Details of the Target
General Information of Target
| Target ID | LDTP13238 | |||||
|---|---|---|---|---|---|---|
| Target Name | Tryptophan--tRNA ligase, mitochondrial (WARS2) | |||||
| Gene Name | WARS2 | |||||
| Gene ID | 10352 | |||||
| Synonyms |
Tryptophan--tRNA ligase, mitochondrial; EC 6.1.1.2; (Mt)TrpRS; Tryptophanyl-tRNA synthetase; TrpRS |
|||||
| 3D Structure | ||||||
| Sequence |
MGLLLPLALCILVLCCGAMSPPQLALNPSALLSRGCNDSDVLAVAGFALRDINKDRKDGY
VLRLNRVNDAQEYRRGGLGSLFYLTLDVLETDCHVLRKKAWQDCGMRIFFESVYGQCKAI FYMNNPSRVLYLAAYNCTLRPVSKKKIYMTCPDCPSSIPTDSSNHQVLEAATESLAKYNN ENTSKQYSLFKVTRASSQWVVGPSYFVEYLIKESPCTKSQASSCSLQSSDSVPVGLCKGS LTRTHWEKFVSVTCDFFESQAPATGSENSAVNQKPTNLPKVEESQQKNTPPTDSPSKAGP RGSVQYLPDLDDKNSQEKGPQEAFPVHLDLTTNPQGETLDISFLFLEPMEEKLVVLPFPK EKARTAECPGPAQNASPLVLPP |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Class-I aminoacyl-tRNA synthetase family
|
|||||
| Subcellular location |
Mitochondrion matrix
|
|||||
| Function | Catalyzes the attachment of tryptophan to tRNA(Trp) in a two-step reaction: tryptophan is first activated by ATP to form Trp-AMP and then transferred to the acceptor end of tRNA(Trp). | |||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
STPyne Probe Info |
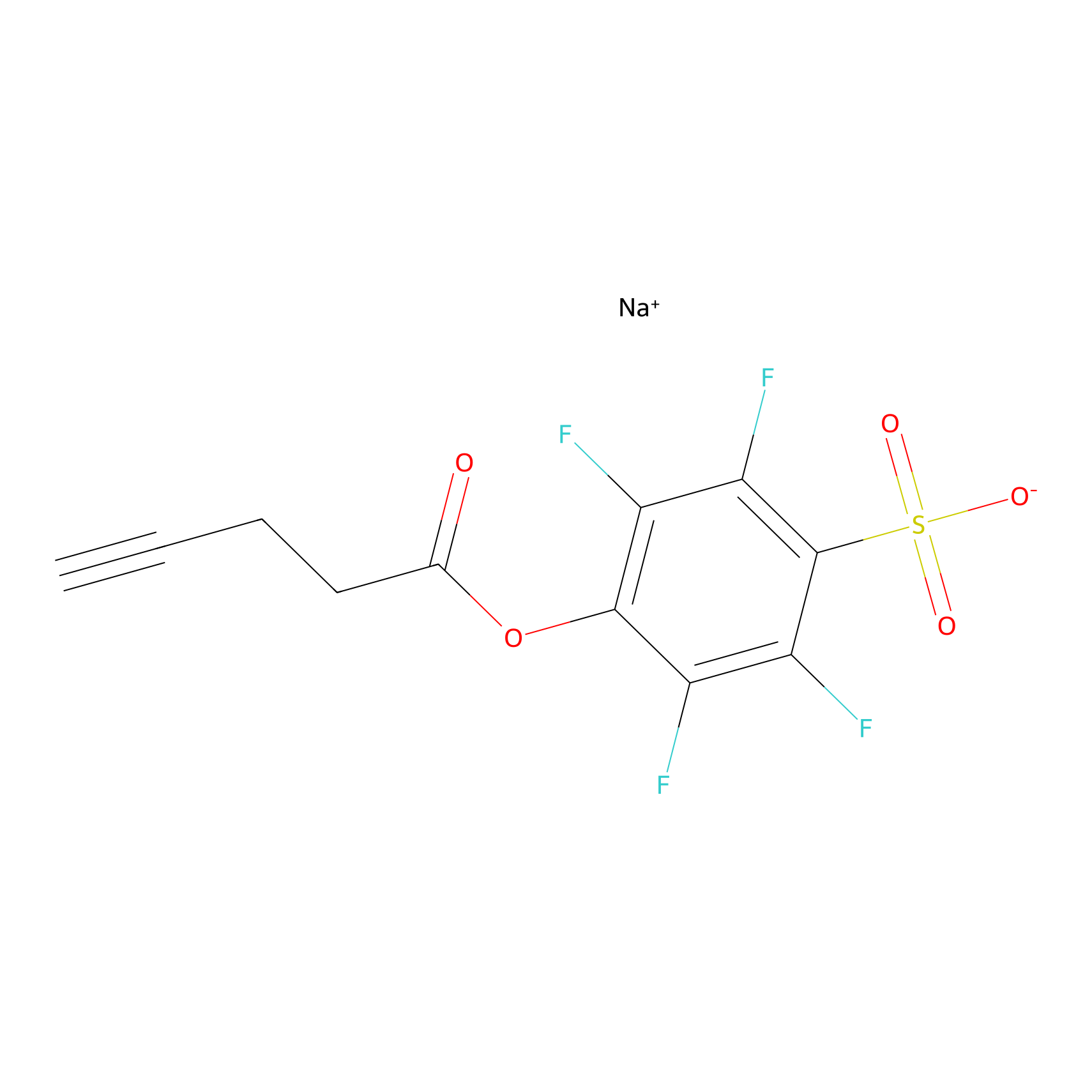 |
K143(6.67); K199(6.25); K226(1.09) | LDD0277 | [1] | |
|
Probe 1 Probe Info |
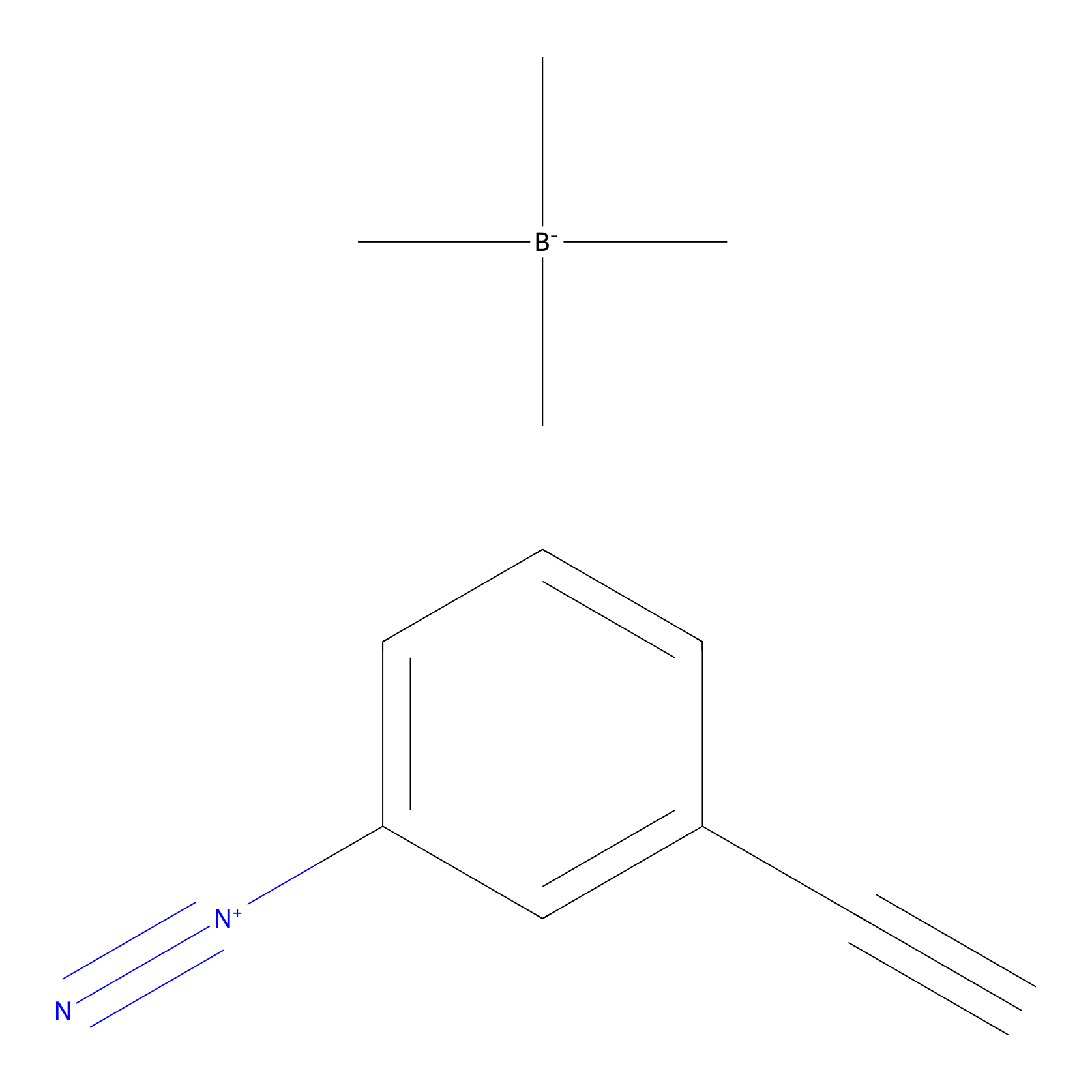 |
Y264(9.80) | LDD3495 | [2] | |
|
DBIA Probe Info |
 |
C102(2.94) | LDD3319 | [3] | |
|
BTD Probe Info |
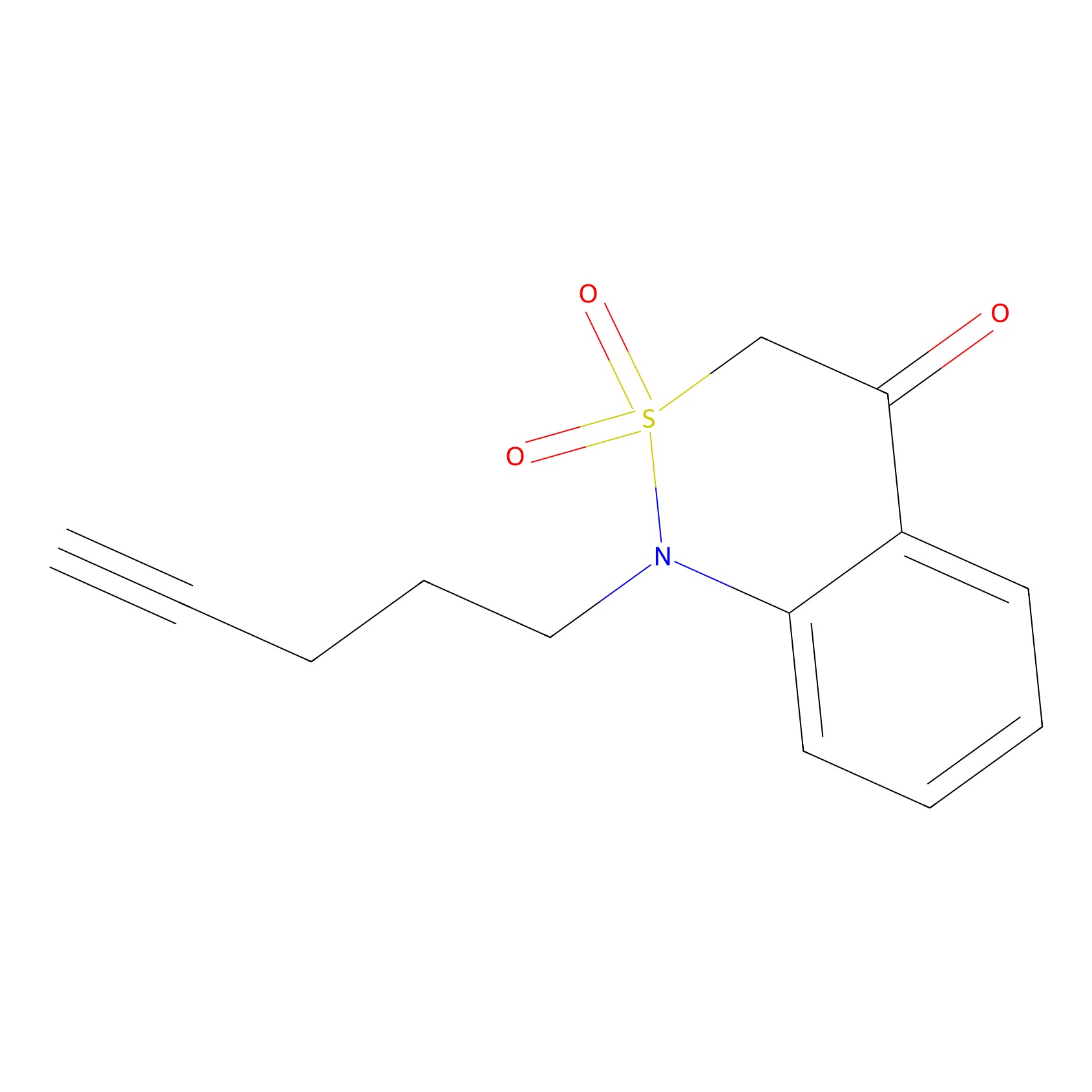 |
C350(1.25) | LDD2117 | [4] | |
|
Acrolein Probe Info |
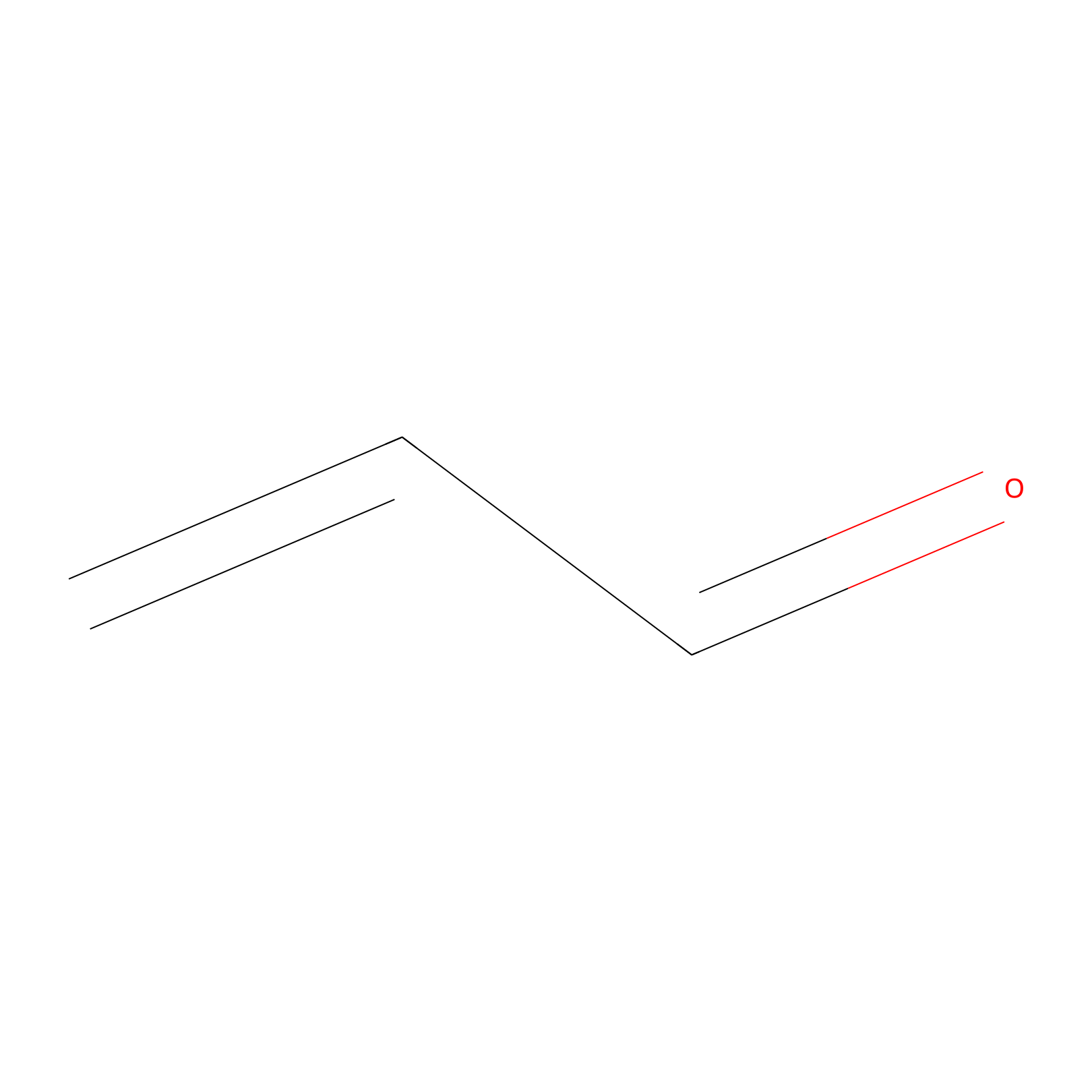 |
N.A. | LDD0224 | [5] | |
|
ATP probe Probe Info |
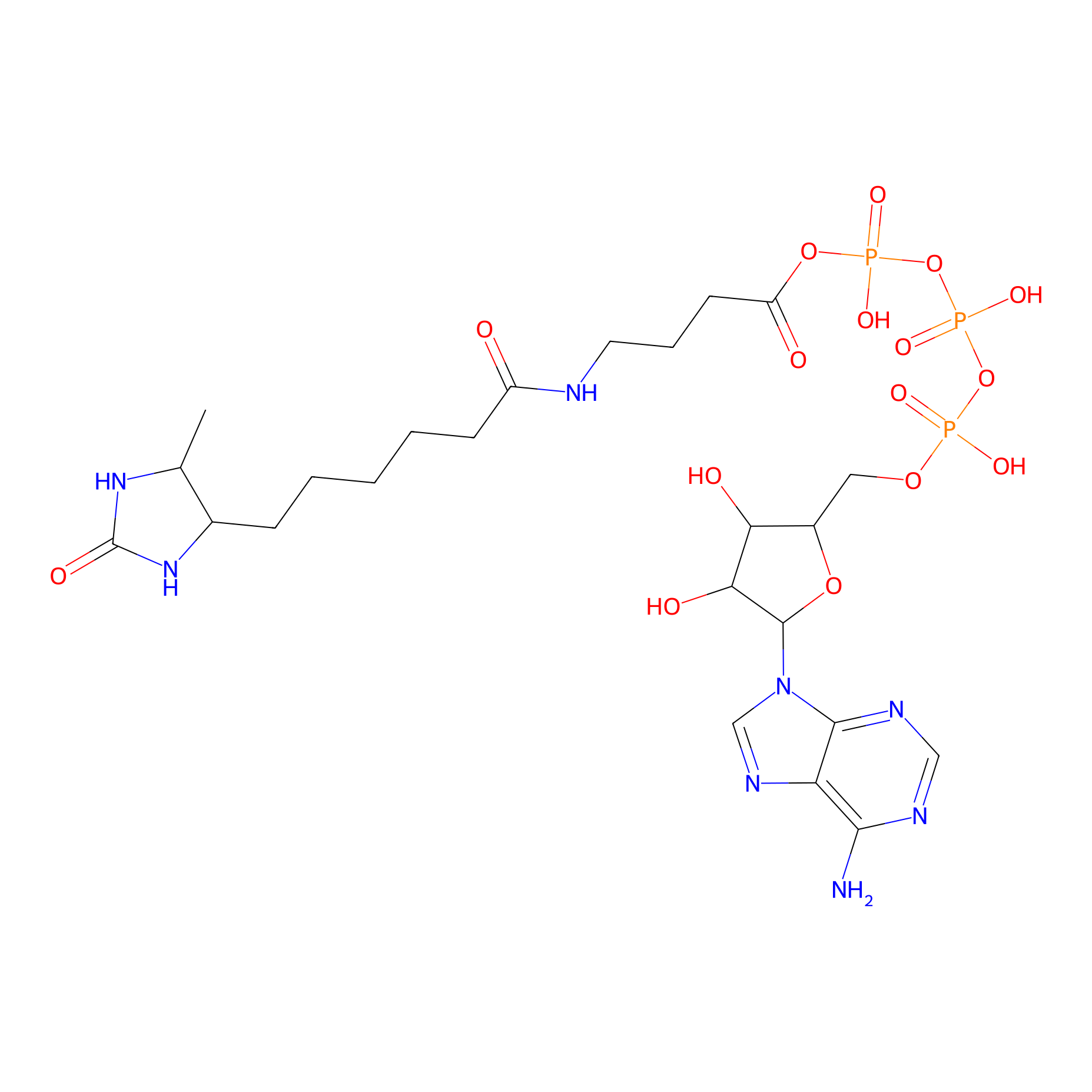 |
K229(0.00); K253(0.00); K226(0.00) | LDD0199 | [6] | |
|
TFBX Probe Info |
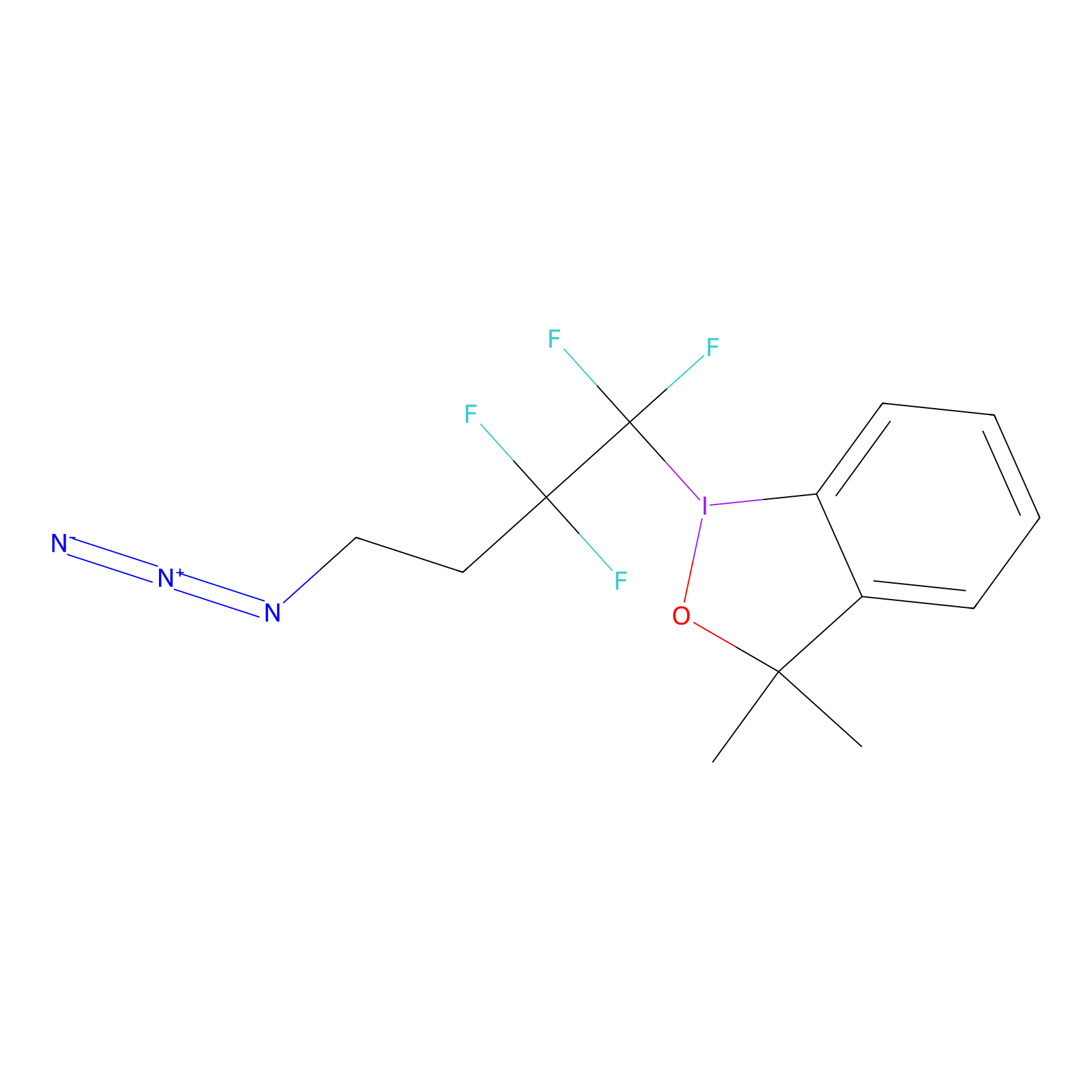 |
N.A. | LDD0027 | [7] | |
PAL-AfBPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
C278 Probe Info |
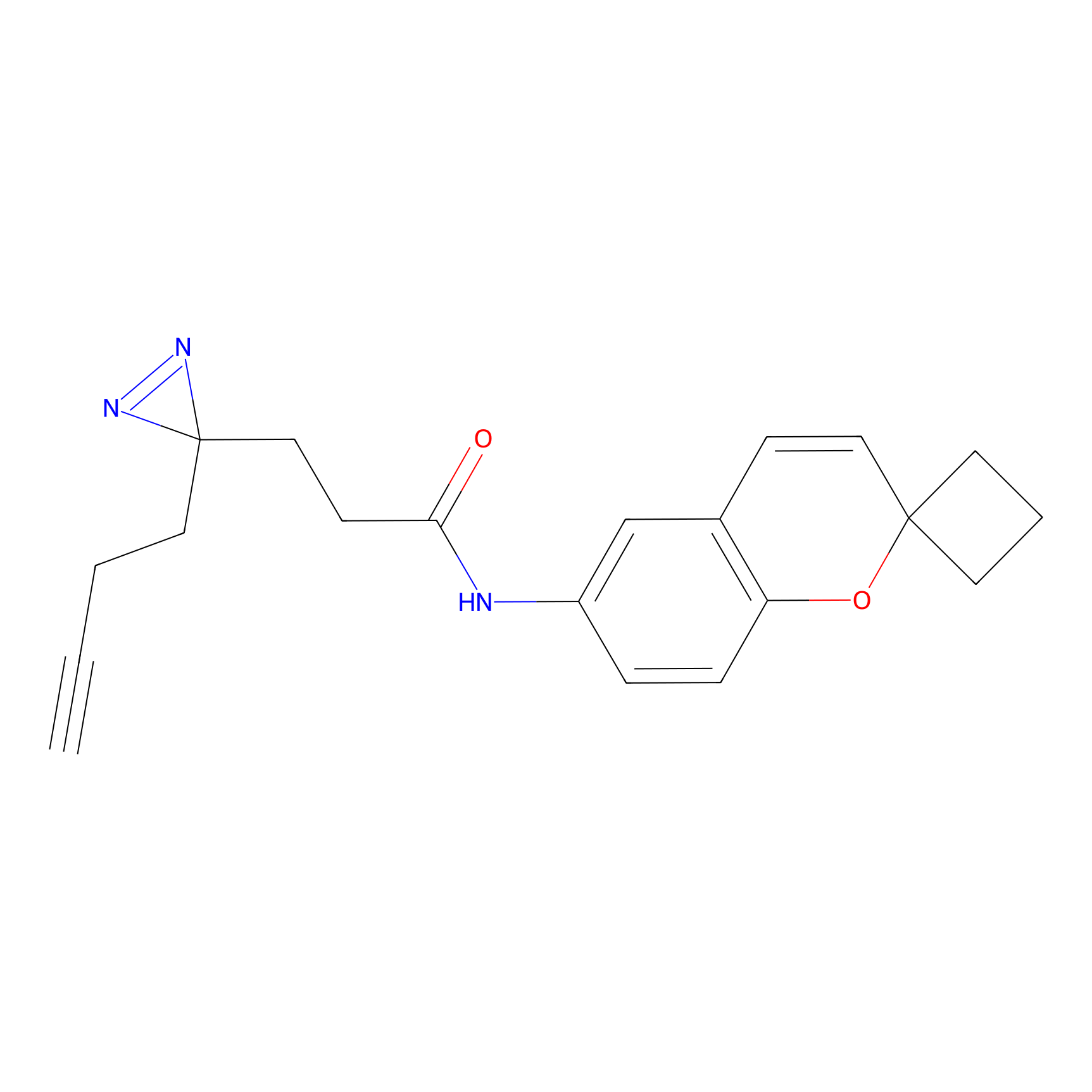 |
81.57 | LDD1948 | [8] | |
|
VE-P Probe Info |
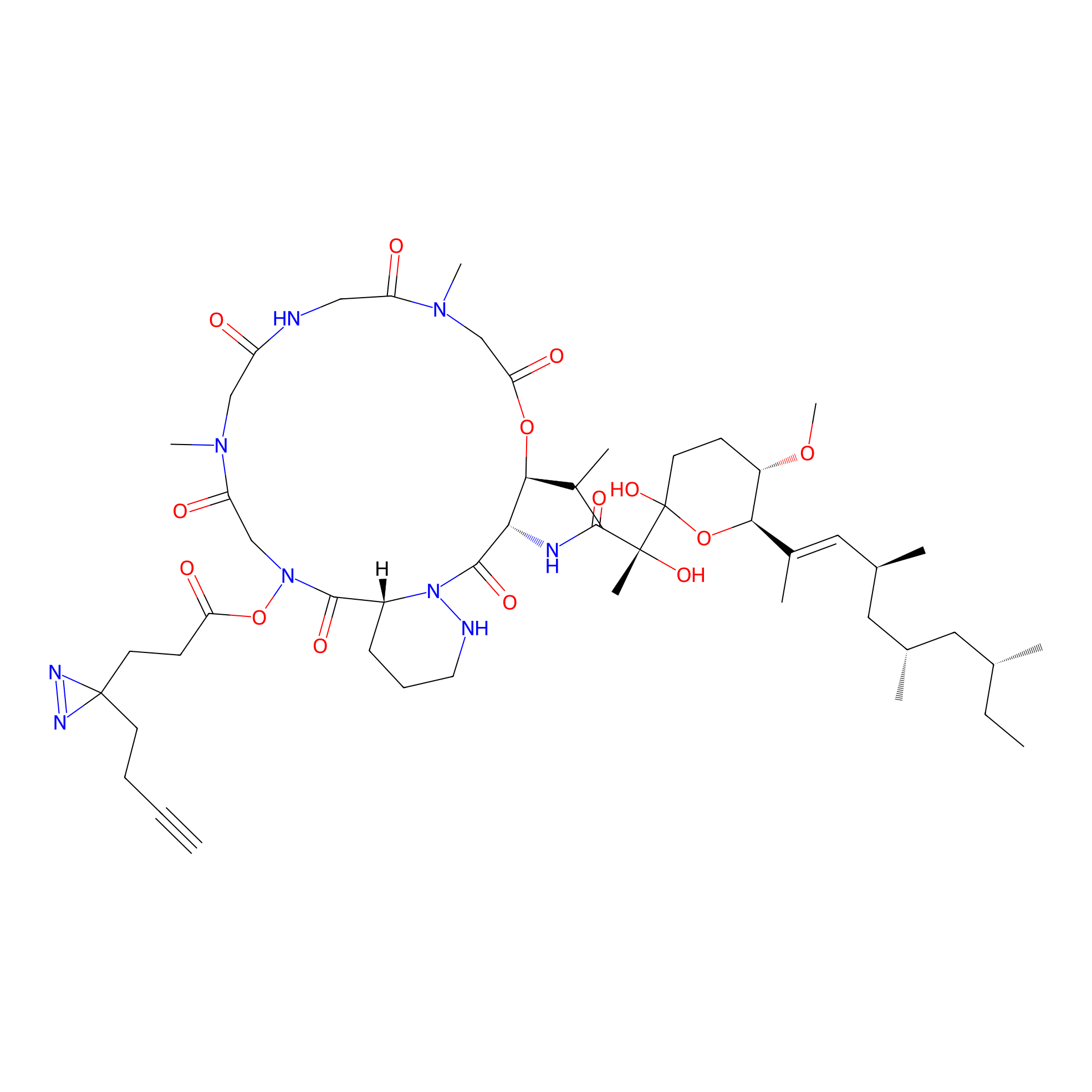 |
N.A. | LDD0396 | [9] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0524 | 2-Cyano-N-(2-morpholin-4-yl-ethyl)-acetamide | MDA-MB-231 | C350(1.25) | LDD2117 | [4] |
| LDCM0022 | KB02 | 22RV1 | C102(1.17) | LDD2243 | [3] |
| LDCM0023 | KB03 | 22RV1 | C102(1.82) | LDD2660 | [3] |
| LDCM0024 | KB05 | MEWO | C102(2.94) | LDD3319 | [3] |
| LDCM0109 | NEM | HeLa | N.A. | LDD0224 | [5] |
The Interaction Atlas With This Target
References
