Details of the Target
General Information of Target
| Target ID | LDTP08504 | |||||
|---|---|---|---|---|---|---|
| Target Name | Prolyl 3-hydroxylase 2 (P3H2) | |||||
| Gene Name | P3H2 | |||||
| Gene ID | 55214 | |||||
| Synonyms |
LEPREL1; MLAT4; Prolyl 3-hydroxylase 2; EC 1.14.11.7; Leprecan-like protein 1; Myxoid liposarcoma-associated protein 4 |
|||||
| 3D Structure | ||||||
| Sequence |
MRERIWAPPLLLLLPLLLPPPLWGGPPDSPRRELELEPGPLQPFDLLYASGAAAYYSGDY
ERAVRDLEAALRSHRRLREIRTRCARHCAARHPLPPPPPGEGPGAELPLFRSLLGRARCY RSCETQRLGGPASRHRVSEDVRSDFQRRVPYNYLQRAYIKLNQLEKAVEAAHTFFVANPE HMEMQQNIENYRATAGVEALQLVDREAKPHMESYNAGVKHYEADDFEMAIRHFEQALREY FVEDTECRTLCEGPQRFEEYEYLGYKAGLYEAIADHYMQVLVCQHECVRELATRPGRLSP IENFLPLHYDYLQFAYYRVGEYVKALECAKAYLLCHPDDEDVLDNVDYYESLLDDSIDPA SIEAREDLTMFVKRHKLESELIKSAAEGLGFSYTEPNYWIRYGGRQDENRVPSGVNVEGA EVHGFSMGKKLSPKIDRDLREGGPLLYENITFVYNSEQLNGTQRVLLDNVLSEEQCRELH SVASGIMLVGDGYRGKTSPHTPNEKFEGATVLKALKSGYEGRVPLKSARLFYDISEKARR IVESYFMLNSTLYFSYTHMVCRTALSGQQDRRNDLSHPIHADNCLLDPEANECWKEPPAY TFRDYSALLYMNDDFEGGEFIFTEMDAKTVTASIKPKCGRMISFSSGGENPHGVKAVTKG KRCAVALWFTLDPLYRELERIQADEVIAILDQEQQGKHELNINPKDEL |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Leprecan family
|
|||||
| Subcellular location |
Endoplasmic reticulum
|
|||||
| Function |
Prolyl 3-hydroxylase that catalyzes the post-translational formation of 3-hydroxyproline on collagens. Contributes to proline 3-hydroxylation of collagen COL4A1 and COL1A1 in tendons, the eye sclera and in the eye lens capsule. Has high activity with the type IV collagen COL4A1, and lower activity with COL1A1. Catalyzes hydroxylation of the first Pro in Gly-Pro-Hyp sequences where Hyp is 4-hydroxyproline. Has no activity on substrates that lack 4-hydroxyproline in the third position.
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
BTD Probe Info |
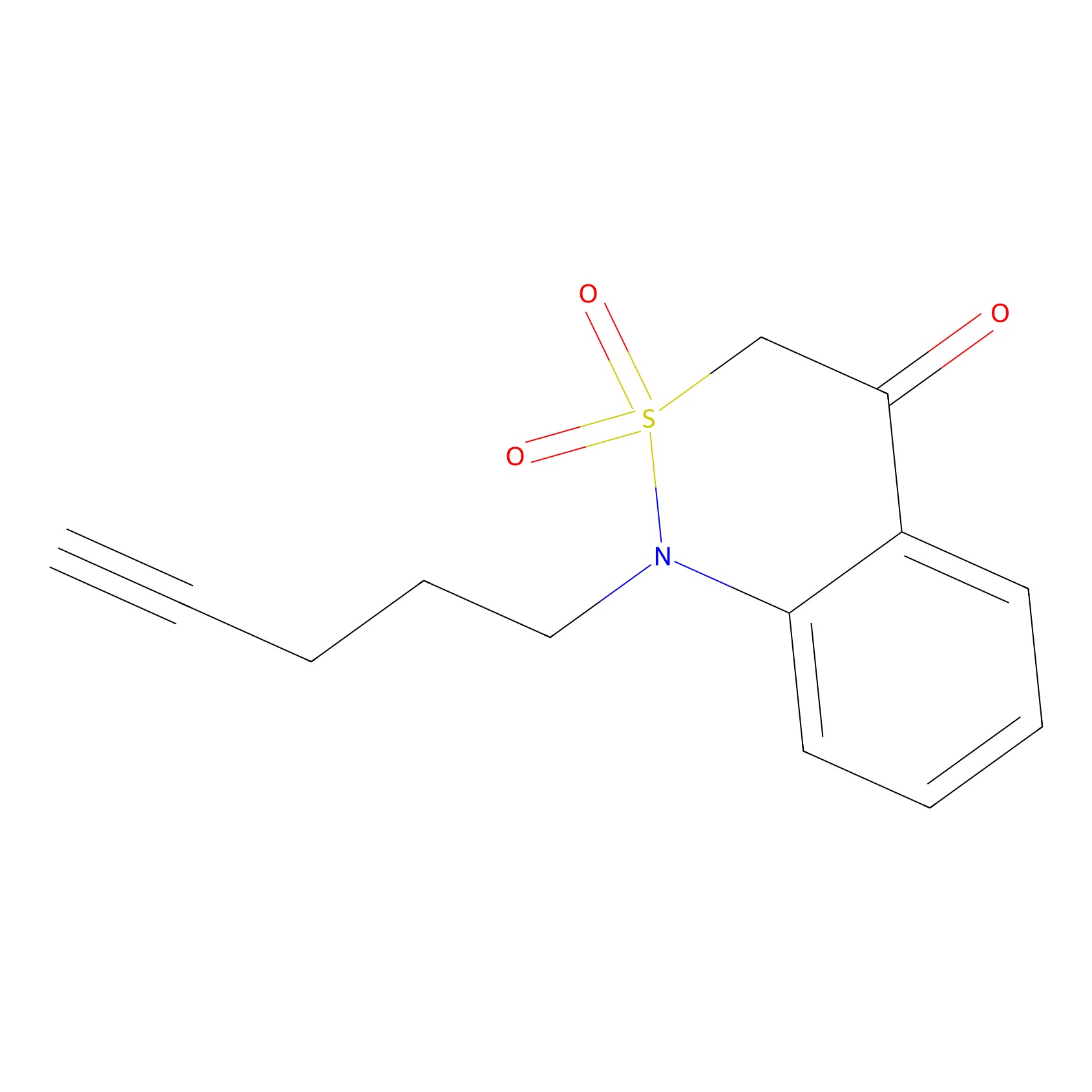 |
C328(1.06) | LDD2090 | [1] | |
|
IPM Probe Info |
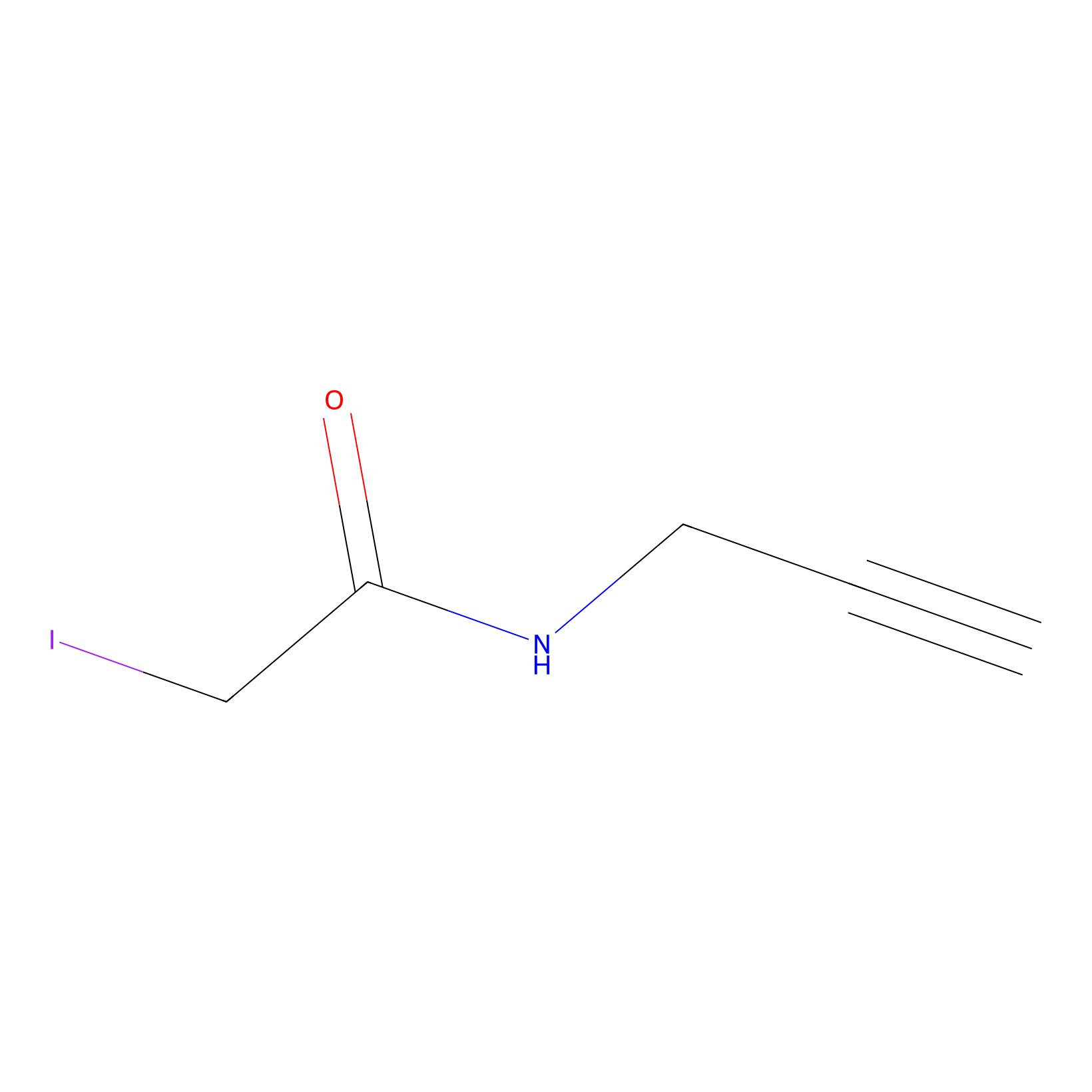 |
C328(10.39) | LDD1701 | [1] | |
|
DBIA Probe Info |
 |
C584(0.99); C593(0.99) | LDD0078 | [2] | |
|
5E-2FA Probe Info |
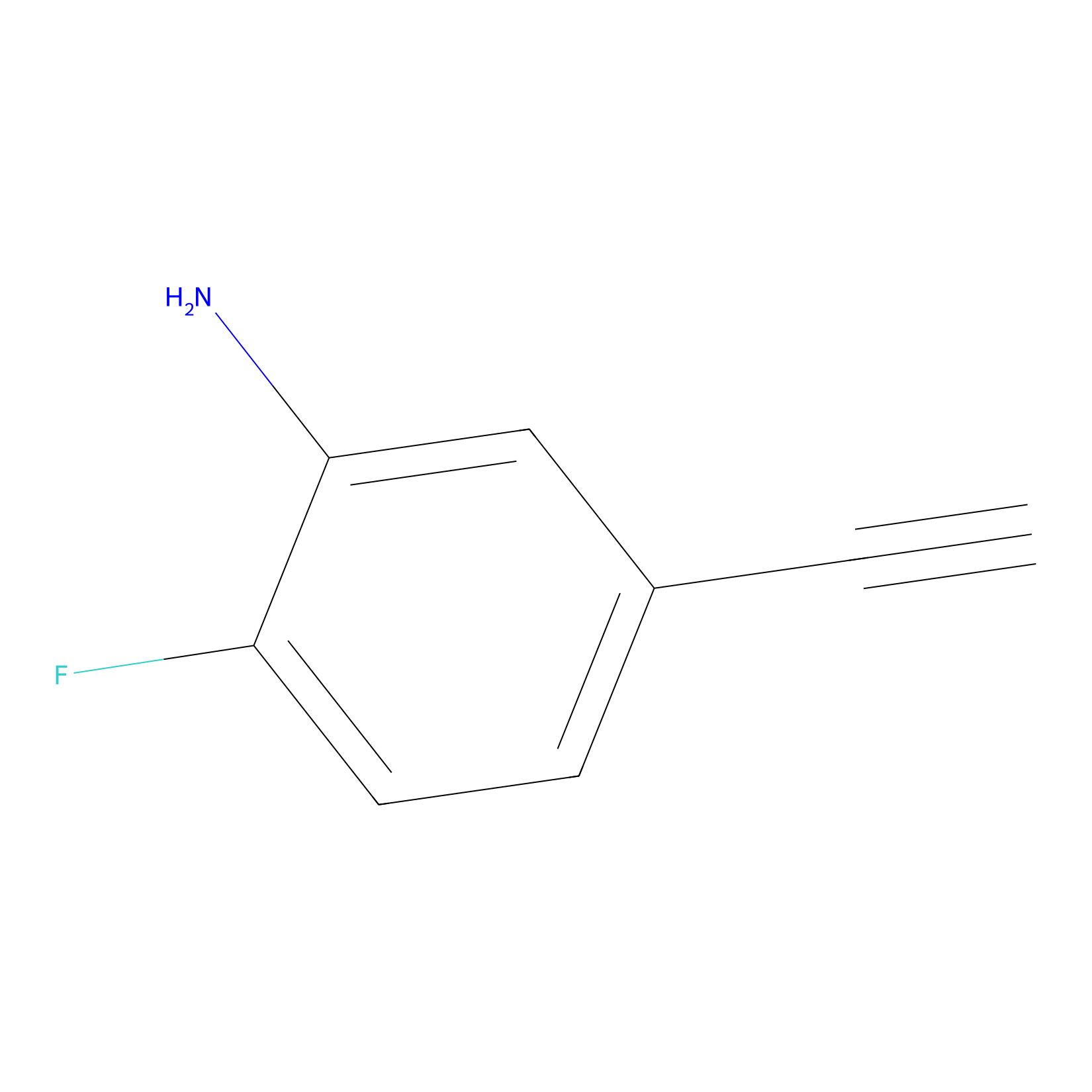 |
H698(0.00); H423(0.00) | LDD2235 | [3] | |
|
m-APA Probe Info |
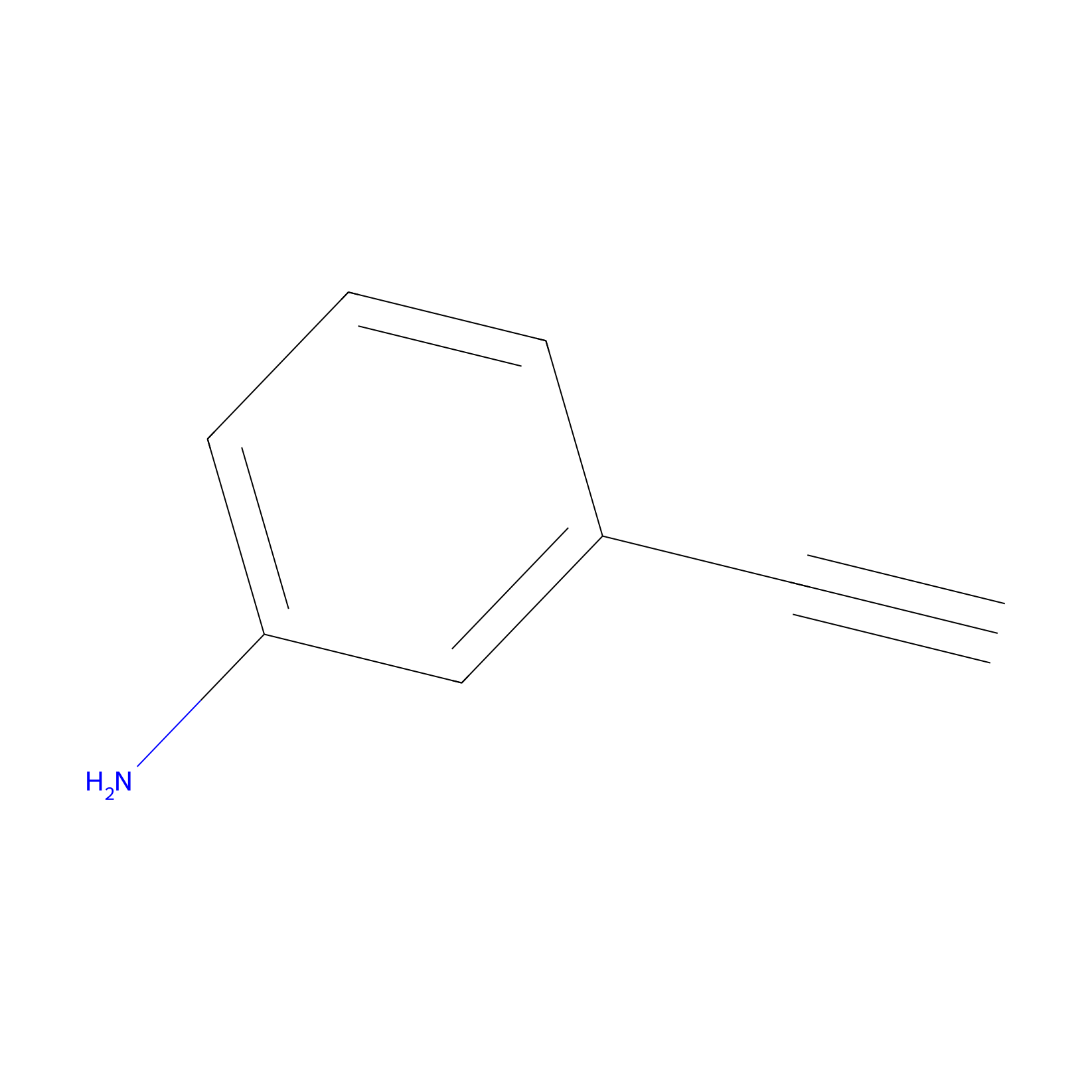 |
H698(0.00); H423(0.00) | LDD2231 | [3] | |
|
Acrolein Probe Info |
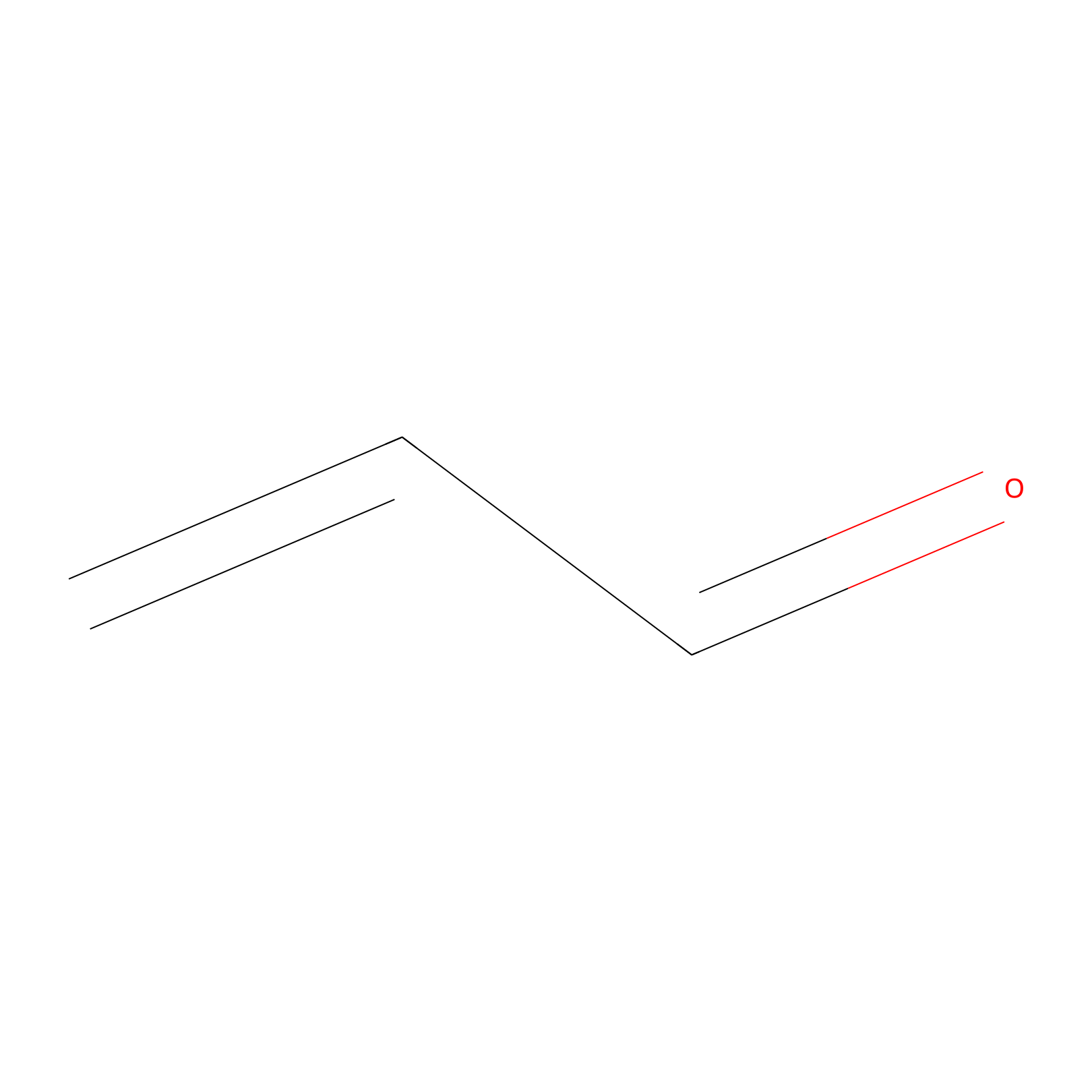 |
H375(0.00); K376(0.00); C476(0.00); H480(0.00) | LDD0217 | [4] | |
|
Crotonaldehyde Probe Info |
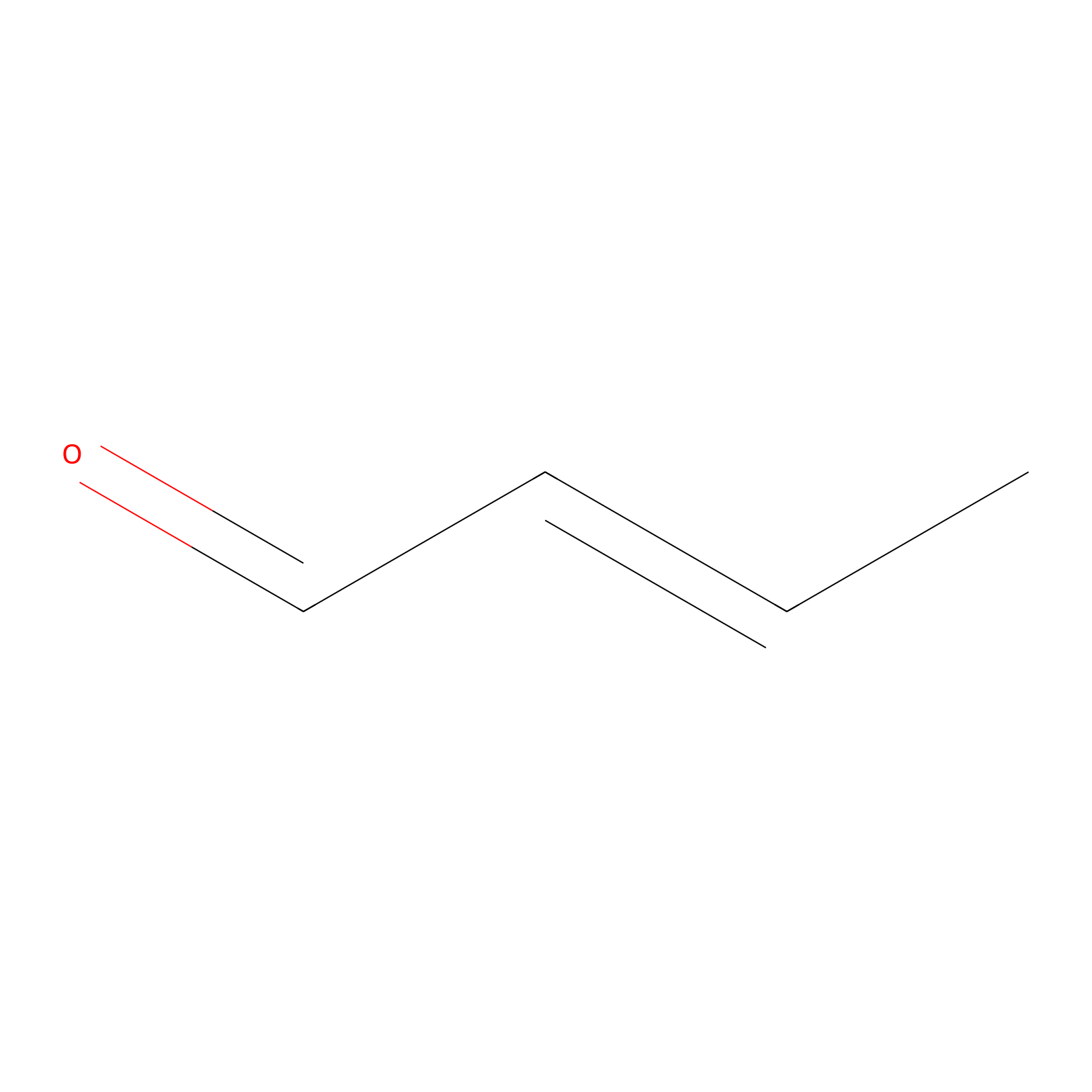 |
C476(0.00); C247(0.00) | LDD0219 | [4] | |
|
Methacrolein Probe Info |
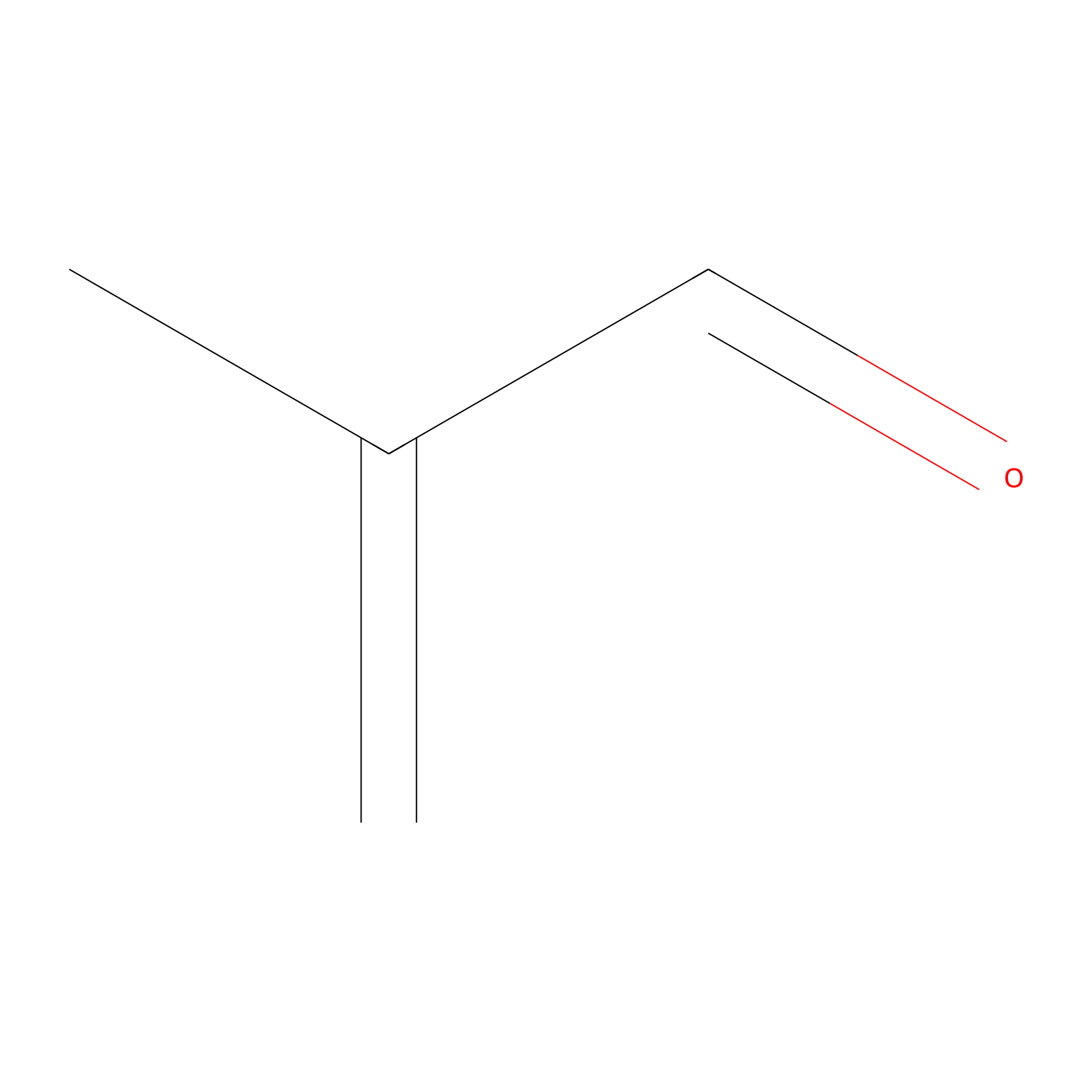 |
C476(0.00); C247(0.00) | LDD0218 | [4] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0510 | 3-(4-(Hydroxydiphenylmethyl)piperidin-1-yl)-3-oxopropanenitrile | MDA-MB-231 | C328(1.12) | LDD2103 | [1] |
| LDCM0020 | ARS-1620 | HCC44 | C584(0.99); C593(0.99) | LDD0078 | [2] |
| LDCM0108 | Chloroacetamide | HeLa | N.A. | LDD0222 | [4] |
| LDCM0022 | KB02 | 786-O | C70(2.48) | LDD2247 | [5] |
| LDCM0023 | KB03 | MDA-MB-231 | C328(10.39) | LDD1701 | [1] |
| LDCM0024 | KB05 | COLO792 | C70(3.26) | LDD3310 | [5] |
| LDCM0509 | N-(4-bromo-3,5-dimethylphenyl)-2-nitroacetamide | MDA-MB-231 | C328(1.06) | LDD2102 | [1] |
| LDCM0109 | NEM | HeLa | H480(0.00); H220(0.00); H232(0.00) | LDD0223 | [4] |
| LDCM0497 | Nucleophilic fragment 11b | MDA-MB-231 | C328(1.06) | LDD2090 | [1] |
| LDCM0499 | Nucleophilic fragment 12b | MDA-MB-231 | C328(1.12) | LDD2092 | [1] |
| LDCM0505 | Nucleophilic fragment 15b | MDA-MB-231 | C328(0.92) | LDD2098 | [1] |
| LDCM0512 | Nucleophilic fragment 19a | MDA-MB-231 | C328(1.68) | LDD2105 | [1] |
| LDCM0527 | Nucleophilic fragment 26b | MDA-MB-231 | C328(0.78) | LDD2120 | [1] |
| LDCM0546 | Nucleophilic fragment 40 | MDA-MB-231 | C328(0.74) | LDD2140 | [1] |
| LDCM0549 | Nucleophilic fragment 43 | MDA-MB-231 | C328(1.06) | LDD2143 | [1] |
| LDCM0553 | Nucleophilic fragment 6b | MDA-MB-231 | C328(1.27) | LDD2147 | [1] |
The Interaction Atlas With This Target
References
