Details of the Target
General Information of Target
| Target ID | LDTP01557 | |||||
|---|---|---|---|---|---|---|
| Target Name | RasGAP-activating-like protein 1 (RASAL1) | |||||
| Gene Name | RASAL1 | |||||
| Gene ID | 8437 | |||||
| Synonyms |
RASAL; RasGAP-activating-like protein 1; RAS protein activator like 1; Ras GTPase-activating-like protein |
|||||
| 3D Structure | ||||||
| Sequence |
MAKSSSLNVRVVEGRALPAKDVSGSSDPYCLVKVDDEVVARTATVWRSLGPFWGEEYTVH
LPLDFHQLAFYVLDEDTVGHDDIIGKISLSREAITADPRGIDSWINLSRVDPDAEVQGEI CLSVQMLEDGQGRCLRCHVLQARDLAPRDISGTSDPFARVFWGSQSLETSTIKKTRFPHW DEVLELREMPGAPSPLRVELWDWDMVGKNDFLGMVEFSPKTLQQKPPKGWFRLLPFPRAE EDSGGNLGALRVKVRLIEDRVLPSQCYQPLMELLMESVQGPAEEDTASPLALLEELTLGD CRQDLATKLVKLFLGRGLAGRFLDYLTRREVARTMDPNTLFRSNSLASKSMEQFMKLVGM PYLHEVLKPVISRVFEEKKYMELDPCKMDLGRTRRISFKGALSEEQMRETSLGLLTGYLG PIVDAIVGSVGRCPPAMRLAFKQLHRRVEERFPQAEHQDVKYLAISGFLFLRFFAPAILT PKLFDLRDQHADPQTSRSLLLLAKAVQSIGNLGQQLGQGKELWMAPLHPFLLQCVSRVRD FLDRLVDVDGDEAGVPARALFPPSAIVREGYLLKRKEEPAGLATRFAFKKRYVWLSGETL SFSKSPEWQMCHSIPVSHIRAVERVDEGAFQLPHVMQVVTQDGTGALHTTYLQCKNVNEL NQWLSALRKASAPNPNKLAACHPGAFRSARWTCCLQAERSAAGCSRTHSAVTLGDWSDPL DPDAEAQTVYRQLLLGRDQLRLKLLEDSNMDTTLEADTGACPEVLARQRAATARLLEVLA DLDRAHEEFQQQERGKAALGPLGP |
|||||
| Target Bioclass |
Other
|
|||||
| Function | Probable inhibitory regulator of the Ras-cyclic AMP pathway. Plays a role in dendrite formation by melanocytes. | |||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
AZ-9 Probe Info |
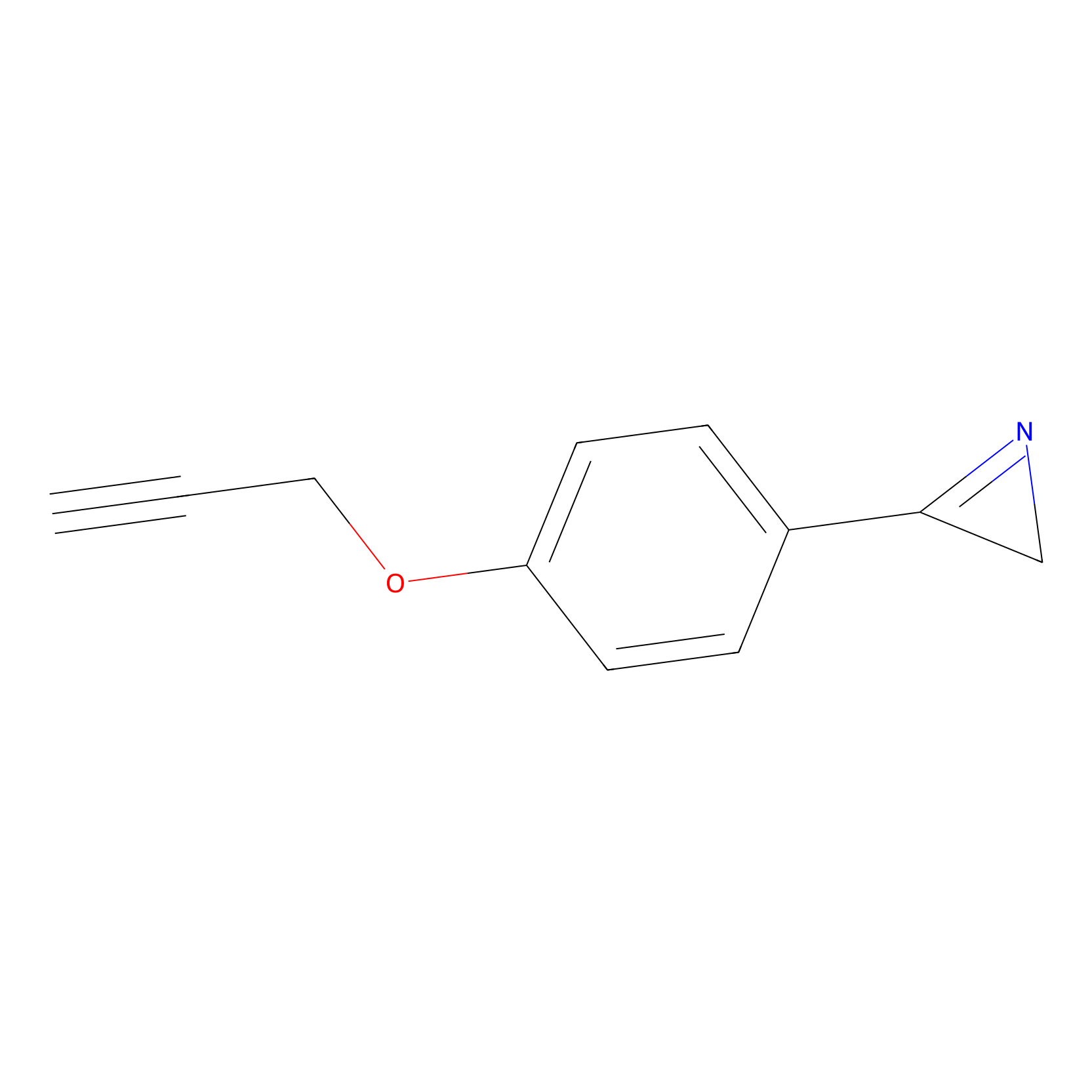 |
10.00 | LDD2154 | [1] | |
|
DBIA Probe Info |
 |
C386(9.20) | LDD0209 | [2] | |
|
4-Iodoacetamidophenylacetylene Probe Info |
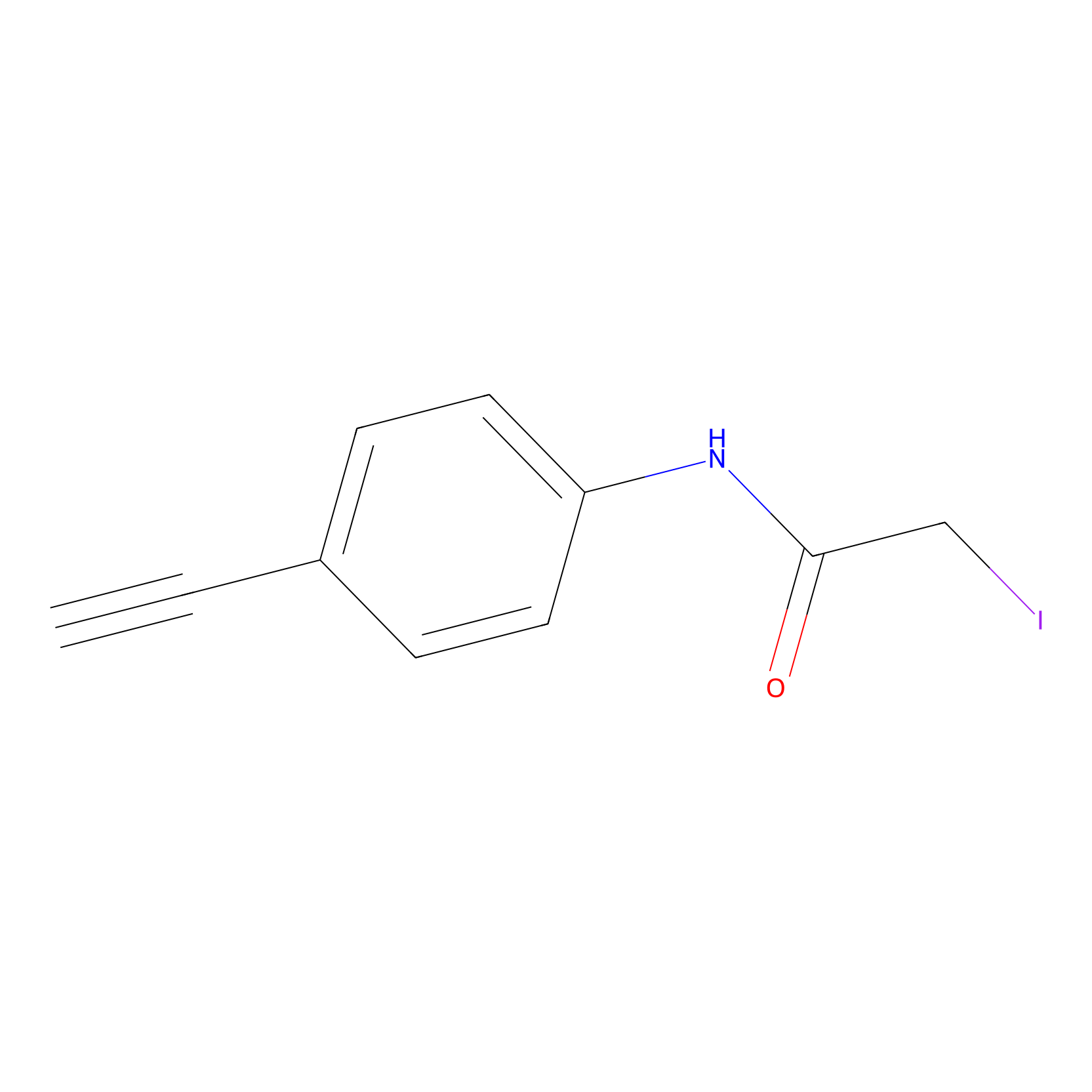 |
N.A. | LDD0038 | [3] | |
|
IA-alkyne Probe Info |
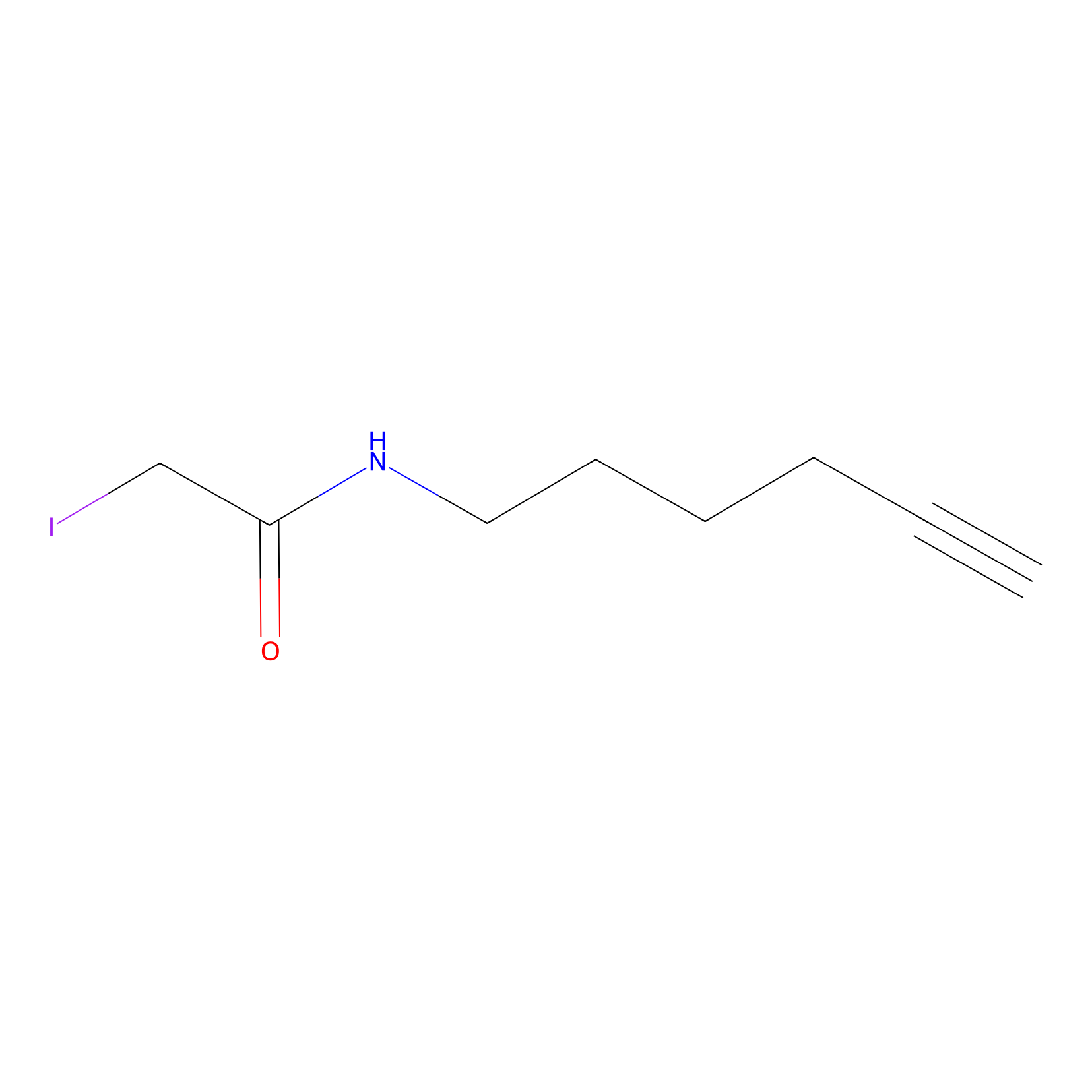 |
C681(0.00); C137(0.00) | LDD0036 | [3] | |
|
NAIA_5 Probe Info |
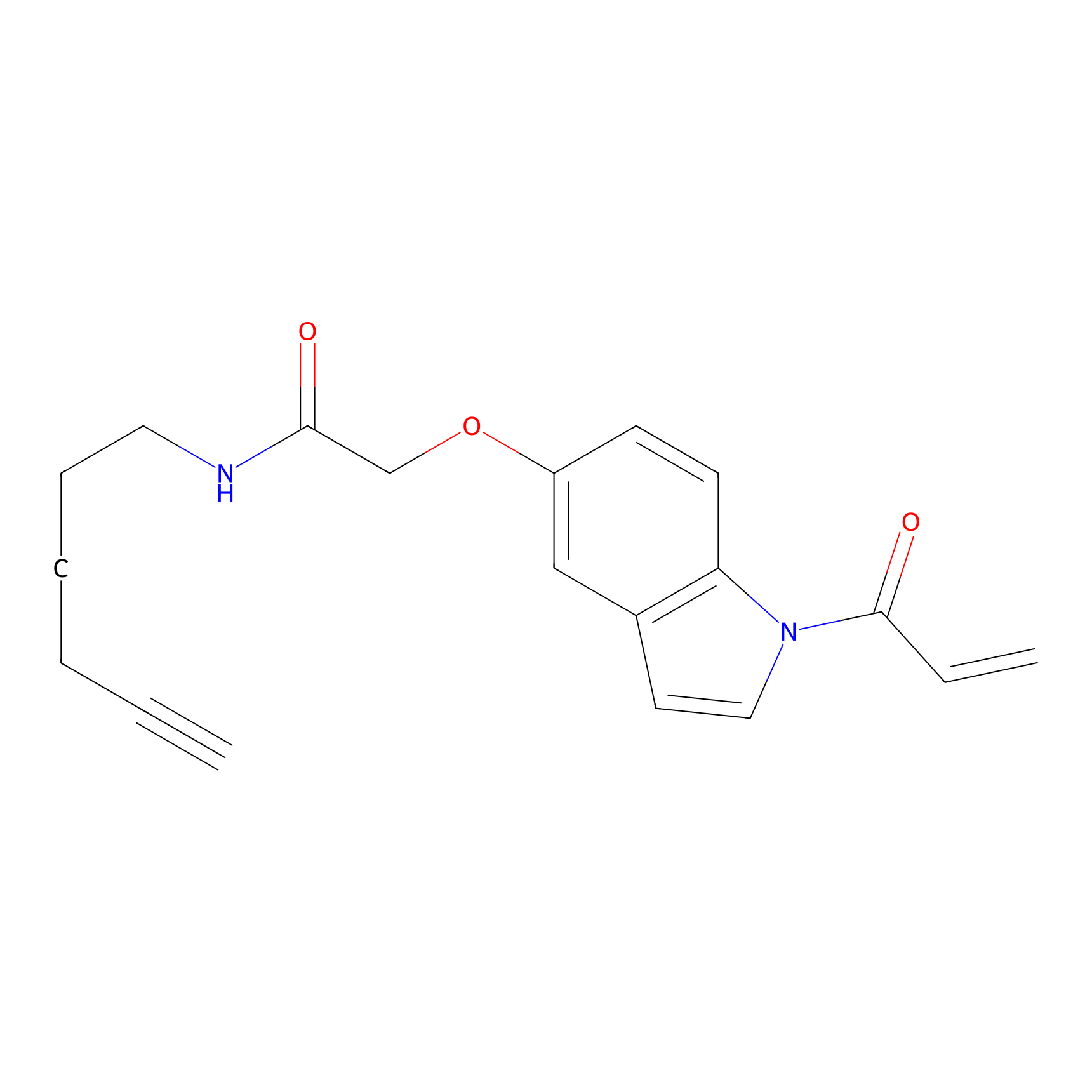 |
N.A. | LDD2223 | [4] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0151 | AZ-11 | HeLa | 10.00 | LDD2154 | [1] |
| LDCM0632 | CL-Sc | Hep-G2 | C611(0.47) | LDD2227 | [4] |
| LDCM0625 | F8 | Ramos | C761(1.39); C137(0.76); C386(2.46); C30(0.76) | LDD2187 | [5] |
| LDCM0572 | Fragment10 | Ramos | C761(3.74); C386(3.15); C30(0.68) | LDD2189 | [5] |
| LDCM0573 | Fragment11 | Ramos | C761(1.23); C137(6.73); C386(1.64); C611(4.29) | LDD2190 | [5] |
| LDCM0574 | Fragment12 | Ramos | C761(2.21); C386(0.86); C30(1.03) | LDD2191 | [5] |
| LDCM0575 | Fragment13 | Ramos | C761(0.95); C30(1.05) | LDD2192 | [5] |
| LDCM0576 | Fragment14 | Ramos | C761(1.00); C386(1.12); C30(0.58); C611(0.84) | LDD2193 | [5] |
| LDCM0579 | Fragment20 | Ramos | C761(1.65); C386(1.30); C30(1.00) | LDD2194 | [5] |
| LDCM0580 | Fragment21 | Ramos | C761(0.96); C386(0.48); C30(0.80) | LDD2195 | [5] |
| LDCM0582 | Fragment23 | Ramos | C761(0.95); C30(1.04) | LDD2196 | [5] |
| LDCM0578 | Fragment27 | Ramos | C761(0.82); C386(1.44); C30(0.84) | LDD2197 | [5] |
| LDCM0586 | Fragment28 | Ramos | C761(0.81); C137(0.41); C386(0.74); C30(1.21) | LDD2198 | [5] |
| LDCM0588 | Fragment30 | Ramos | C761(1.17); C386(0.89); C30(0.86) | LDD2199 | [5] |
| LDCM0589 | Fragment31 | Ramos | C761(0.71); C30(0.83) | LDD2200 | [5] |
| LDCM0590 | Fragment32 | Ramos | C761(18.20); C30(0.72) | LDD2201 | [5] |
| LDCM0468 | Fragment33 | Ramos | C761(1.23); C386(0.98); C30(0.78) | LDD2202 | [5] |
| LDCM0596 | Fragment38 | Ramos | C761(0.56); C386(0.61); C30(0.83) | LDD2203 | [5] |
| LDCM0566 | Fragment4 | Ramos | C761(1.21); C137(0.49); C386(1.12); C30(0.62) | LDD2184 | [5] |
| LDCM0610 | Fragment52 | Ramos | C761(0.93); C30(1.04) | LDD2204 | [5] |
| LDCM0614 | Fragment56 | Ramos | C761(0.76); C30(1.04) | LDD2205 | [5] |
| LDCM0569 | Fragment7 | Ramos | C761(1.40); C137(0.57); C386(1.12); C30(0.66) | LDD2186 | [5] |
| LDCM0571 | Fragment9 | Ramos | C761(2.03); C30(1.00) | LDD2188 | [5] |
| LDCM0022 | KB02 | Ramos | C761(2.87); C137(0.44); C386(1.87); C30(0.52) | LDD2182 | [5] |
| LDCM0023 | KB03 | Jurkat | C386(9.20) | LDD0209 | [2] |
| LDCM0024 | KB05 | RVH421 | C706(1.22) | LDD3320 | [6] |
| LDCM0131 | RA190 | MM1.R | C761(1.21) | LDD0304 | [7] |
References
