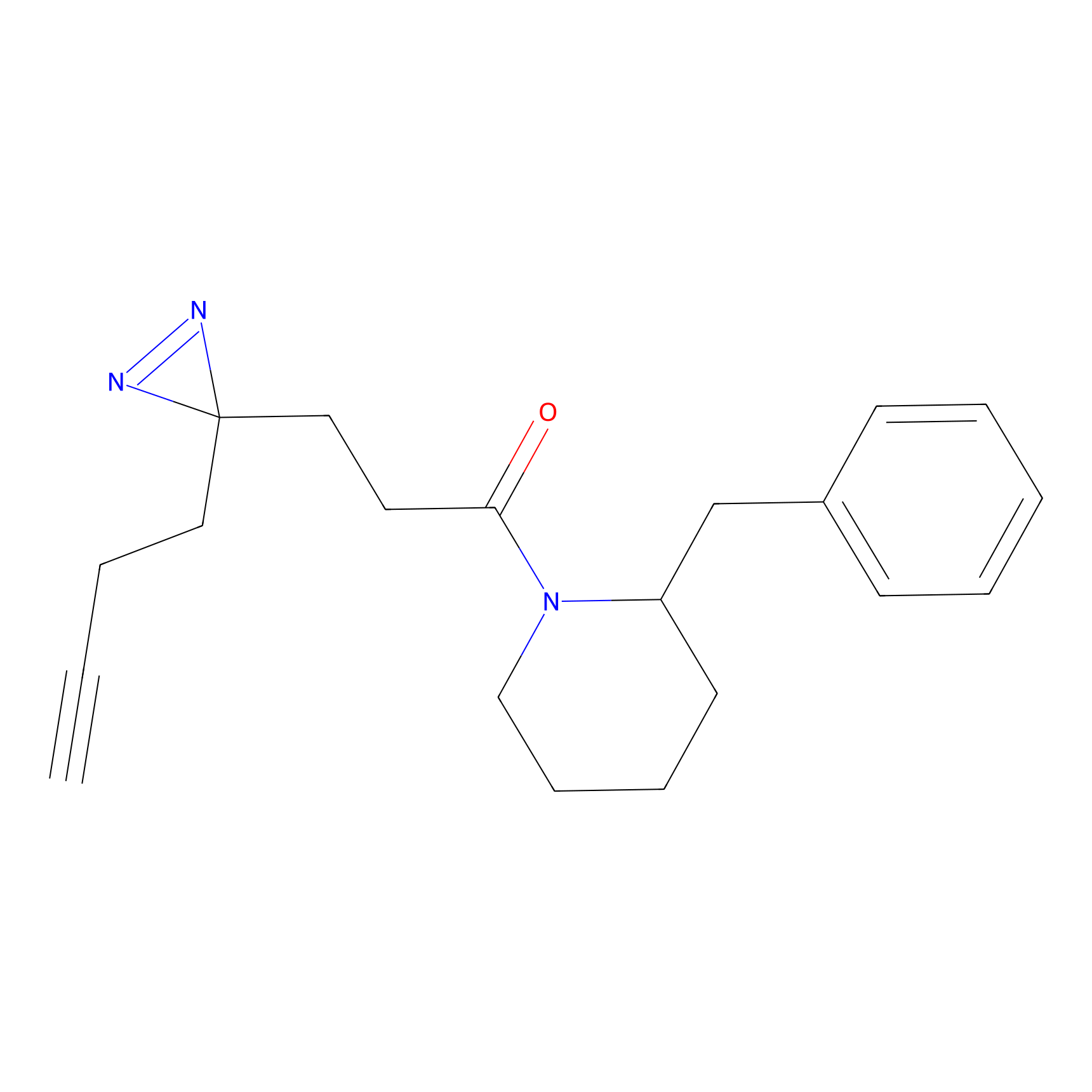 |
20.00 | LDD0475 | [28] | |
|
FFF probe2 Probe Info |
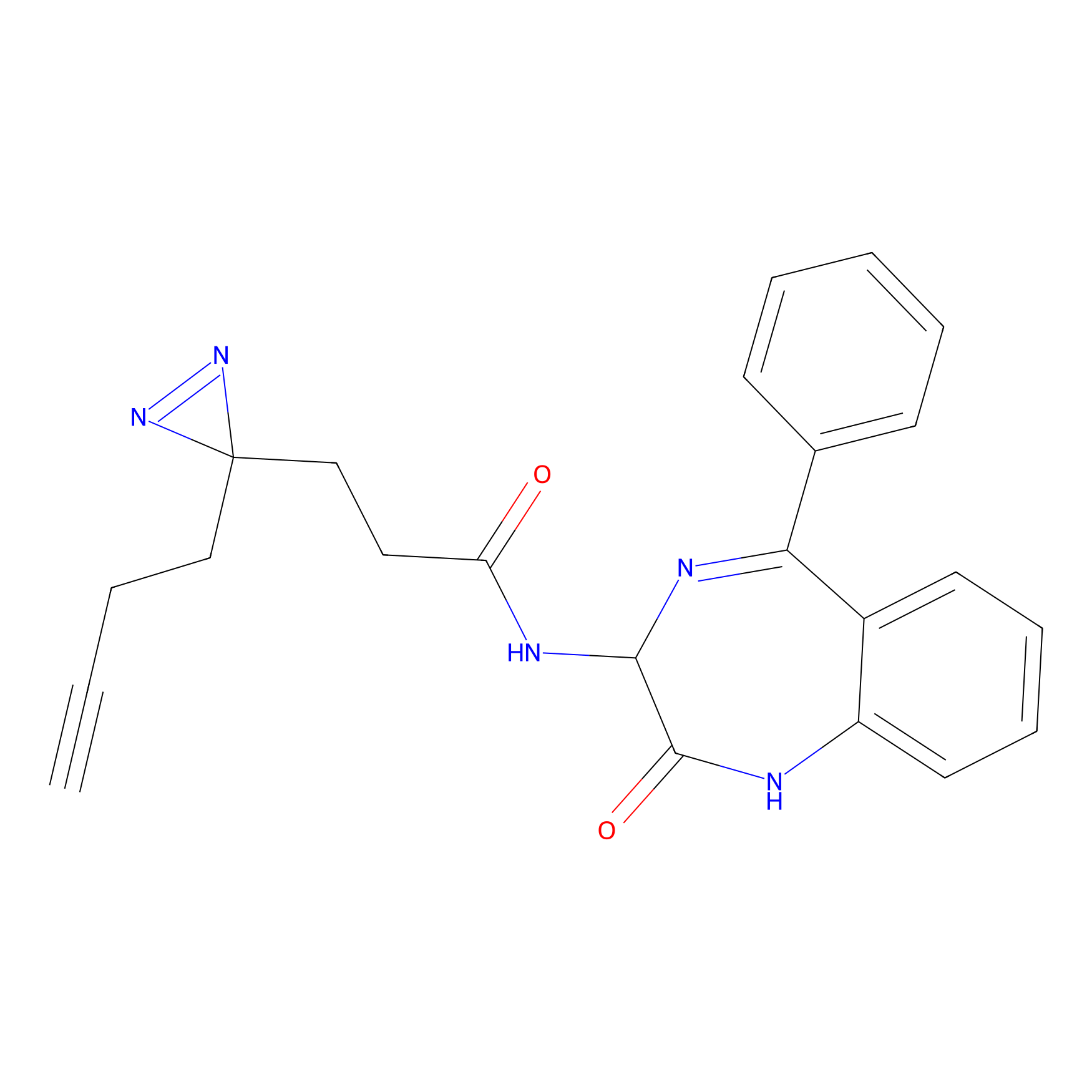 |
5.50 | LDD0463 | [28] | |
|
FFF probe3 Probe Info |
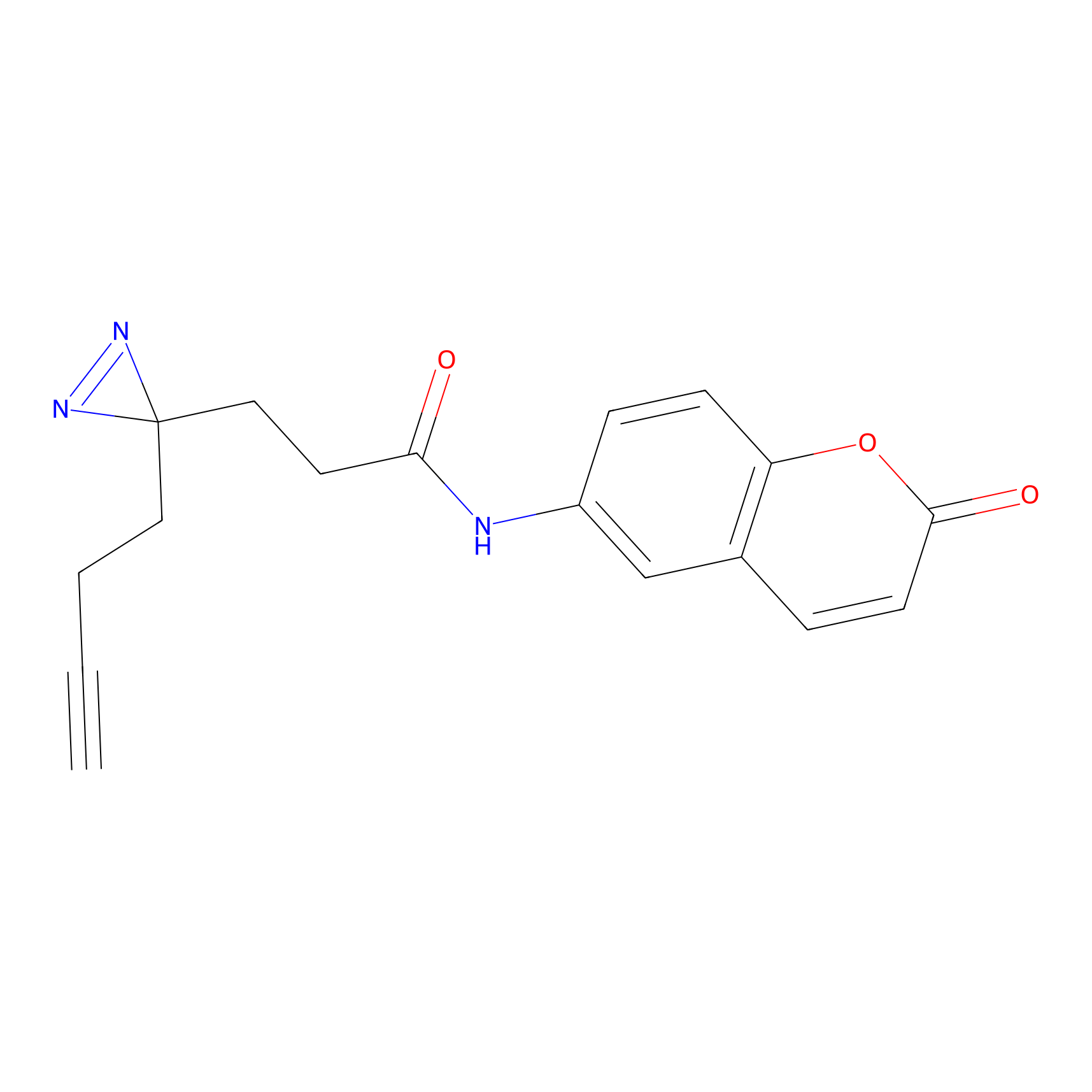 |
5.15 | LDD0465 | [28] | |
|
STS-1 Probe Info |
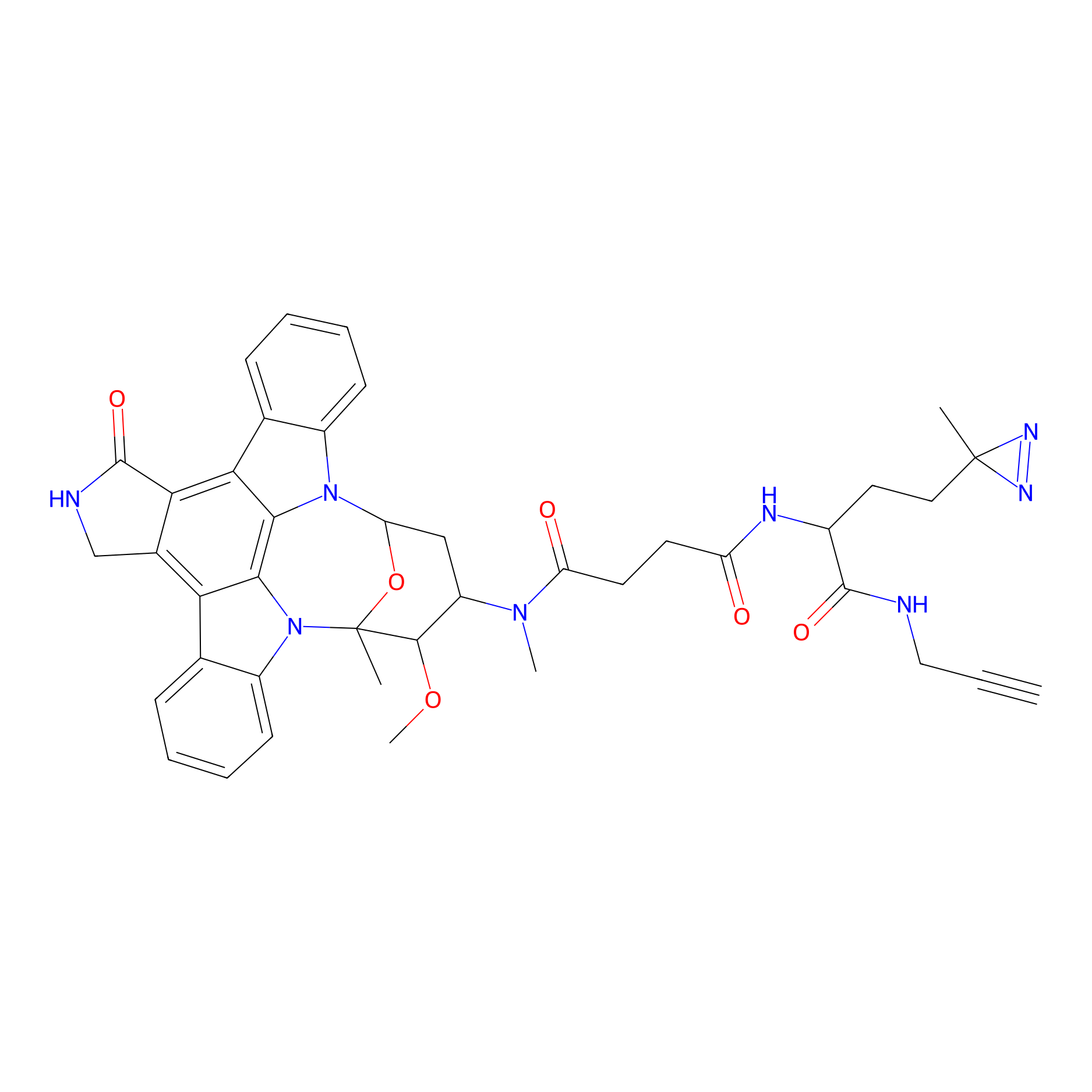 |
N.A. | LDD0136 | [29] | |
|
STS-2 Probe Info |
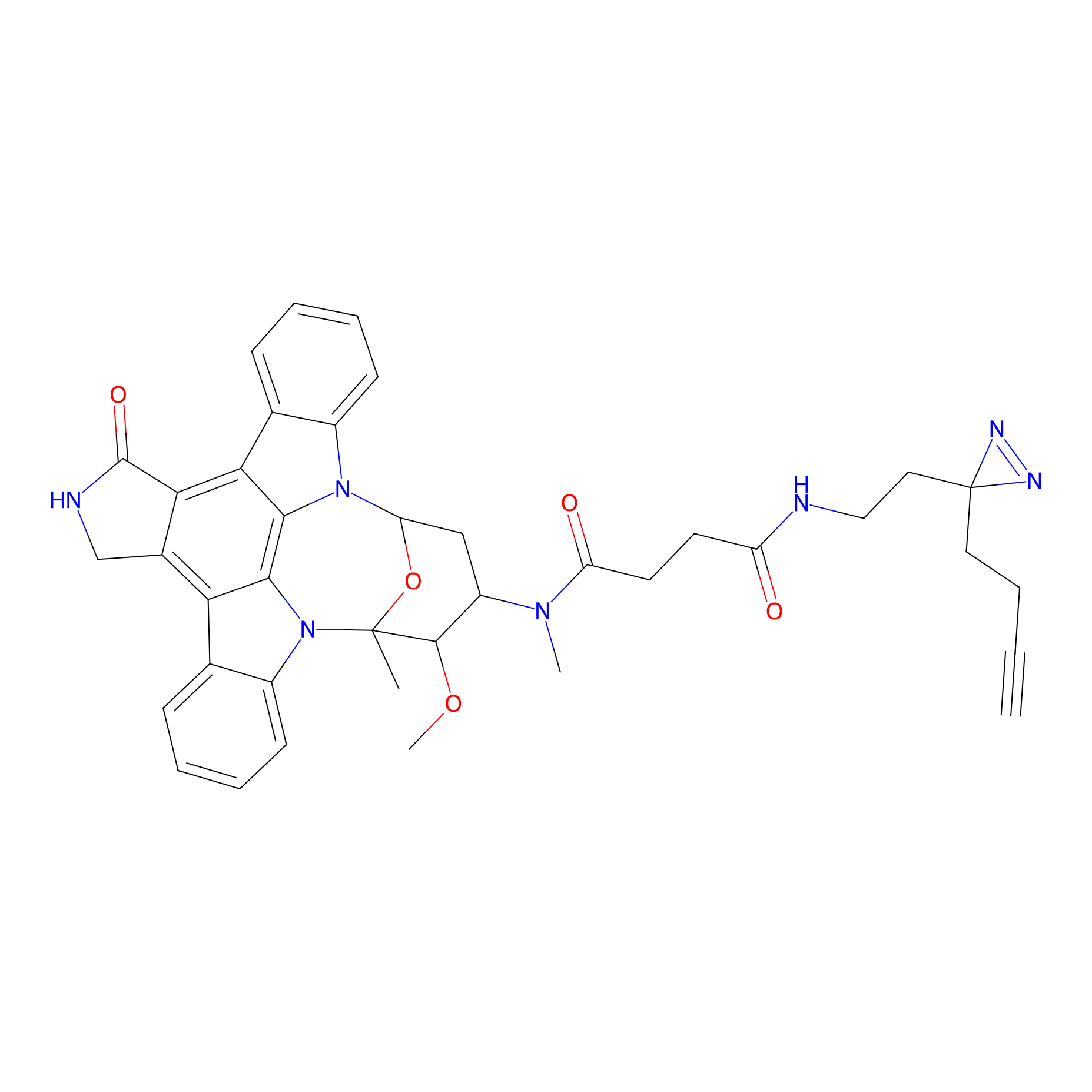 |
N.A. | LDD0138 | [29] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0548 | 1-(4-(Benzo[d][1,3]dioxol-5-ylmethyl)piperazin-1-yl)-2-nitroethan-1-one | MDA-MB-231 | C41(0.78) | LDD2142 | [7] |
| LDCM0519 | 1-(6-methoxy-3,4-dihydroquinolin-1(2H)-yl)-2-nitroethan-1-one | MDA-MB-231 | C41(1.09); C261(0.72) | LDD2112 | [7] |
| LDCM0502 | 1-(Cyanoacetyl)piperidine | MDA-MB-231 | C41(0.76) | LDD2095 | [7] |
| LDCM0537 | 2-Cyano-N,N-dimethylacetamide | MDA-MB-231 | C41(0.97); C273(1.01) | LDD2130 | [7] |
| LDCM0524 | 2-Cyano-N-(2-morpholin-4-yl-ethyl)-acetamide | MDA-MB-231 | C41(1.09); C273(1.06); C261(0.93) | LDD2117 | [7] |
| LDCM0558 | 2-Cyano-N-phenylacetamide | MDA-MB-231 | C41(1.04); C273(1.15) | LDD2152 | [7] |
| LDCM0510 | 3-(4-(Hydroxydiphenylmethyl)piperidin-1-yl)-3-oxopropanenitrile | MDA-MB-231 | C41(1.39) | LDD2103 | [7] |
| LDCM0539 | 3-(4-Isopropylpiperazin-1-yl)-3-oxopropanenitrile | MDA-MB-231 | C41(0.59) | LDD2132 | [7] |
| LDCM0025 | 4SU-RNA | DM93 | C273(6.24) | LDD0170 | [8] |
| LDCM0545 | Acetamide | MDA-MB-231 | C41(0.42); C273(0.81) | LDD2138 | [7] |
| LDCM0520 | AKOS000195272 | MDA-MB-231 | C41(0.77); C273(0.99) | LDD2113 | [7] |
| LDCM0498 | BS-3668 | MDA-MB-231 | C41(0.45); C273(0.82) | LDD2091 | [7] |
| LDCM0108 | Chloroacetamide | HeLa | C181(0.00); C261(0.00); H292(0.00); C218(0.00) | LDD0222 | [23] |
| LDCM0213 | Electrophilic fragment 2 | MDA-MB-231 | C273(2.17); C261(2.00); C41(1.34) | LDD1702 | [7] |
| LDCM0175 | Ethacrynic acid | HeLa | C181(0.88) | LDD2210 | [9] |
| LDCM0625 | F8 | Ramos | C273(0.85); C261(0.70); C181(1.10); 0.73 | LDD2187 | [30] |
| LDCM0572 | Fragment10 | Ramos | C273(0.64); C261(0.54); C181(0.54); 0.68 | LDD2189 | [30] |
| LDCM0573 | Fragment11 | Ramos | C273(0.06); C261(0.28); C181(0.12); 0.22 | LDD2190 | [30] |
| LDCM0574 | Fragment12 | Ramos | C273(0.70); C261(0.79); C181(0.55); 0.72 | LDD2191 | [30] |
| LDCM0575 | Fragment13 | Ramos | C273(1.08); C261(1.23); C181(0.98); 0.41 | LDD2192 | [30] |
| LDCM0576 | Fragment14 | Ramos | C273(0.68); C261(0.66); C181(0.64); 0.89 | LDD2193 | [30] |
| LDCM0579 | Fragment20 | Ramos | C273(0.56); C261(0.56); C181(0.56); 0.40 | LDD2194 | [30] |
| LDCM0580 | Fragment21 | Ramos | C273(0.88); C261(1.08); C181(0.88); 0.87 | LDD2195 | [30] |
| LDCM0582 | Fragment23 | Ramos | C273(0.46); C261(0.52); C181(0.74); 0.64 | LDD2196 | [30] |
| LDCM0578 | Fragment27 | Ramos | C273(1.00); C261(1.09); C181(1.07); 1.49 | LDD2197 | [30] |
| LDCM0586 | Fragment28 | Ramos | C273(0.85); C261(0.34); C181(1.30); 0.03 | LDD2198 | [30] |
| LDCM0588 | Fragment30 | Ramos | C273(1.13); C261(1.16); C181(0.70); 0.54 | LDD2199 | [30] |
| LDCM0589 | Fragment31 | Ramos | C273(0.88); C261(1.15); C181(0.78); 1.05 | LDD2200 | [30] |
| LDCM0590 | Fragment32 | Ramos | C273(0.47); C261(0.60); C181(0.47); 0.65 | LDD2201 | [30] |
| LDCM0468 | Fragment33 | Ramos | C273(0.95); C261(0.88); C181(0.65); 0.70 | LDD2202 | [30] |
| LDCM0596 | Fragment38 | Ramos | C273(0.97); C261(0.94); C181(0.93) | LDD2203 | [30] |
| LDCM0566 | Fragment4 | Ramos | C273(0.84); C261(0.78); C181(0.66); 0.64 | LDD2184 | [30] |
| LDCM0610 | Fragment52 | Ramos | C273(1.28); C261(0.98); C181(0.83); 1.21 | LDD2204 | [30] |
| LDCM0614 | Fragment56 | Ramos | C273(1.02); C261(1.10); C181(0.89); 0.62 | LDD2205 | [30] |
| LDCM0569 | Fragment7 | Ramos | C273(0.64); C261(0.55); C181(0.59); 0.38 | LDD2186 | [30] |
| LDCM0571 | Fragment9 | Ramos | C273(0.51); C261(0.57); C181(0.45); 0.42 | LDD2188 | [30] |
| LDCM0107 | IAA | HeLa | C261(0.00); H292(0.00); C218(0.00); C273(0.00) | LDD0221 | [23] |
| LDCM0123 | JWB131 | DM93 | Y156(0.59); Y227(0.71) | LDD0285 | [11] |
| LDCM0124 | JWB142 | DM93 | Y156(0.87); Y227(0.05) | LDD0286 | [11] |
| LDCM0125 | JWB146 | DM93 | Y156(0.50); Y227(0.05) | LDD0287 | [11] |
| LDCM0126 | JWB150 | DM93 | Y156(1.51); Y227(4.55) | LDD0288 | [11] |
| LDCM0127 | JWB152 | DM93 | Y156(1.14); Y227(3.45) | LDD0289 | [11] |
| LDCM0128 | JWB198 | DM93 | Y156(0.13); Y227(0.12) | LDD0290 | [11] |
| LDCM0129 | JWB202 | DM93 | Y156(0.64); Y227(0.36) | LDD0291 | [11] |
| LDCM0130 | JWB211 | DM93 | Y156(0.52); Y227(0.56) | LDD0292 | [11] |
| LDCM0022 | KB02 | HEK-293T | C273(1.03); C261(0.97); C218(1.15); C222(1.00) | LDD1492 | [31] |
| LDCM0023 | KB03 | Jurkat | C261(4.31) | LDD0209 | [10] |
| LDCM0024 | KB05 | COLO792 | C219(0.69); C299(1.28) | LDD3310 | [32] |
| LDCM0528 | N-(4-bromophenyl)-2-cyano-N-phenylacetamide | MDA-MB-231 | C41(0.58); C273(0.88) | LDD2121 | [7] |
| LDCM0109 | NEM | HeLa | H292(0.00); C261(0.00); H290(0.00); H121(0.00) | LDD0223 | [23] |
| LDCM0496 | Nucleophilic fragment 11a | MDA-MB-231 | C41(0.52); C273(1.07) | LDD2089 | [7] |
| LDCM0499 | Nucleophilic fragment 12b | MDA-MB-231 | C41(0.76) | LDD2092 | [7] |
| LDCM0500 | Nucleophilic fragment 13a | MDA-MB-231 | C41(1.04); C273(1.19) | LDD2093 | [7] |
| LDCM0501 | Nucleophilic fragment 13b | MDA-MB-231 | C41(1.14) | LDD2094 | [7] |
| LDCM0503 | Nucleophilic fragment 14b | MDA-MB-231 | C41(0.60) | LDD2096 | [7] |
| LDCM0504 | Nucleophilic fragment 15a | MDA-MB-231 | C41(1.23); C273(0.74) | LDD2097 | [7] |
| LDCM0505 | Nucleophilic fragment 15b | MDA-MB-231 | C41(0.98) | LDD2098 | [7] |
| LDCM0506 | Nucleophilic fragment 16a | MDA-MB-231 | C41(1.00); C273(1.11); C261(1.12) | LDD2099 | [7] |
| LDCM0507 | Nucleophilic fragment 16b | MDA-MB-231 | C41(0.70) | LDD2100 | [7] |
| LDCM0508 | Nucleophilic fragment 17a | MDA-MB-231 | C41(1.07); C273(0.89) | LDD2101 | [7] |
| LDCM0511 | Nucleophilic fragment 18b | MDA-MB-231 | C41(0.74) | LDD2104 | [7] |
| LDCM0512 | Nucleophilic fragment 19a | MDA-MB-231 | C41(1.37) | LDD2105 | [7] |
| LDCM0513 | Nucleophilic fragment 19b | MDA-MB-231 | C41(0.62) | LDD2106 | [7] |
| LDCM0514 | Nucleophilic fragment 20a | MDA-MB-231 | C41(0.94); C261(1.33) | LDD2107 | [7] |
| LDCM0515 | Nucleophilic fragment 20b | MDA-MB-231 | C41(0.82); C222(1.06) | LDD2108 | [7] |
| LDCM0516 | Nucleophilic fragment 21a | MDA-MB-231 | C41(0.83); C273(0.87); C261(0.93) | LDD2109 | [7] |
| LDCM0517 | Nucleophilic fragment 21b | MDA-MB-231 | C41(1.18) | LDD2110 | [7] |
| LDCM0518 | Nucleophilic fragment 22a | MDA-MB-231 | C41(0.97); C273(1.27) | LDD2111 | [7] |
| LDCM0522 | Nucleophilic fragment 24a | MDA-MB-231 | C41(0.76) | LDD2115 | [7] |
| LDCM0523 | Nucleophilic fragment 24b | MDA-MB-231 | C41(0.93) | LDD2116 | [7] |
| LDCM0525 | Nucleophilic fragment 25b | MDA-MB-231 | C41(0.83) | LDD2118 | [7] |
| LDCM0526 | Nucleophilic fragment 26a | MDA-MB-231 | C41(1.62); C273(1.84) | LDD2119 | [7] |
| LDCM0527 | Nucleophilic fragment 26b | MDA-MB-231 | C41(0.63); C273(1.53) | LDD2120 | [7] |
| LDCM0529 | Nucleophilic fragment 27b | MDA-MB-231 | C41(0.86) | LDD2122 | [7] |
| LDCM0530 | Nucleophilic fragment 28a | MDA-MB-231 | C41(1.02); C273(1.19); C261(1.05) | LDD2123 | [7] |
| LDCM0531 | Nucleophilic fragment 28b | MDA-MB-231 | C41(0.82) | LDD2124 | [7] |
| LDCM0532 | Nucleophilic fragment 29a | MDA-MB-231 | C41(0.93); C273(0.95); C261(1.06) | LDD2125 | [7] |
| LDCM0533 | Nucleophilic fragment 29b | MDA-MB-231 | C41(1.01) | LDD2126 | [7] |
| LDCM0534 | Nucleophilic fragment 30a | MDA-MB-231 | C41(1.15); C273(1.07) | LDD2127 | [7] |
| LDCM0535 | Nucleophilic fragment 30b | MDA-MB-231 | C41(0.91) | LDD2128 | [7] |
| LDCM0536 | Nucleophilic fragment 31 | MDA-MB-231 | C41(1.06); C273(1.37) | LDD2129 | [7] |
| LDCM0540 | Nucleophilic fragment 35 | MDA-MB-231 | C41(0.65); C273(0.81) | LDD2133 | [7] |
| LDCM0541 | Nucleophilic fragment 36 | MDA-MB-231 | C41(0.62); C273(0.54) | LDD2134 | [7] |
| LDCM0542 | Nucleophilic fragment 37 | MDA-MB-231 | C41(1.06); C273(1.21) | LDD2135 | [7] |
| LDCM0543 | Nucleophilic fragment 38 | MDA-MB-231 | C41(1.13); C273(1.32); C261(1.40) | LDD2136 | [7] |
| LDCM0544 | Nucleophilic fragment 39 | MDA-MB-231 | C41(1.01); C261(1.02); C222(0.91) | LDD2137 | [7] |
| LDCM0211 | Nucleophilic fragment 3b | MDA-MB-231 | C273(1.80); C41(1.57) | LDD1700 | [7] |
| LDCM0546 | Nucleophilic fragment 40 | MDA-MB-231 | C41(0.93); C273(0.94); C261(0.95) | LDD2140 | [7] |
| LDCM0547 | Nucleophilic fragment 41 | MDA-MB-231 | C41(0.95); C261(0.71) | LDD2141 | [7] |
| LDCM0549 | Nucleophilic fragment 43 | MDA-MB-231 | C41(0.99) | LDD2143 | [7] |
| LDCM0550 | Nucleophilic fragment 5a | MDA-MB-231 | C41(2.16); C273(1.87) | LDD2144 | [7] |
| LDCM0551 | Nucleophilic fragment 5b | MDA-MB-231 | C41(1.05); C261(1.01) | LDD2145 | [7] |
| LDCM0552 | Nucleophilic fragment 6a | MDA-MB-231 | C41(0.85); C273(1.08) | LDD2146 | [7] |
| LDCM0554 | Nucleophilic fragment 7a | MDA-MB-231 | C41(0.98); C273(0.56) | LDD2148 | [7] |
| LDCM0555 | Nucleophilic fragment 7b | MDA-MB-231 | C41(1.29) | LDD2149 | [7] |
| LDCM0556 | Nucleophilic fragment 8a | MDA-MB-231 | C41(0.31); C273(0.63) | LDD2150 | [7] |
| LDCM0557 | Nucleophilic fragment 8b | MDA-MB-231 | C41(0.84) | LDD2151 | [7] |
| LDCM0628 | OTUB2-COV-1 | HEK-293T | C181(0.70) | LDD2207 | [33] |
| LDCM0131 | RA190 | MM1.R | C273(1.52); C261(1.24) | LDD0304 | [34] |
The Interaction Atlas With This Target
The Protein(s) Related To This Target
Enzyme
Transporter and channel
| Protein name | Family | Uniprot ID | |||
|---|---|---|---|---|---|
| MICOS complex subunit MIC60 (IMMT) | MICOS complex subunit Mic60 family | Q16891 | |||
References
