Details of the Target
General Information of Target
| Target ID | LDTP05474 | |||||
|---|---|---|---|---|---|---|
| Target Name | ATP synthase F(0) complex subunit C2, mitochondrial (ATP5MC2) | |||||
| Gene Name | ATP5MC2 | |||||
| Gene ID | 517 | |||||
| Synonyms |
ATP5G2; ATP synthase F(0) complex subunit C2, mitochondrial; ATP synthase lipid-binding protein; ATP synthase membrane subunit c locus 2; ATP synthase proteolipid P2; ATP synthase proton-transporting mitochondrial F(0) complex subunit C2; ATPase protein 9; ATPase subunit c
|
|||||
| 3D Structure | ||||||
| Sequence |
MFACSKFVSTPSLVKSTSQLLSRPLSAVVLKRPEILTDESLSSLAVSCPLTSLVSSRSFQ
TSAISRDIDTAAKFIGAGAATVGVAGSGAGIGTVFGSLIIGYARNPSLKQQLFSYAILGF ALSEAMGLFCLMVAFLILFAM |
|||||
| Target Bioclass |
Transporter and channel
|
|||||
| Family |
ATPase C chain family
|
|||||
| Subcellular location |
Mitochondrion membrane
|
|||||
| Function |
Mitochondrial membrane ATP synthase (F(1)F(0) ATP synthase or Complex V) produces ATP from ADP in the presence of a proton gradient across the membrane which is generated by electron transport complexes of the respiratory chain. F-type ATPases consist of two structural domains, F(1) - containing the extramembraneous catalytic core and F(0) - containing the membrane proton channel, linked together by a central stalk and a peripheral stalk. During catalysis, ATP synthesis in the catalytic domain of F(1) is coupled via a rotary mechanism of the central stalk subunits to proton translocation. Part of the complex F(0) domain. A homomeric c-ring of probably 10 subunits is part of the complex rotary element.
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
DBIA Probe Info |
 |
C48(1.05) | LDD1513 | [1] | |
PAL-AfBPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
C112 Probe Info |
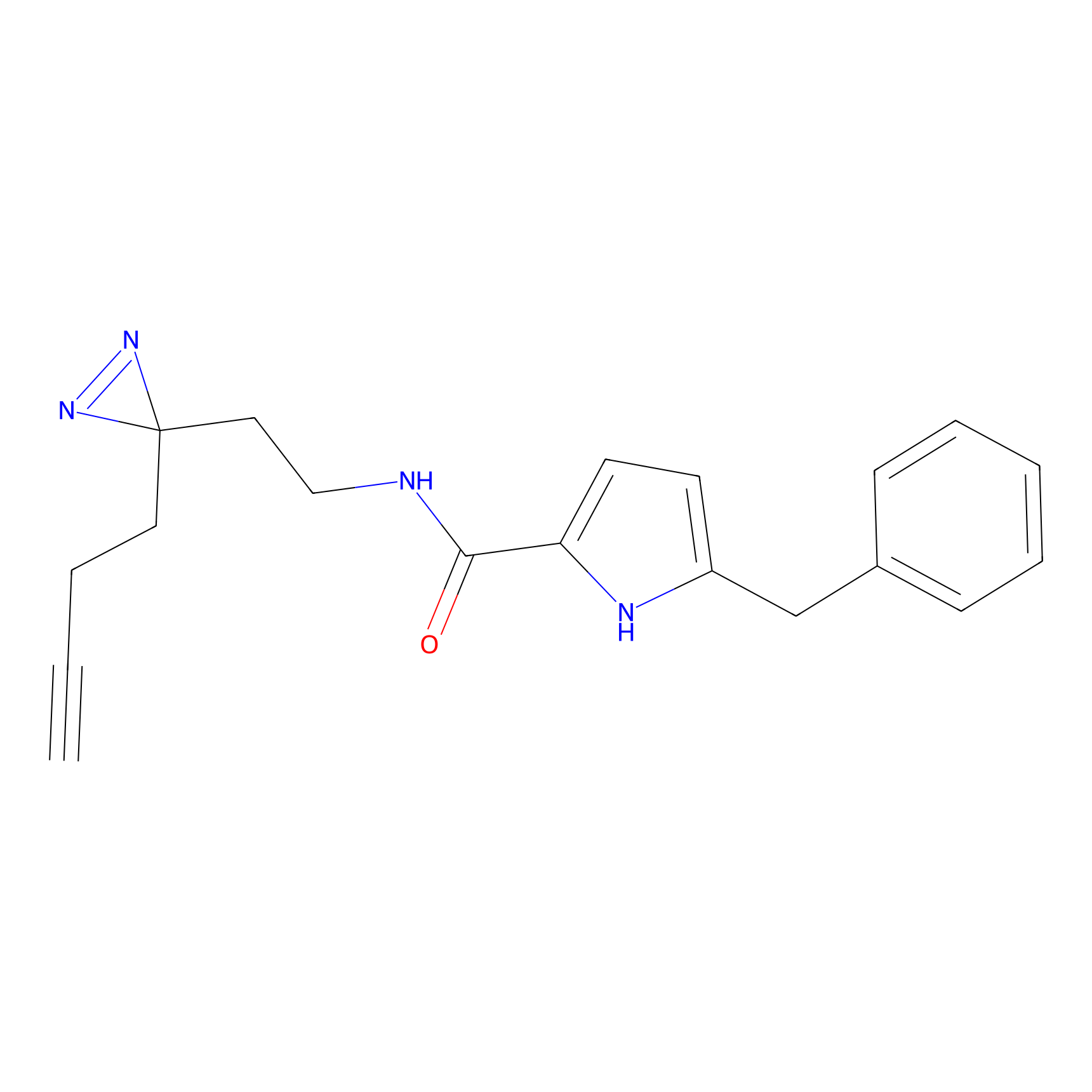 |
28.25 | LDD1799 | [2] | |
|
C278 Probe Info |
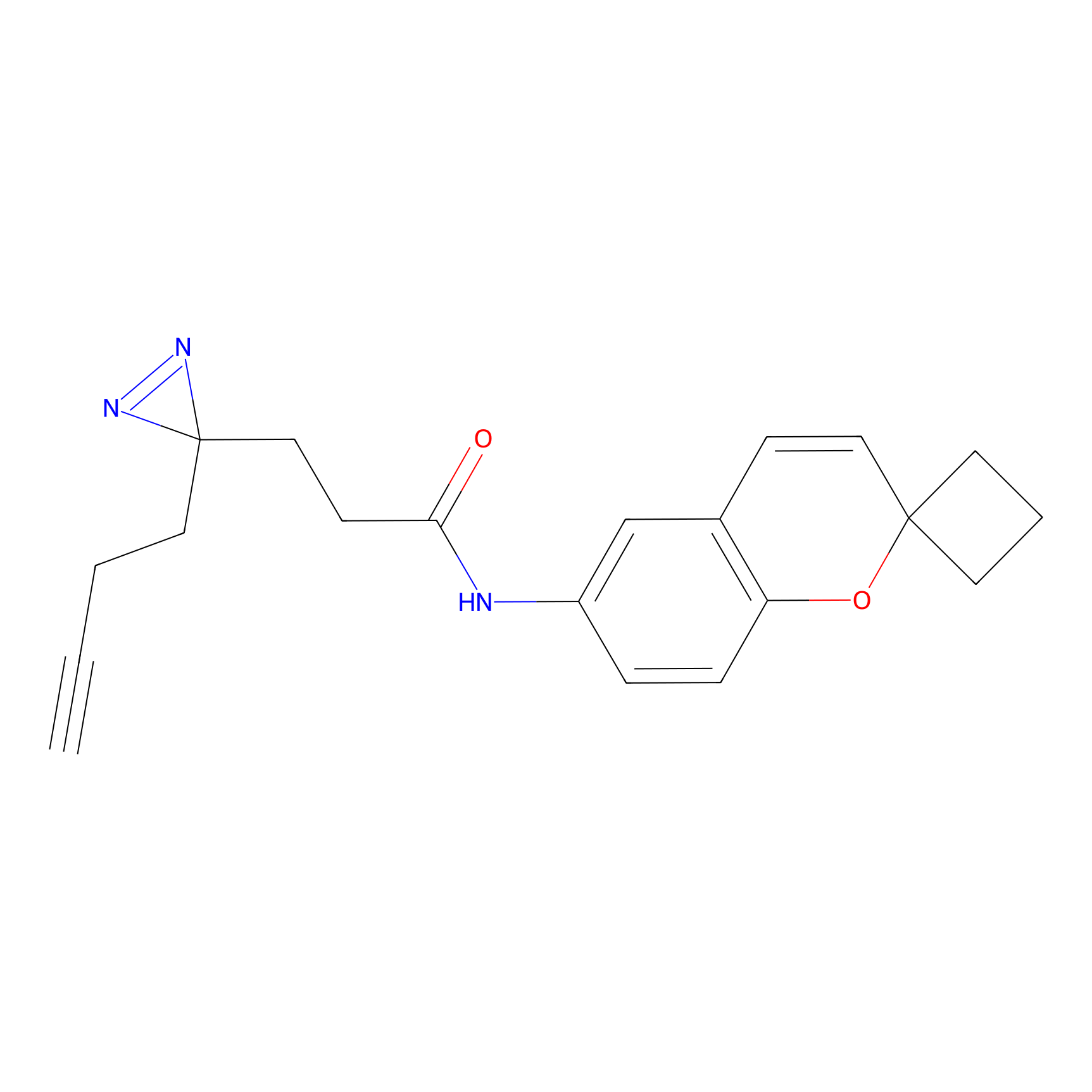 |
56.49 | LDD1948 | [2] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0270 | AC15 | HEK-293T | C48(1.05) | LDD1513 | [1] |
| LDCM0283 | AC23 | HEK-293T | C48(1.06) | LDD1522 | [1] |
| LDCM0284 | AC24 | HEK-293T | C48(1.07) | LDD1523 | [1] |
| LDCM0292 | AC31 | HEK-293T | C48(1.28) | LDD1531 | [1] |
| LDCM0293 | AC32 | HEK-293T | C48(0.91) | LDD1532 | [1] |
| LDCM0300 | AC39 | HEK-293T | C48(1.04) | LDD1539 | [1] |
| LDCM0302 | AC40 | HEK-293T | C48(0.97) | LDD1541 | [1] |
| LDCM0309 | AC47 | HEK-293T | C48(0.91) | LDD1548 | [1] |
| LDCM0310 | AC48 | HEK-293T | C48(1.05) | LDD1549 | [1] |
| LDCM0318 | AC55 | HEK-293T | C48(0.99) | LDD1557 | [1] |
| LDCM0319 | AC56 | HEK-293T | C48(1.07) | LDD1558 | [1] |
| LDCM0327 | AC63 | HEK-293T | C48(1.21) | LDD1566 | [1] |
| LDCM0328 | AC64 | HEK-293T | C48(1.02) | LDD1567 | [1] |
| LDCM0334 | AC7 | HEK-293T | C48(1.12) | LDD1568 | [1] |
| LDCM0345 | AC8 | HEK-293T | C48(1.09) | LDD1569 | [1] |
| LDCM0275 | AKOS034007705 | HEK-293T | C48(0.97) | LDD1514 | [1] |
| LDCM0379 | CL11 | HEK-293T | C48(1.23) | LDD1583 | [1] |
| LDCM0390 | CL12 | HEK-293T | C48(1.15) | LDD1594 | [1] |
| LDCM0411 | CL23 | HEK-293T | C48(1.32) | LDD1615 | [1] |
| LDCM0412 | CL24 | HEK-293T | C48(1.03) | LDD1616 | [1] |
| LDCM0424 | CL35 | HEK-293T | C48(1.15) | LDD1628 | [1] |
| LDCM0425 | CL36 | HEK-293T | C48(1.07) | LDD1629 | [1] |
| LDCM0437 | CL47 | HEK-293T | C48(1.23) | LDD1641 | [1] |
| LDCM0438 | CL48 | HEK-293T | C48(1.20) | LDD1642 | [1] |
| LDCM0450 | CL59 | HEK-293T | C48(1.35) | LDD1653 | [1] |
| LDCM0452 | CL60 | HEK-293T | C48(1.25) | LDD1655 | [1] |
| LDCM0464 | CL71 | HEK-293T | C48(1.46) | LDD1667 | [1] |
| LDCM0465 | CL72 | HEK-293T | C48(1.16) | LDD1668 | [1] |
| LDCM0477 | CL83 | HEK-293T | C48(1.15) | LDD1680 | [1] |
| LDCM0478 | CL84 | HEK-293T | C48(1.21) | LDD1681 | [1] |
| LDCM0490 | CL95 | HEK-293T | C48(1.11) | LDD1693 | [1] |
| LDCM0491 | CL96 | HEK-293T | C48(1.26) | LDD1694 | [1] |
References
