Details of the Target
General Information of Target
| Target ID | LDTP11903 | |||||
|---|---|---|---|---|---|---|
| Target Name | Ribokinase (RBKS) | |||||
| Gene Name | RBKS | |||||
| Gene ID | 64080 | |||||
| Synonyms |
RBSK; Ribokinase; RK; EC 2.7.1.15 |
|||||
| 3D Structure | ||||||
| Sequence |
MEPPMEPSGGEQEPGAVRFLDLPWEDVLLPHVLNRVPLRQLLRLQRVSRAFRSLVQLHLA
GLRRFDAAQVGPQIPRAALARLLRDAEGLQELALAPCHEWLSDEDLVPVLARNPQLRSVA LGGCGQLSRRALGALAEGCPRLQRLSLAHCDWVDGLALRGLADRCPALEELDLTACRQLK DEAIVYLAQRRGAGLRSLSLAVNANVGDAAVQELARNCPELHHLDLTGCLRVGSDGVRTL AEYCPVLRSLRVRHCHHVAESSLSRLRKRGVDIDVEPPLHQALVLLQDMAGFAPFVNLQV |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Carbohydrate kinase PfkB family, Ribokinase subfamily
|
|||||
| Subcellular location |
Cytoplasm
|
|||||
| Function |
Catalyzes the phosphorylation of ribose at O-5 in a reaction requiring ATP and magnesium. The resulting D-ribose-5-phosphate can then be used either for sythesis of nucleotides, histidine, and tryptophan, or as a component of the pentose phosphate pathway. {|HAMAP-Rule:MF_03215}.
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
DBIA Probe Info |
 |
C59(1.75) | LDD3378 | [1] | |
|
BTD Probe Info |
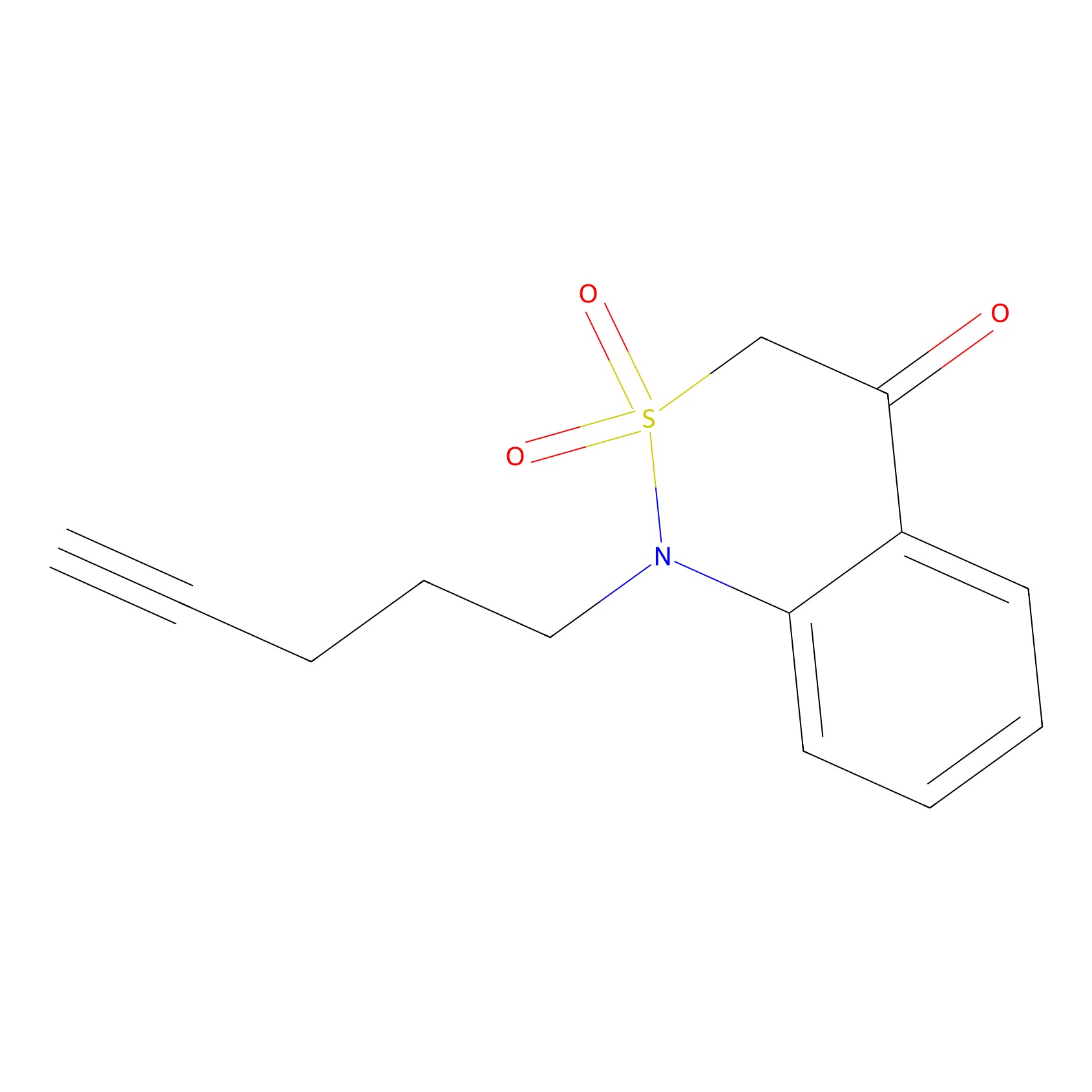 |
C59(1.35) | LDD2090 | [2] | |
|
ATP probe Probe Info |
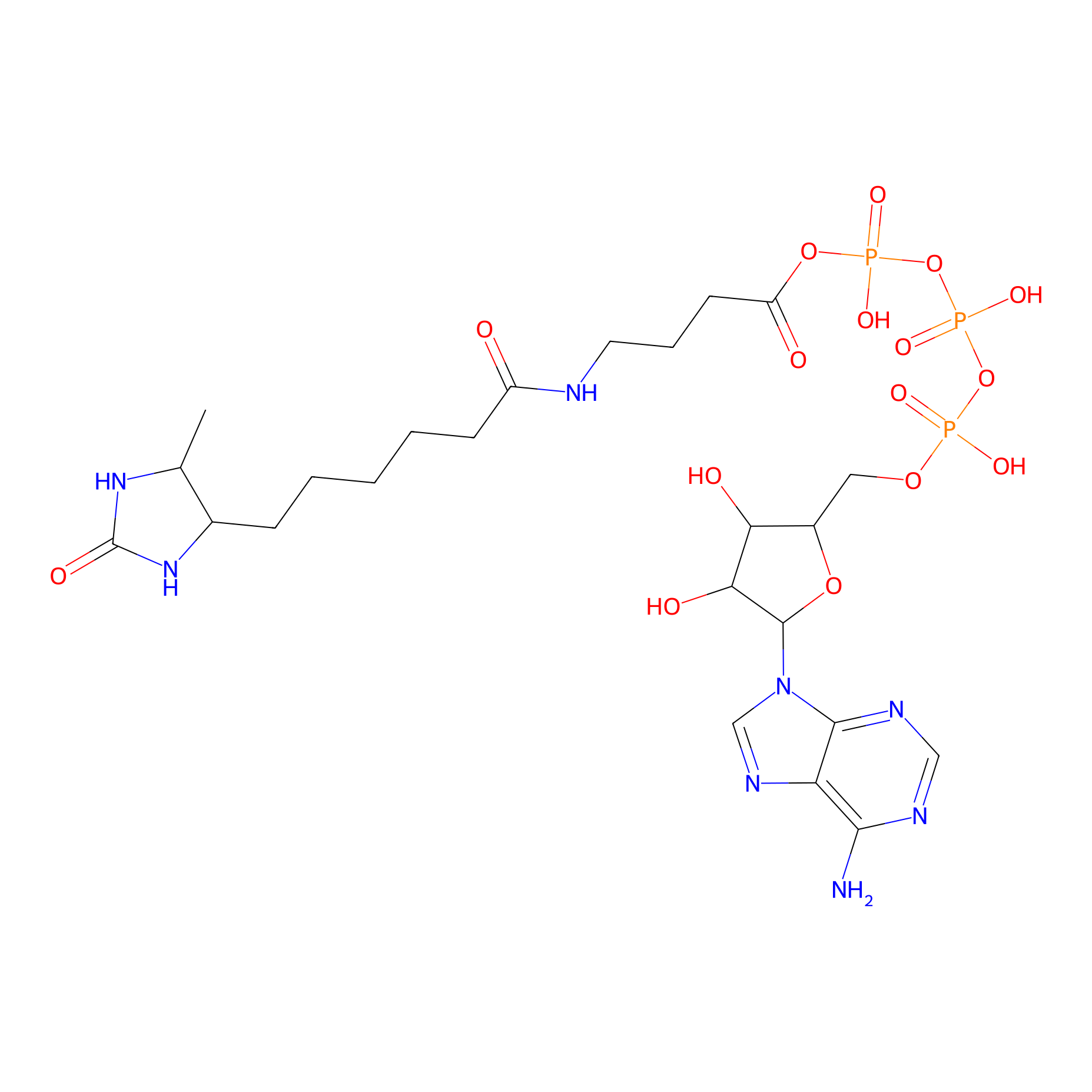 |
N.A. | LDD0199 | [3] | |
|
NAIA_5 Probe Info |
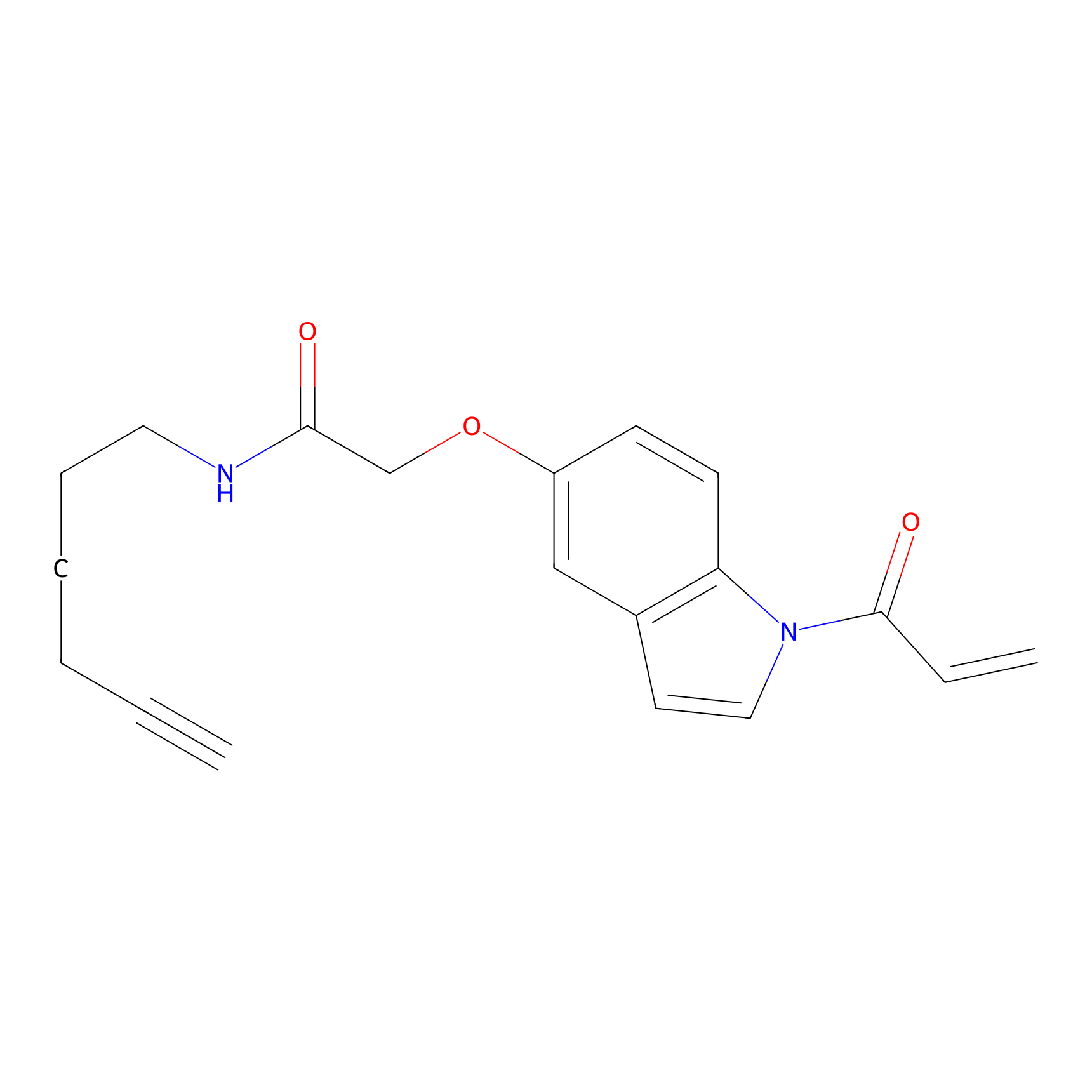 |
C59(0.00); C24(0.00) | LDD2223 | [4] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0214 | AC1 | HEK-293T | C59(1.25) | LDD1507 | [5] |
| LDCM0276 | AC17 | HEK-293T | C59(1.04) | LDD1515 | [5] |
| LDCM0281 | AC21 | HEK-293T | C59(1.22) | LDD1520 | [5] |
| LDCM0285 | AC25 | HEK-293T | C59(0.76) | LDD1524 | [5] |
| LDCM0289 | AC29 | HEK-293T | C59(1.20) | LDD1528 | [5] |
| LDCM0294 | AC33 | HEK-293T | C59(1.29) | LDD1533 | [5] |
| LDCM0298 | AC37 | HEK-293T | C59(0.96) | LDD1537 | [5] |
| LDCM0303 | AC41 | HEK-293T | C59(1.61) | LDD1542 | [5] |
| LDCM0307 | AC45 | HEK-293T | C59(1.31) | LDD1546 | [5] |
| LDCM0311 | AC49 | HEK-293T | C59(0.95) | LDD1550 | [5] |
| LDCM0312 | AC5 | HEK-293T | C59(0.71) | LDD1551 | [5] |
| LDCM0316 | AC53 | HEK-293T | C59(0.97) | LDD1555 | [5] |
| LDCM0320 | AC57 | HEK-293T | C59(1.27) | LDD1559 | [5] |
| LDCM0325 | AC61 | HEK-293T | C59(1.12) | LDD1564 | [5] |
| LDCM0248 | AKOS034007472 | HEK-293T | C59(1.02) | LDD1511 | [5] |
| LDCM0356 | AKOS034007680 | HEK-293T | C59(1.46) | LDD1570 | [5] |
| LDCM0404 | CL17 | HEK-293T | C59(1.16) | LDD1608 | [5] |
| LDCM0409 | CL21 | HEK-293T | C59(1.06) | LDD1613 | [5] |
| LDCM0417 | CL29 | HEK-293T | C59(1.07) | LDD1621 | [5] |
| LDCM0422 | CL33 | HEK-293T | C59(0.76) | LDD1626 | [5] |
| LDCM0431 | CL41 | HEK-293T | C59(0.96) | LDD1635 | [5] |
| LDCM0435 | CL45 | HEK-293T | C59(1.01) | LDD1639 | [5] |
| LDCM0440 | CL5 | HEK-293T | C59(1.07) | LDD1644 | [5] |
| LDCM0444 | CL53 | HEK-293T | C59(0.80) | LDD1647 | [5] |
| LDCM0448 | CL57 | HEK-293T | C59(1.14) | LDD1651 | [5] |
| LDCM0457 | CL65 | HEK-293T | C59(0.92) | LDD1660 | [5] |
| LDCM0461 | CL69 | HEK-293T | C59(0.95) | LDD1664 | [5] |
| LDCM0470 | CL77 | HEK-293T | C59(0.96) | LDD1673 | [5] |
| LDCM0475 | CL81 | HEK-293T | C59(1.22) | LDD1678 | [5] |
| LDCM0483 | CL89 | HEK-293T | C59(0.72) | LDD1686 | [5] |
| LDCM0484 | CL9 | HEK-293T | C59(0.97) | LDD1687 | [5] |
| LDCM0488 | CL93 | HEK-293T | C59(1.04) | LDD1691 | [5] |
| LDCM0022 | KB02 | 769-P | C59(1.77) | LDD2246 | [1] |
| LDCM0023 | KB03 | 769-P | C59(2.24) | LDD2663 | [1] |
| LDCM0024 | KB05 | OS-RC-2 | C59(1.75) | LDD3378 | [1] |
| LDCM0497 | Nucleophilic fragment 11b | MDA-MB-231 | C59(1.35) | LDD2090 | [2] |
| LDCM0503 | Nucleophilic fragment 14b | MDA-MB-231 | C59(0.31) | LDD2096 | [2] |
| LDCM0507 | Nucleophilic fragment 16b | MDA-MB-231 | C59(0.42) | LDD2100 | [2] |
| LDCM0512 | Nucleophilic fragment 19a | MDA-MB-231 | C59(1.28) | LDD2105 | [2] |
| LDCM0513 | Nucleophilic fragment 19b | MDA-MB-231 | C59(0.50) | LDD2106 | [2] |
| LDCM0522 | Nucleophilic fragment 24a | MDA-MB-231 | C59(0.54) | LDD2115 | [2] |
| LDCM0525 | Nucleophilic fragment 25b | MDA-MB-231 | C59(0.81) | LDD2118 | [2] |
| LDCM0527 | Nucleophilic fragment 26b | MDA-MB-231 | C59(0.93) | LDD2120 | [2] |
| LDCM0529 | Nucleophilic fragment 27b | MDA-MB-231 | C59(0.61) | LDD2122 | [2] |
| LDCM0531 | Nucleophilic fragment 28b | MDA-MB-231 | C59(0.58) | LDD2124 | [2] |
| LDCM0533 | Nucleophilic fragment 29b | MDA-MB-231 | C59(0.56) | LDD2126 | [2] |
| LDCM0551 | Nucleophilic fragment 5b | MDA-MB-231 | C59(0.69) | LDD2145 | [2] |
| LDCM0554 | Nucleophilic fragment 7a | MDA-MB-231 | C59(0.47) | LDD2148 | [2] |
| LDCM0555 | Nucleophilic fragment 7b | MDA-MB-231 | C59(0.75) | LDD2149 | [2] |
| LDCM0557 | Nucleophilic fragment 8b | MDA-MB-231 | C59(0.66) | LDD2151 | [2] |
| LDCM0559 | Nucleophilic fragment 9b | MDA-MB-231 | C59(1.27) | LDD2153 | [2] |
The Interaction Atlas With This Target
References
