Details of the Target
General Information of Target
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
DBIA Probe Info |
 |
C92(1.65) | LDD3310 | [1] | |
|
JZ128-DTB Probe Info |
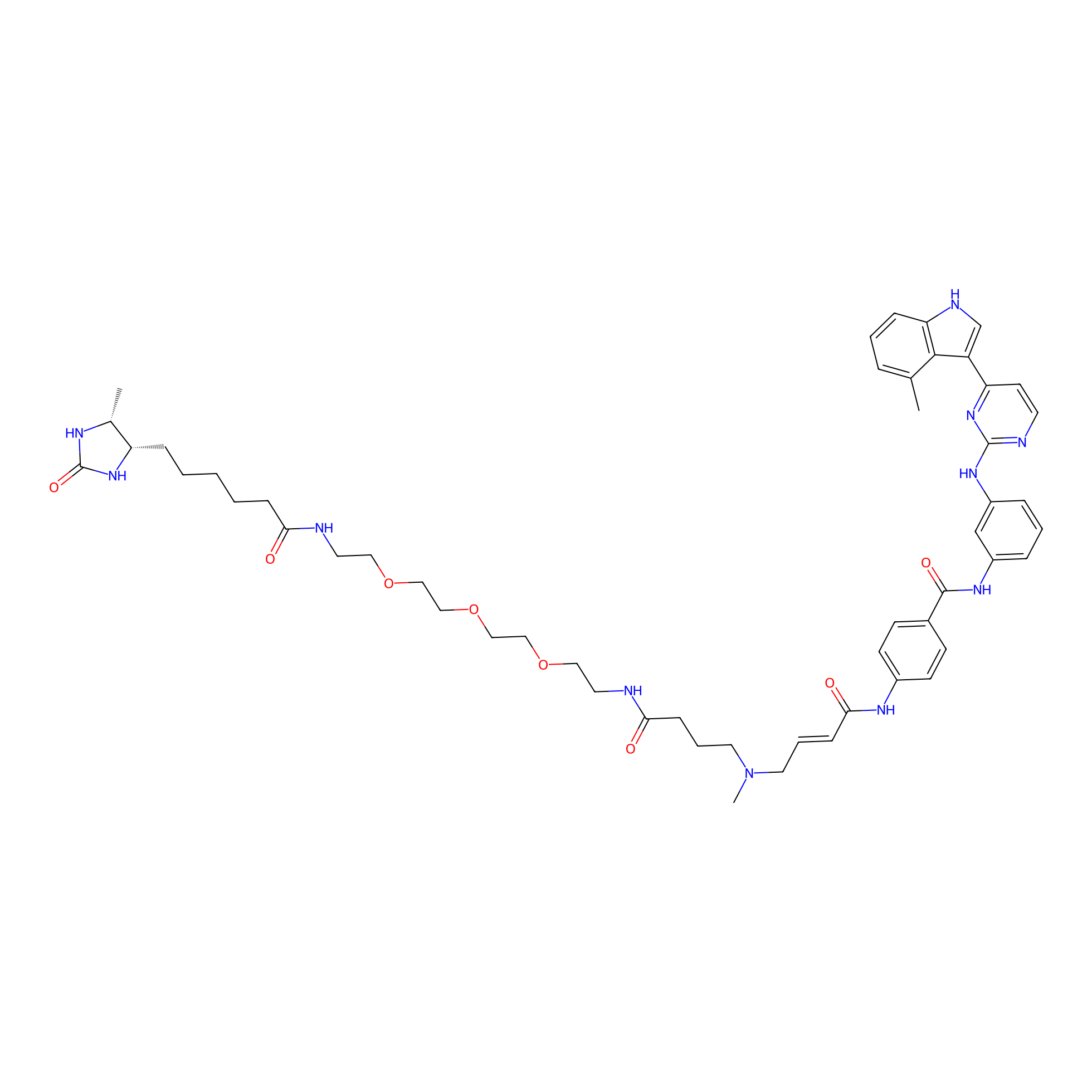 |
N.A. | LDD0462 | [2] | |
|
THZ1-DTB Probe Info |
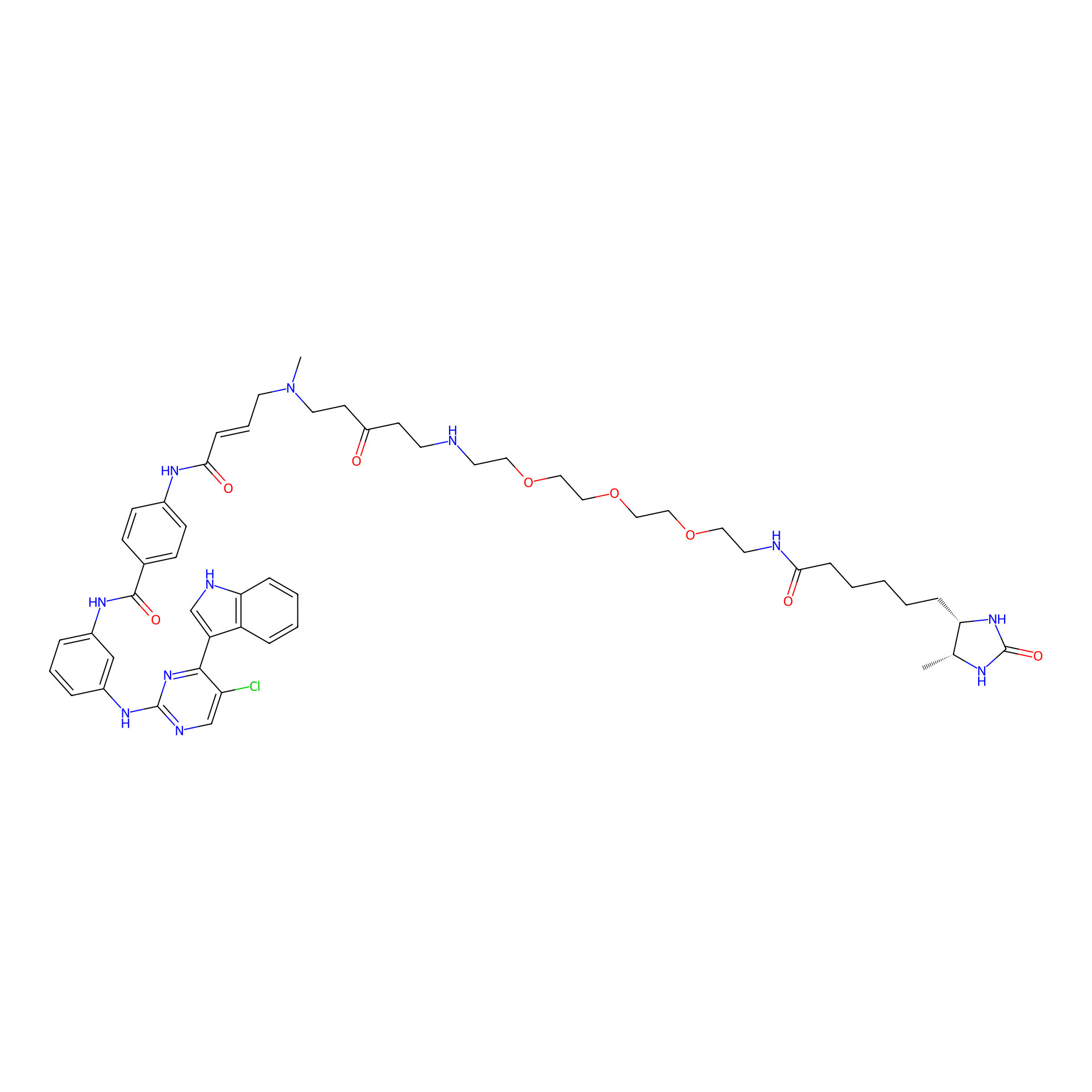 |
C92(1.73) | LDD0460 | [2] | |
|
Jackson_14 Probe Info |
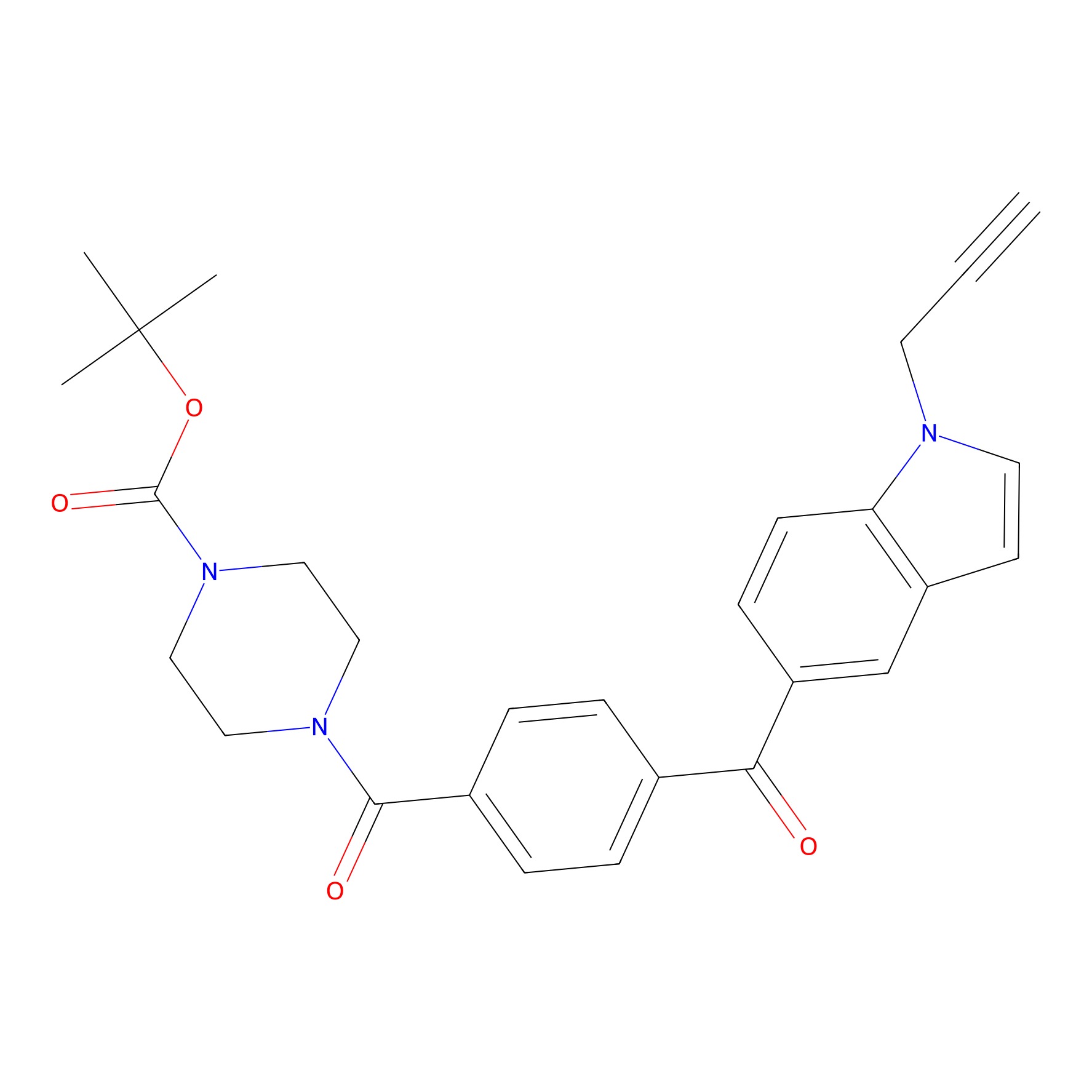 |
3.57 | LDD0123 | [3] | |
|
NAIA_5 Probe Info |
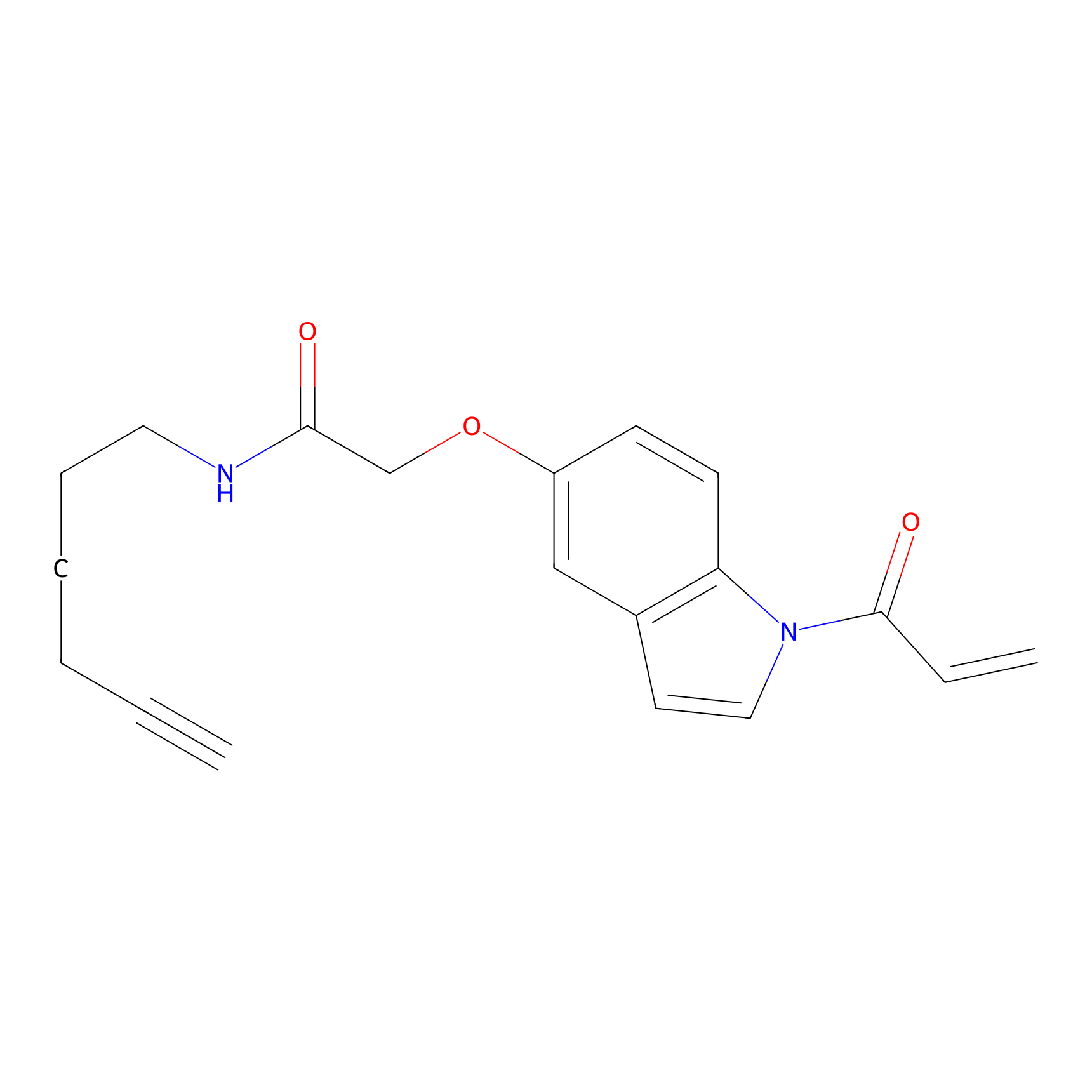 |
C280(20.00) | LDD2227 | [4] | |
|
Acrolein Probe Info |
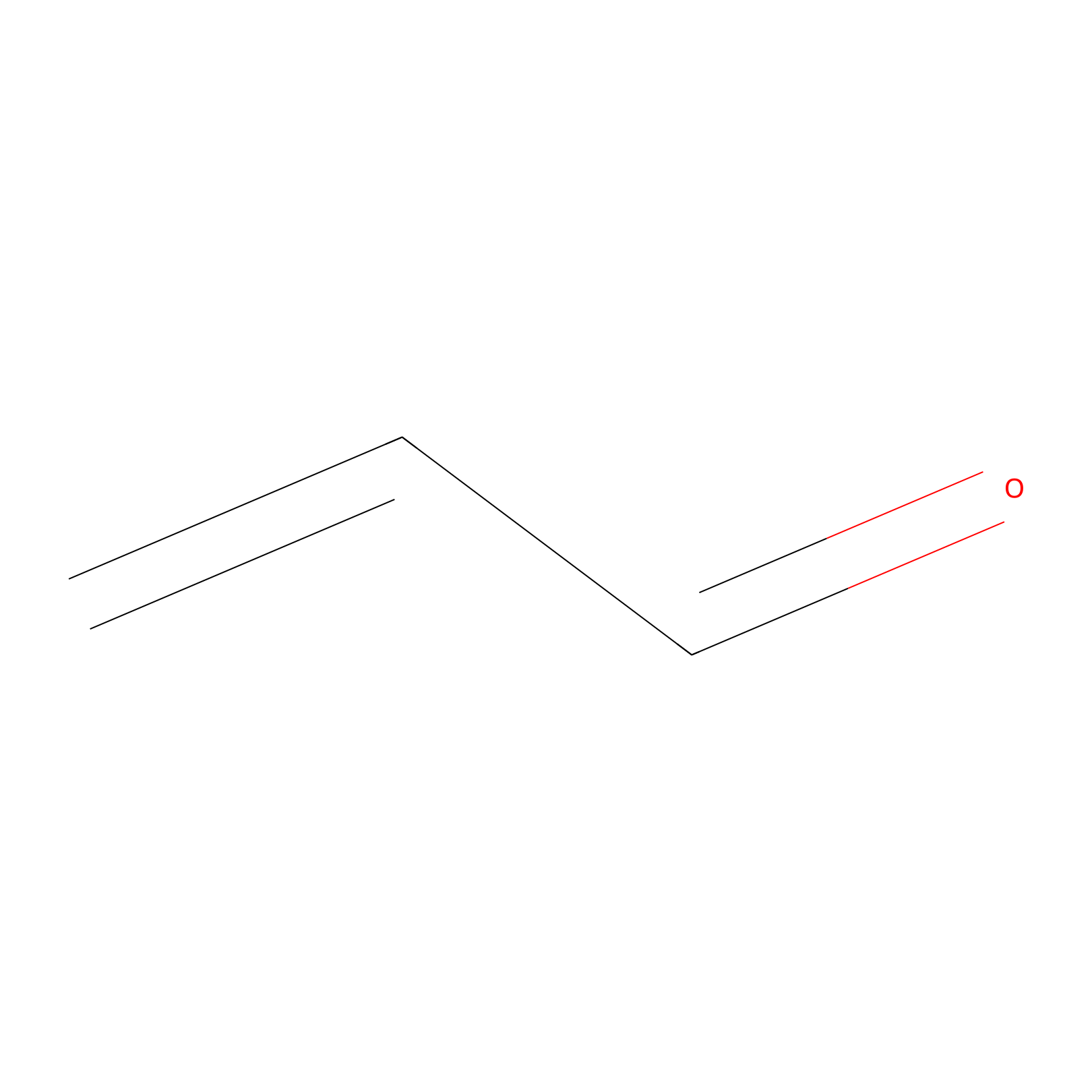 |
H279(0.00); C280(0.00) | LDD0222 | [5] | |
|
JW-RF-010 Probe Info |
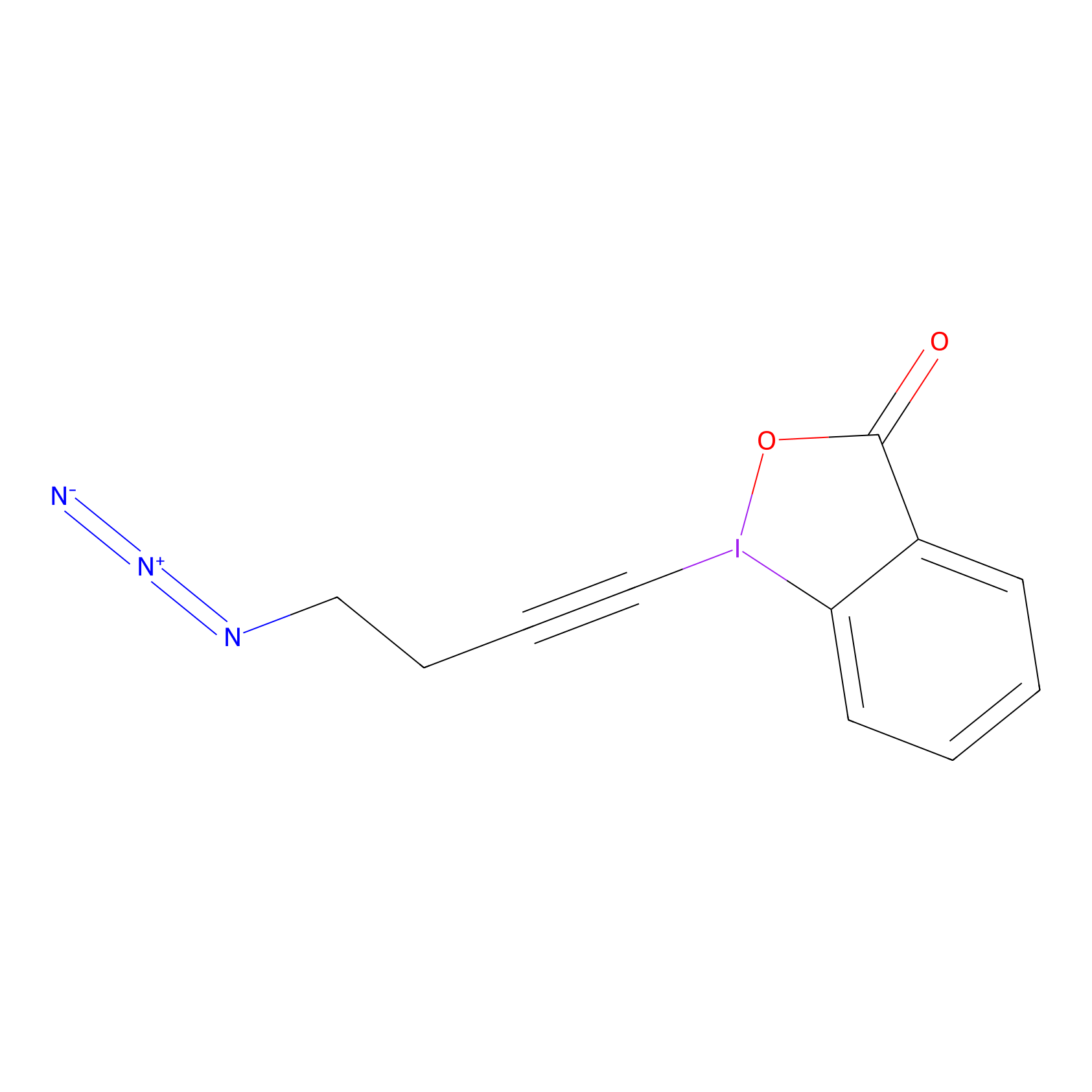 |
N.A. | LDD0026 | [6] | |
|
TFBX Probe Info |
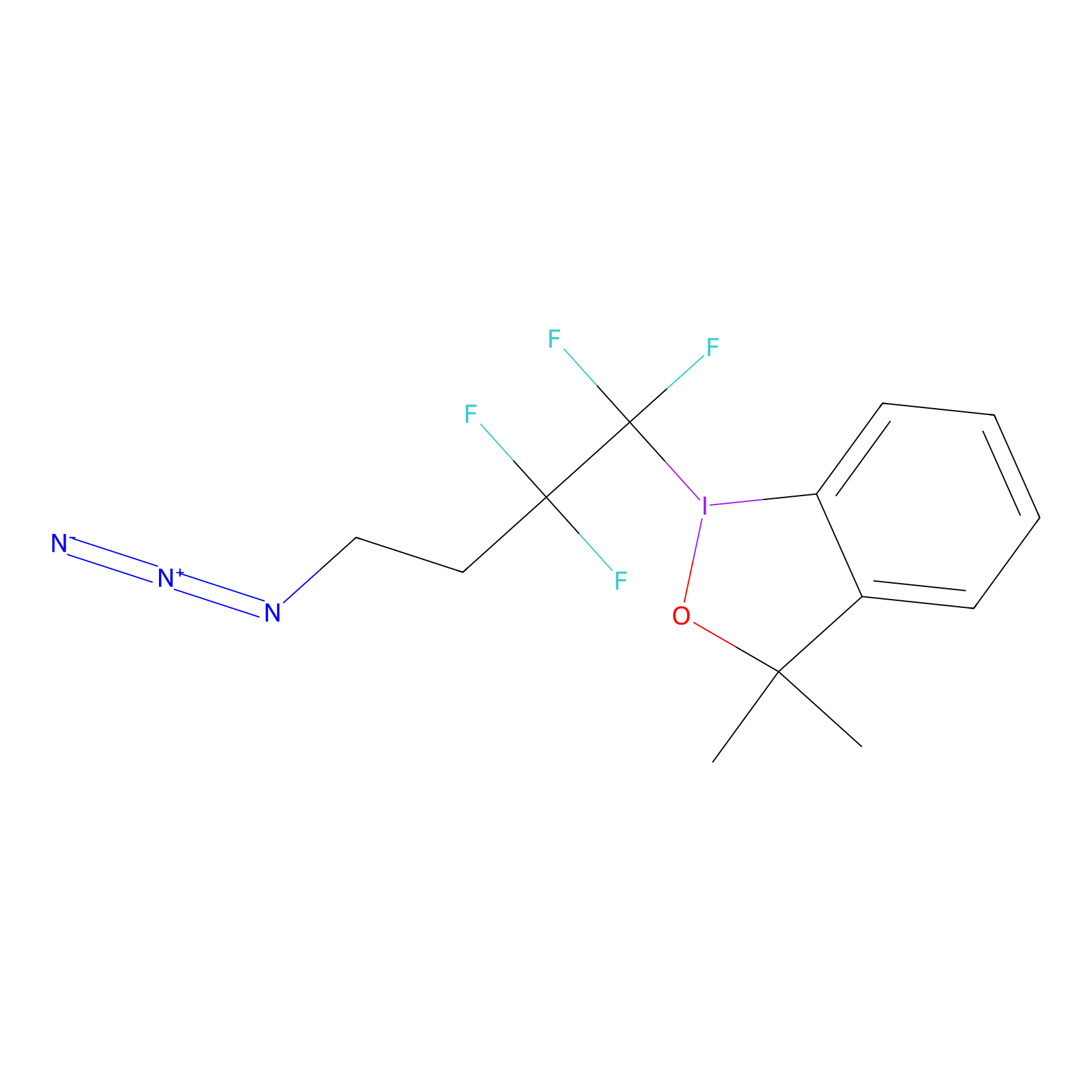 |
N.A. | LDD0027 | [6] | |
|
IPM Probe Info |
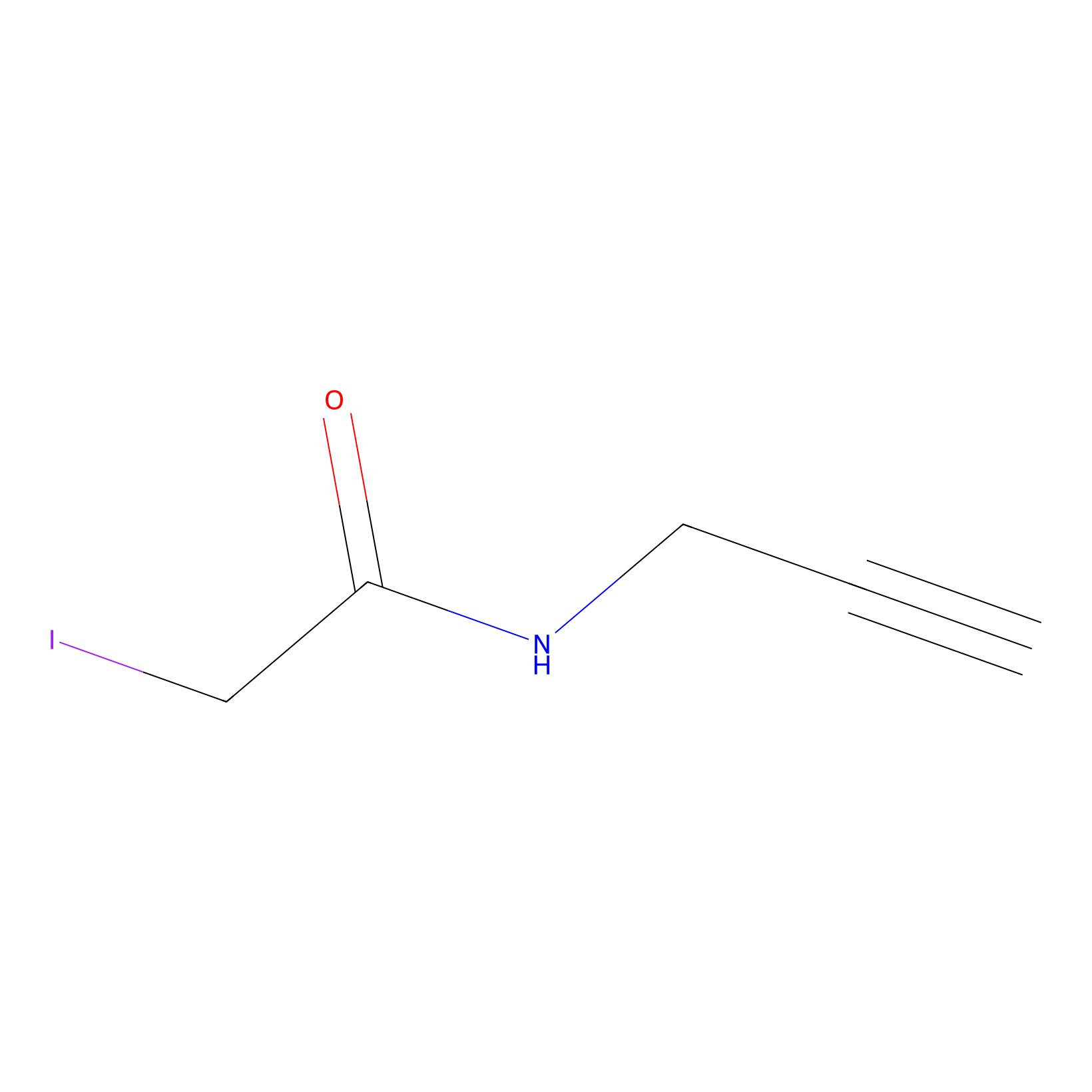 |
C280(0.00); C92(0.00) | LDD0147 | [6] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0108 | Chloroacetamide | HeLa | H279(0.00); C280(0.00) | LDD0222 | [5] |
| LDCM0632 | CL-Sc | Hep-G2 | C280(20.00) | LDD2227 | [4] |
| LDCM0179 | JZ128 | PC-3 | N.A. | LDD0462 | [2] |
| LDCM0022 | KB02 | A2780 | C280(2.16); C92(2.35) | LDD2254 | [1] |
| LDCM0023 | KB03 | 786-O | C280(1.69) | LDD2664 | [1] |
| LDCM0024 | KB05 | COLO792 | C92(1.65) | LDD3310 | [1] |
| LDCM0016 | Ranjitkar_cp1 | MDA-MB-231 | 3.57 | LDD0123 | [3] |
| LDCM0021 | THZ1 | HeLa S3 | C92(1.73) | LDD0460 | [2] |
The Interaction Atlas With This Target
The Protein(s) Related To This Target
Enzyme
| Protein name | Family | Uniprot ID | |||
|---|---|---|---|---|---|
| Angiopoietin-1 receptor (TEK) | Tyr protein kinase family | Q02763 | |||
The Drug(s) Related To This Target
Approved
Phase 2
| Drug Name | Drug Type | External ID | |||
|---|---|---|---|---|---|
| Pinocembrin | Small molecular drug | D0G5CS | |||
Phase 1
| Drug Name | Drug Type | External ID | |||
|---|---|---|---|---|---|
| Naringenin | Small molecular drug | D02ABO | |||
Investigative
References
