Details of the Target
General Information of Target
| Target ID | LDTP02902 | |||||
|---|---|---|---|---|---|---|
| Target Name | Aldo-keto reductase family 1 member C4 (AKR1C4) | |||||
| Gene Name | AKR1C4 | |||||
| Gene ID | 1109 | |||||
| Synonyms |
CHDR; Aldo-keto reductase family 1 member C4; EC 1.1.1.-; EC 1.1.1.209; EC 1.1.1.210; EC 1.1.1.51; EC 1.1.1.53; EC 1.1.1.62; 3-alpha-hydroxysteroid dehydrogenase type I; 3-alpha-HSD1; EC 1.1.1.357; 3alpha-hydroxysteroid 3-dehydrogenase; Chlordecone reductase; CDR; EC 1.1.1.225; Dihydrodiol dehydrogenase 4; DD-4; DD4; HAKRA
|
|||||
| 3D Structure | ||||||
| Sequence |
MDPKYQRVELNDGHFMPVLGFGTYAPPEVPRNRAVEVTKLAIEAGFRHIDSAYLYNNEEQ
VGLAIRSKIADGSVKREDIFYTSKLWCTFFQPQMVQPALESSLKKLQLDYVDLYLLHFPM ALKPGETPLPKDENGKVIFDTVDLSATWEVMEKCKDAGLAKSIGVSNFNCRQLEMILNKP GLKYKPVCNQVECHPYLNQSKLLDFCKSKDIVLVAHSALGTQRHKLWVDPNSPVLLEDPV LCALAKKHKQTPALIALRYQLQRGVVVLAKSYNEQRIRENIQVFEFQLTSEDMKVLDGLN RNYRYVVMDFLMDHPDYPFSDEY |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Aldo/keto reductase family
|
|||||
| Subcellular location |
Cytoplasm, cytosol
|
|||||
| Function |
Cytosolic aldo-keto reductase that catalyzes the NADH and NADPH-dependent reduction of ketosteroids to hydroxysteroids. Liver specific enzyme that acts as an NAD(P)(H)-dependent 3-, 17- and 20-ketosteroid reductase on the steroid nucleus and side chain. Displays the ability to catalyze both oxidation and reduction in vitro, but most probably acts as a reductase in vivo since the oxidase activity measured in vitro is inhibited by physiological concentration of NADPH. Acts preferentially as a 3-alpha-hydroxysteroid dehydrogenase (HSD) with a subsidiary 3-beta-HSD activity. Catalyzes efficiently the transformation of the potent androgen 5-alpha-dihydrotestosterone (5alpha-DHT or 17beta-hydroxy-5alpha-androstan-3-one) into the less active form, 5-alpha-androstan-3-alpha,17-beta-diol (3-alpha-diol). Catalyzes the reduction of estrone into 17beta-estradiol but with low efficiency. Metabolizes a broad spectrum of natural and synthetic therapeutic steroid and plays an important role in metabolism of androgens, estrogens, progestereone and conjugated steroids. Catalyzes the biotransformation of the pesticide chlordecone (kepone) to its corresponding alcohol leading to increased biliary excretion of the pesticide and concomitant reduction of its neurotoxicity since bile is the major excretory route.
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
| ChEMBL ID | ||||||
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
IPM Probe Info |
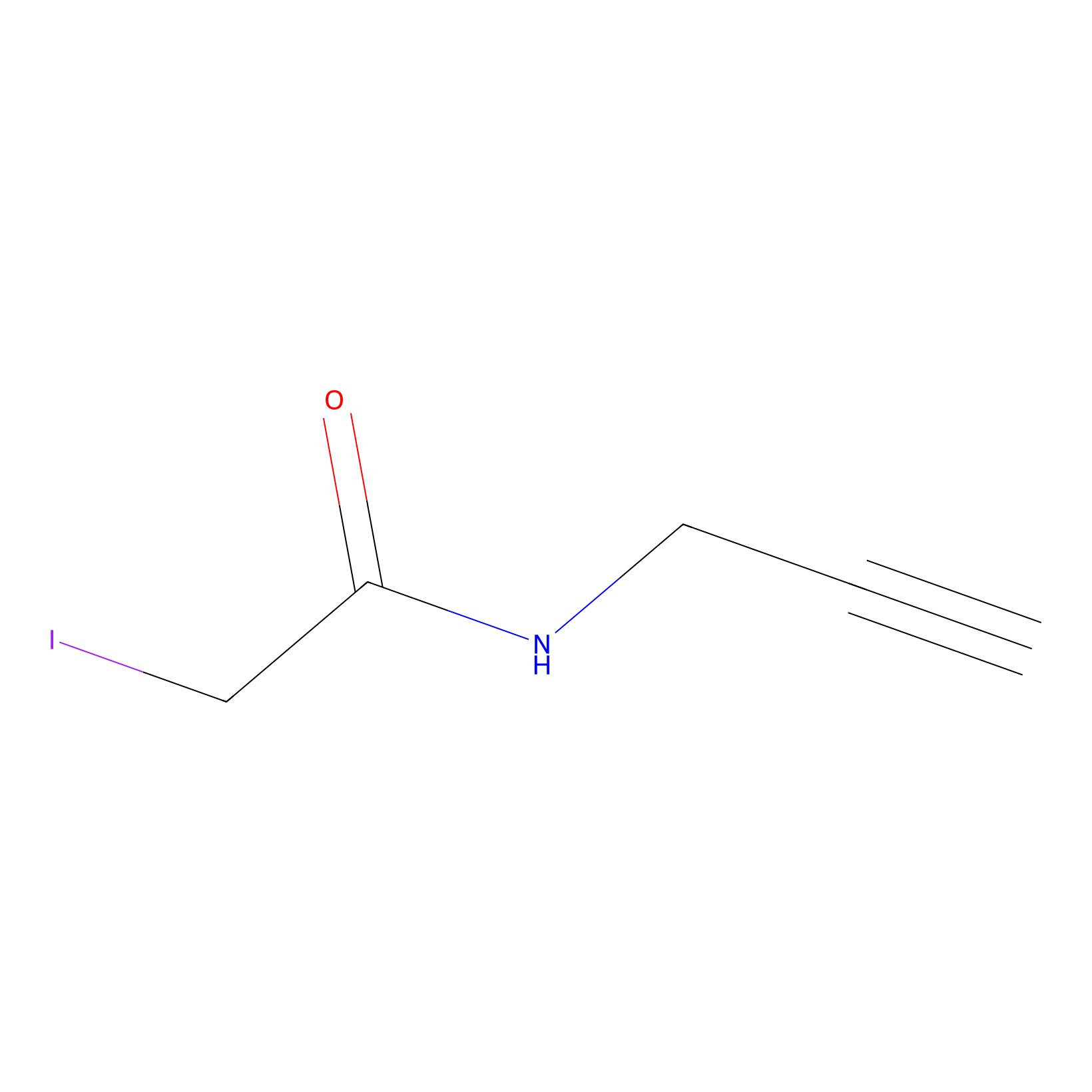 |
C154(0.00); C193(0.00); C188(0.00) | LDD0241 | [1] | |
|
OPA-S-S-alkyne Probe Info |
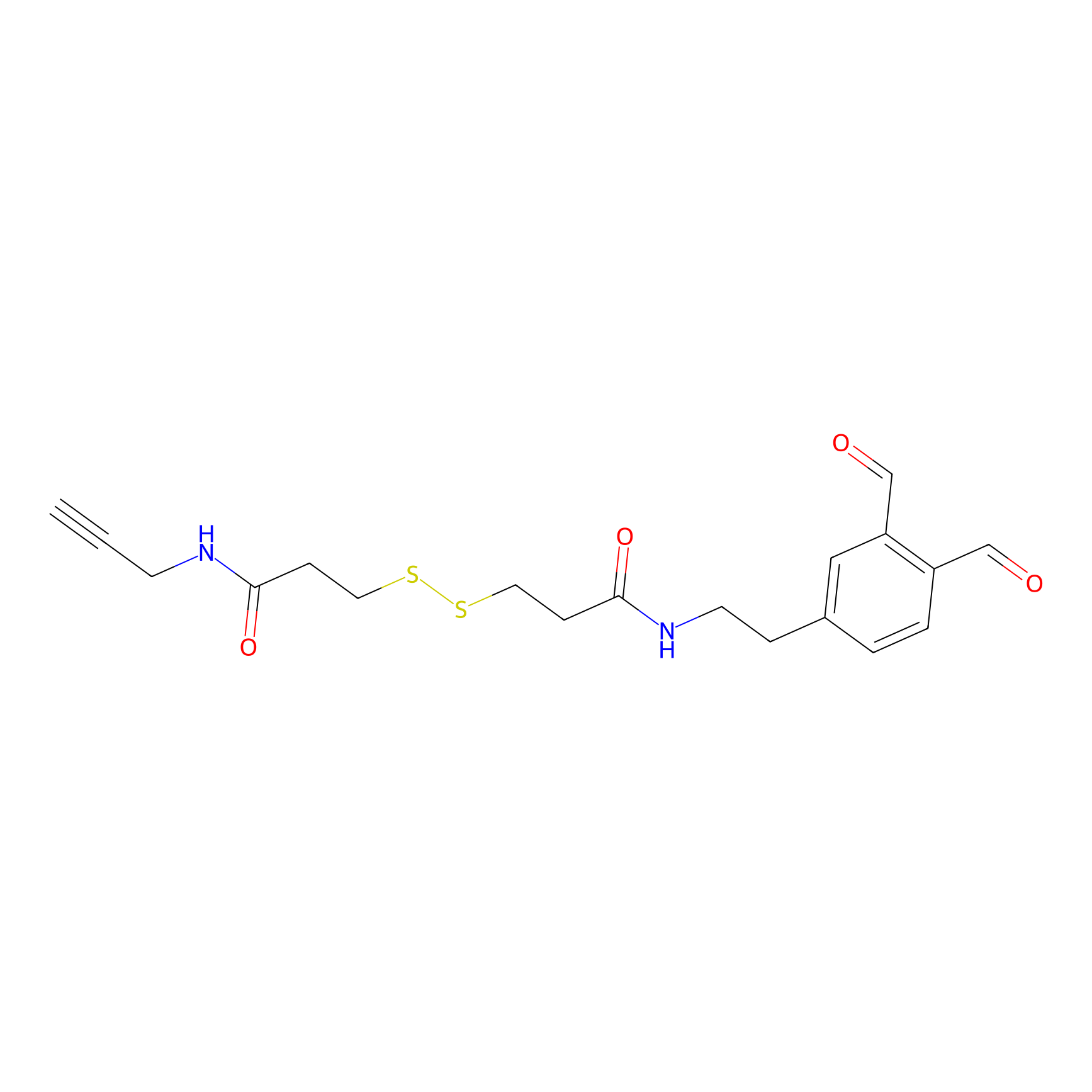 |
K68(2.24); K75(2.78); K270(2.79) | LDD3494 | [2] | |
|
DBIA Probe Info |
 |
C242(0.90) | LDD3364 | [3] | |
|
5E-2FA Probe Info |
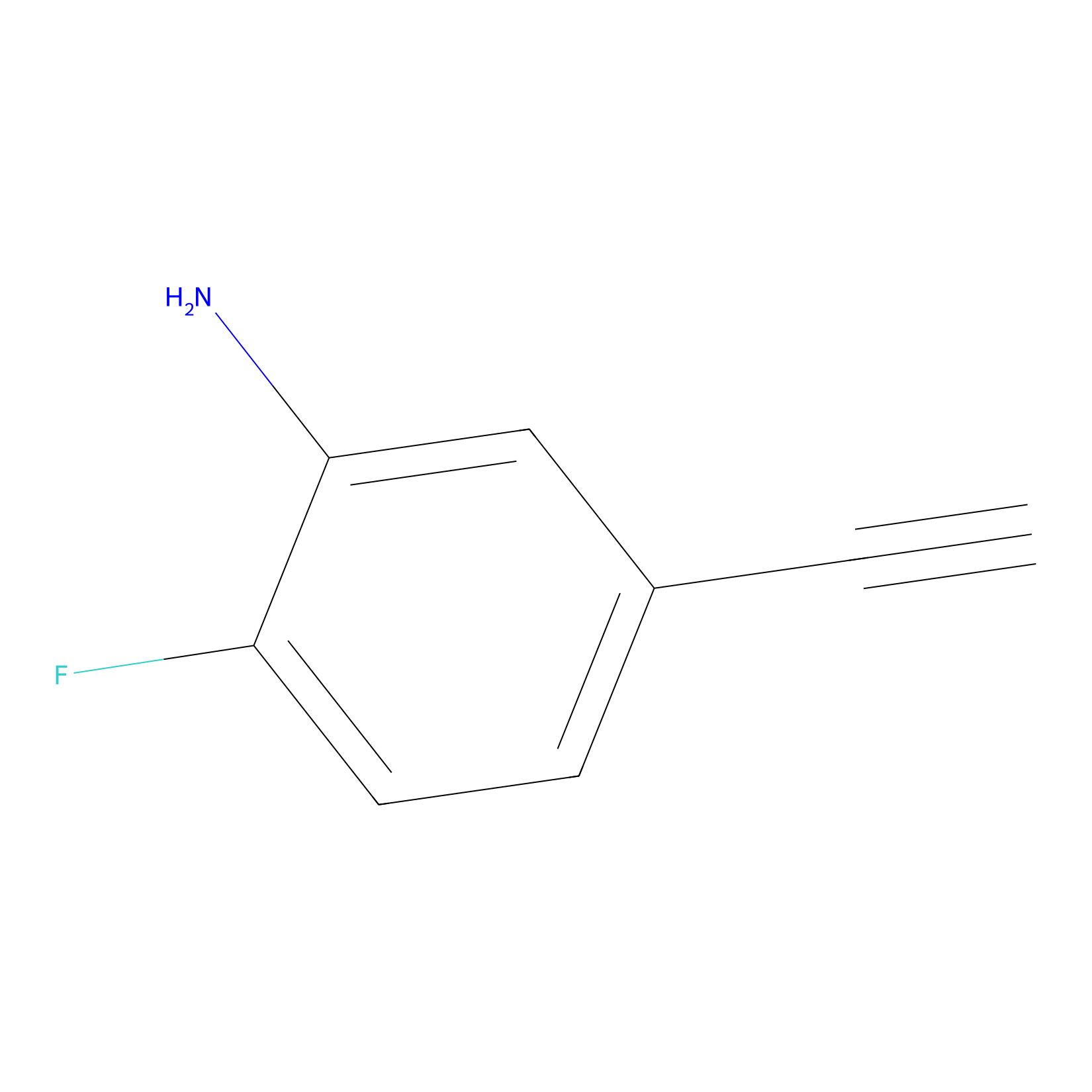 |
N.A. | LDD2235 | [4] | |
|
m-APA Probe Info |
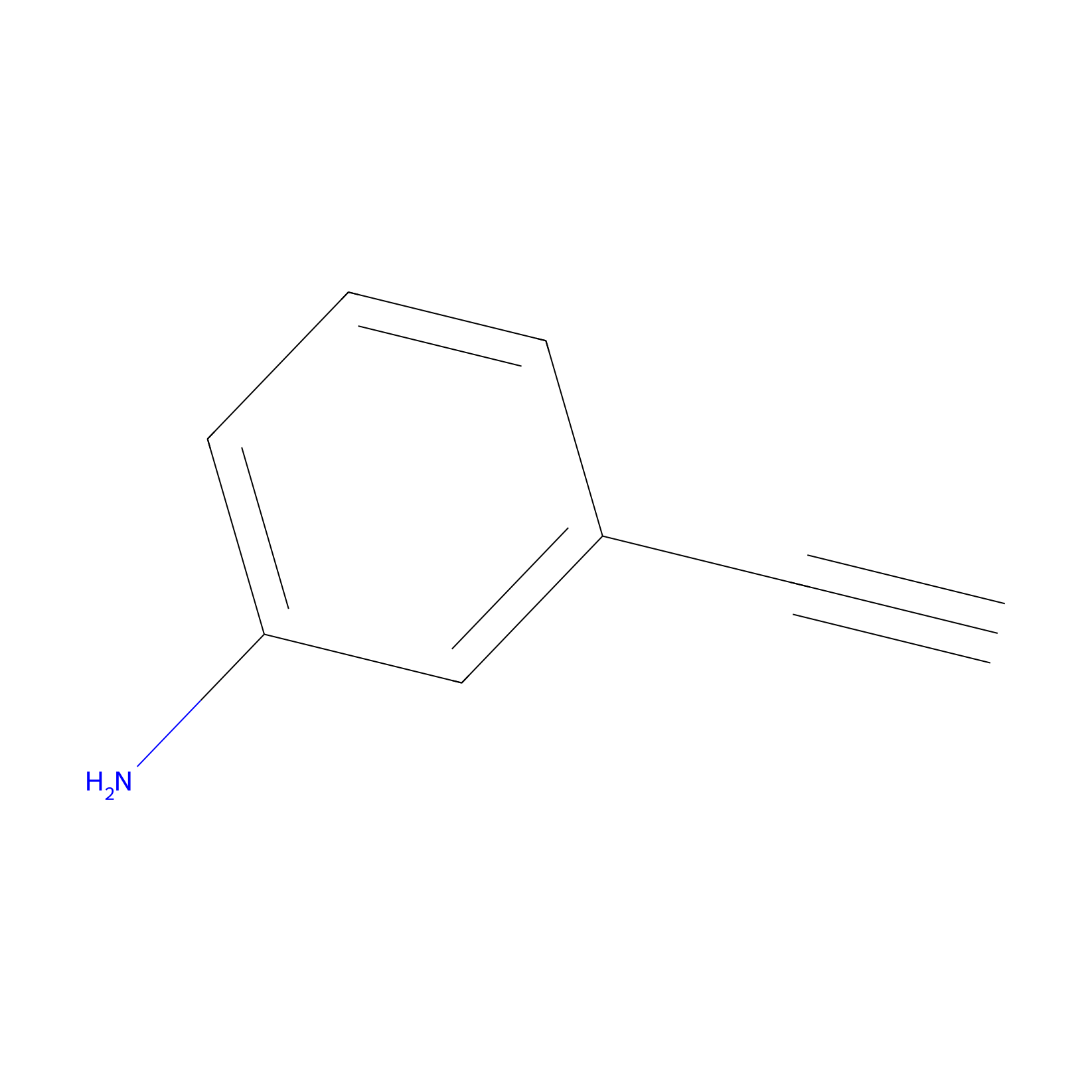 |
N.A. | LDD2231 | [4] | |
|
KY-26 Probe Info |
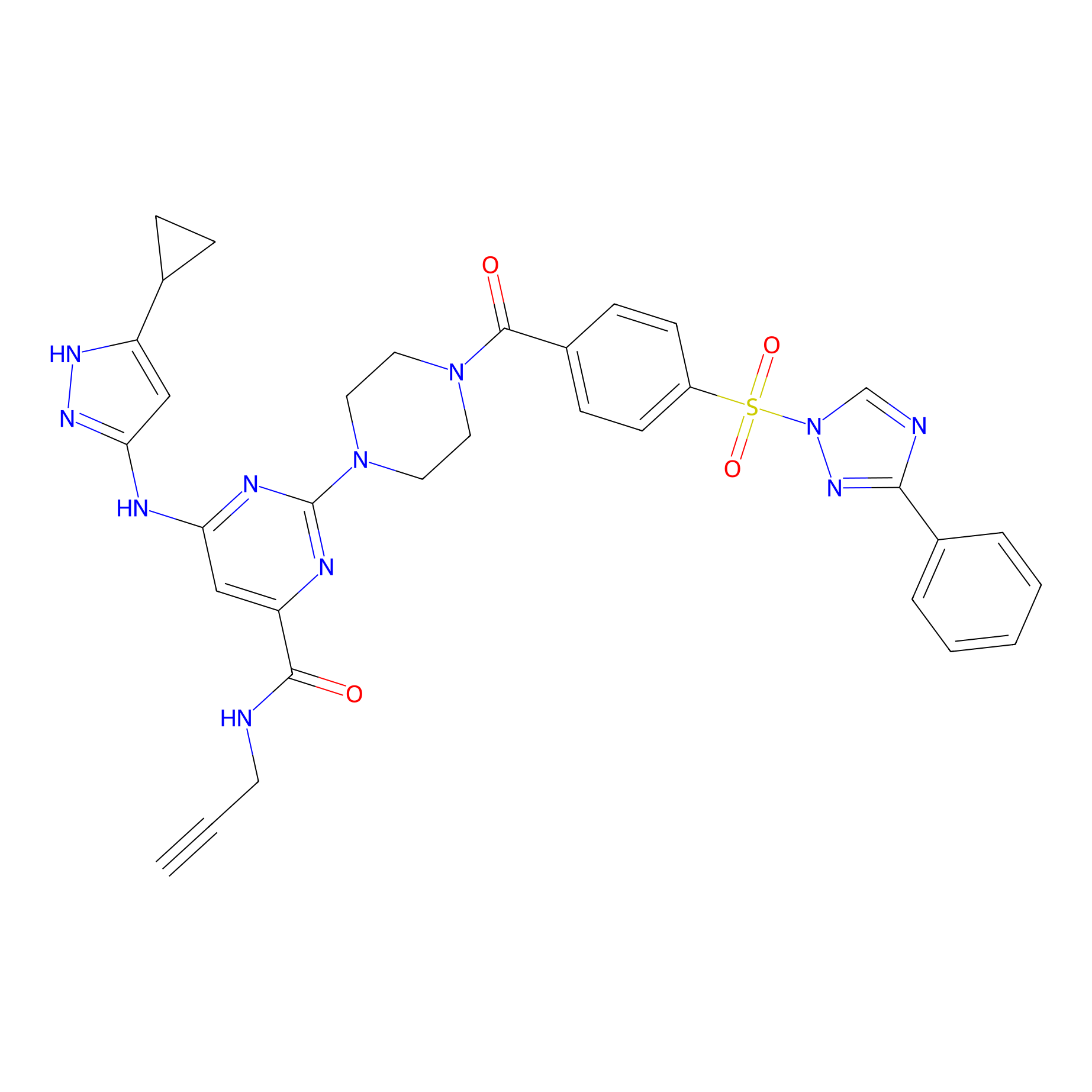 |
N.A. | LDD0301 | [5] | |
|
Acrolein Probe Info |
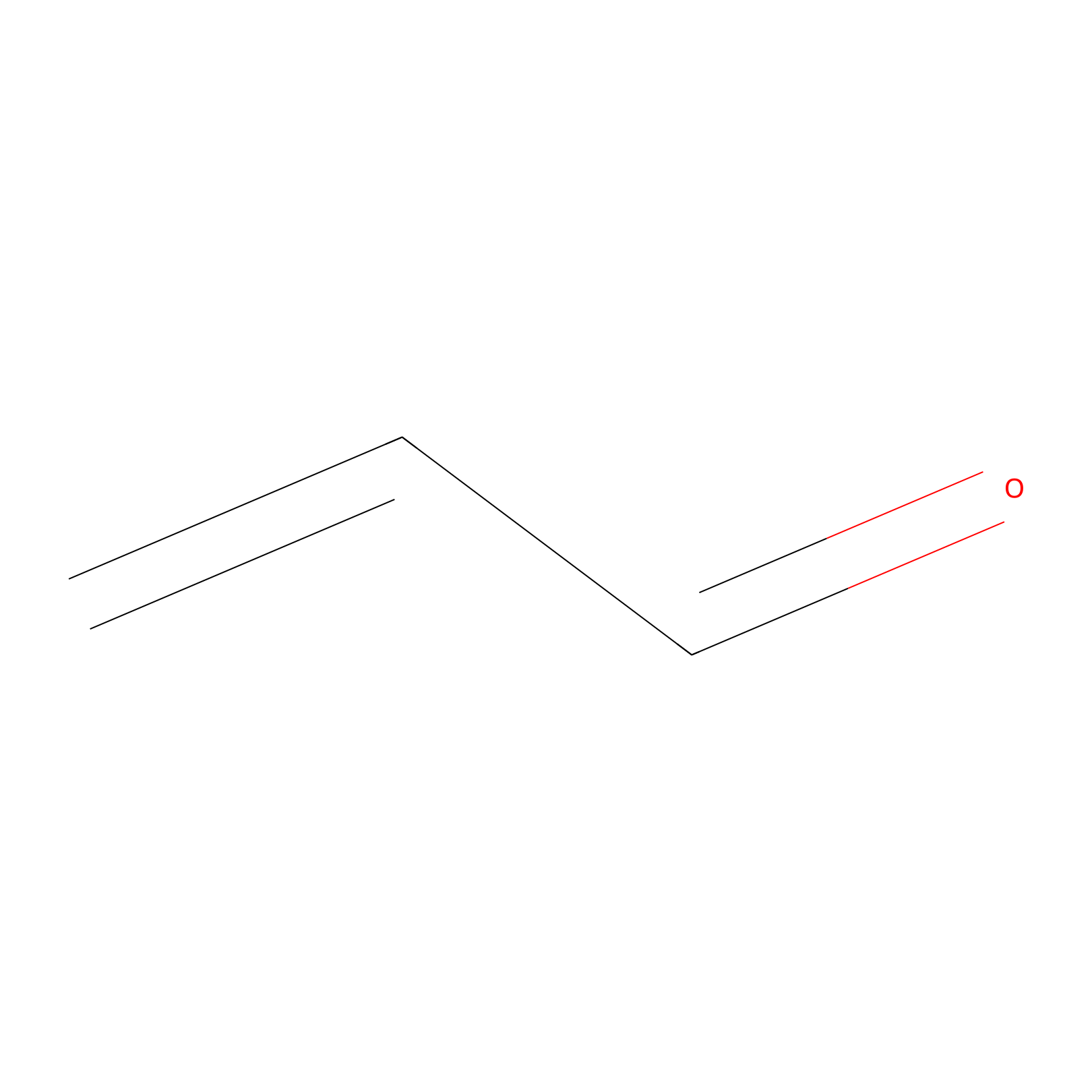 |
C242(0.00); H14(0.00); C206(0.00) | LDD0217 | [6] | |
|
Crotonaldehyde Probe Info |
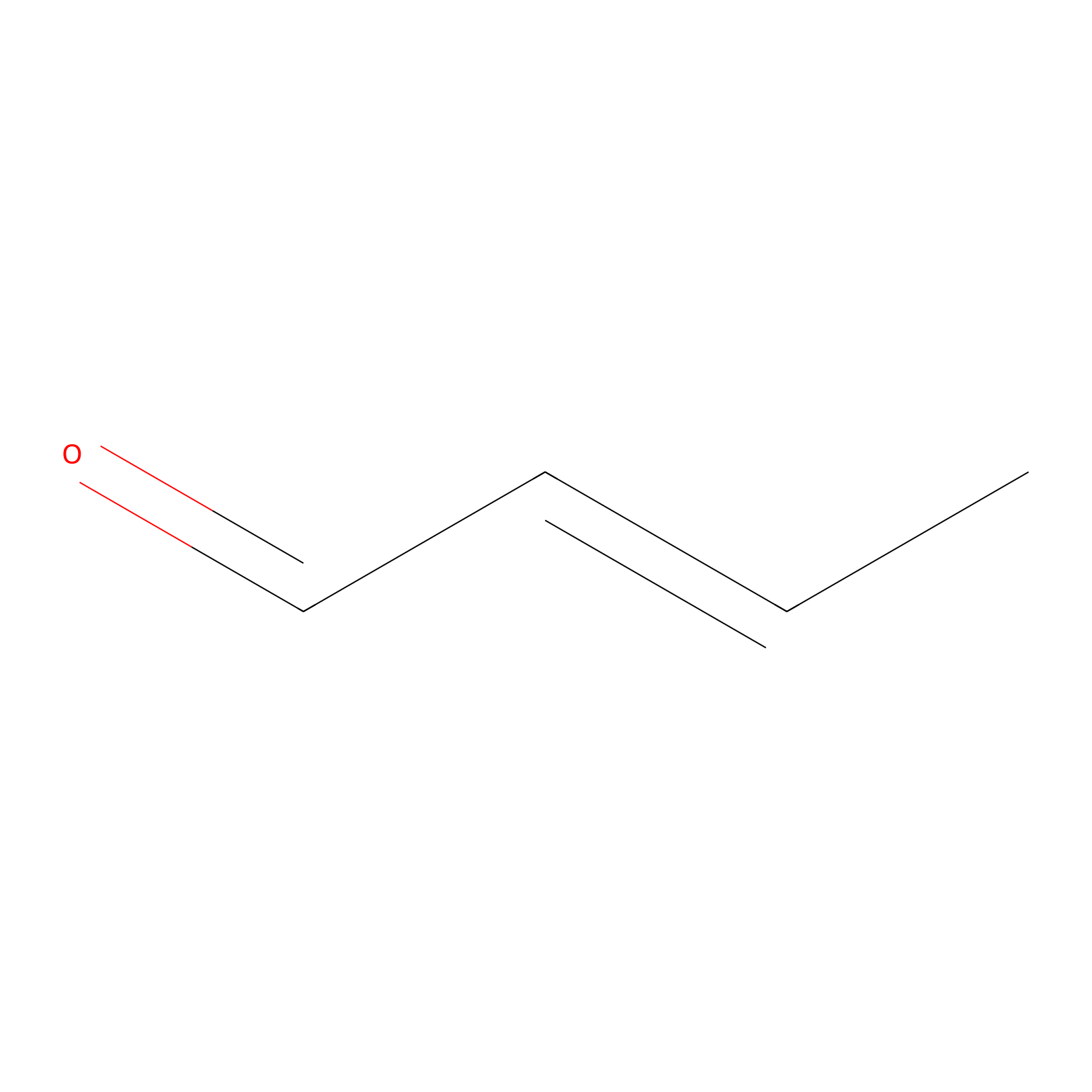 |
N.A. | LDD0219 | [6] | |
|
Methacrolein Probe Info |
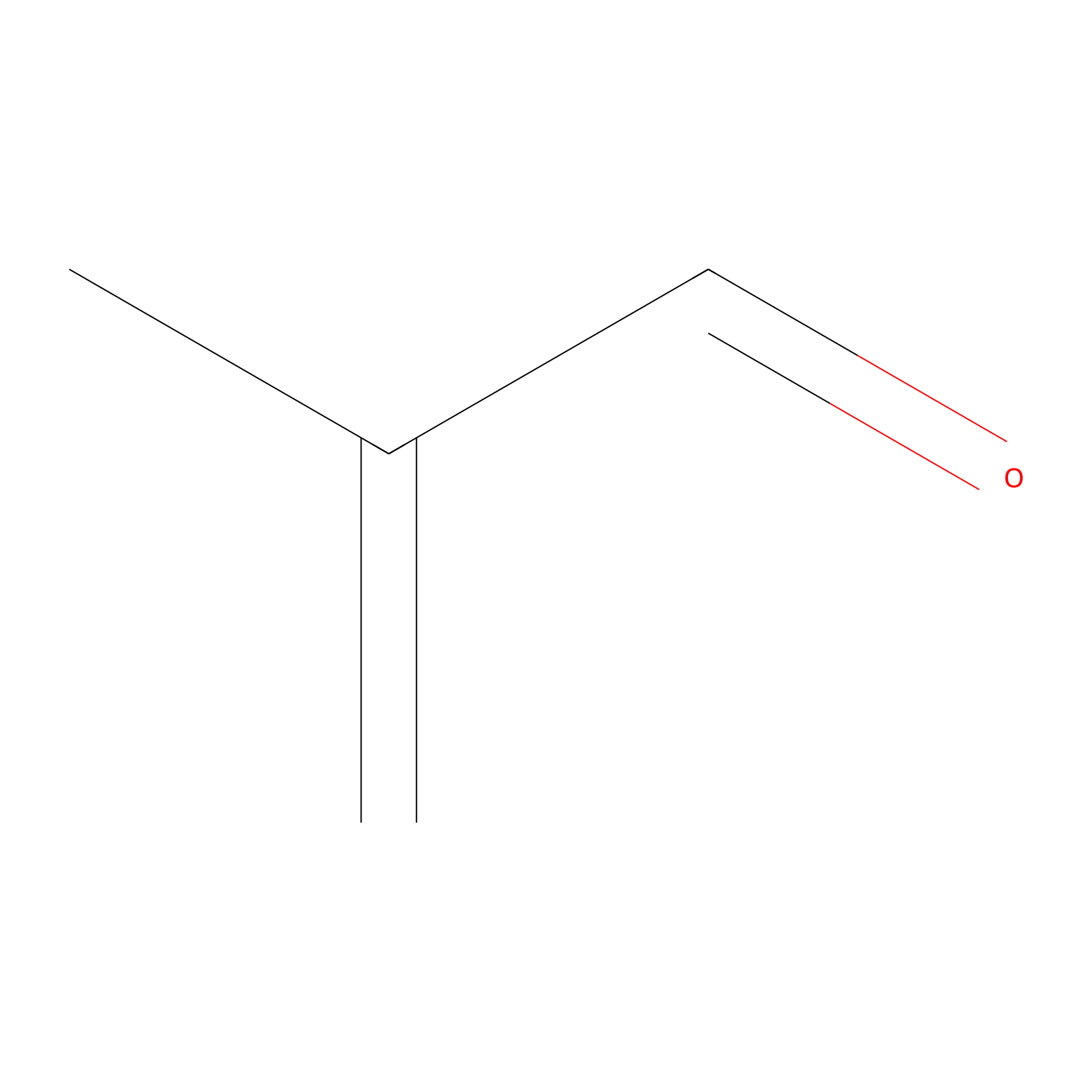 |
C242(0.00); C206(0.00) | LDD0218 | [6] | |
|
NAIA_5 Probe Info |
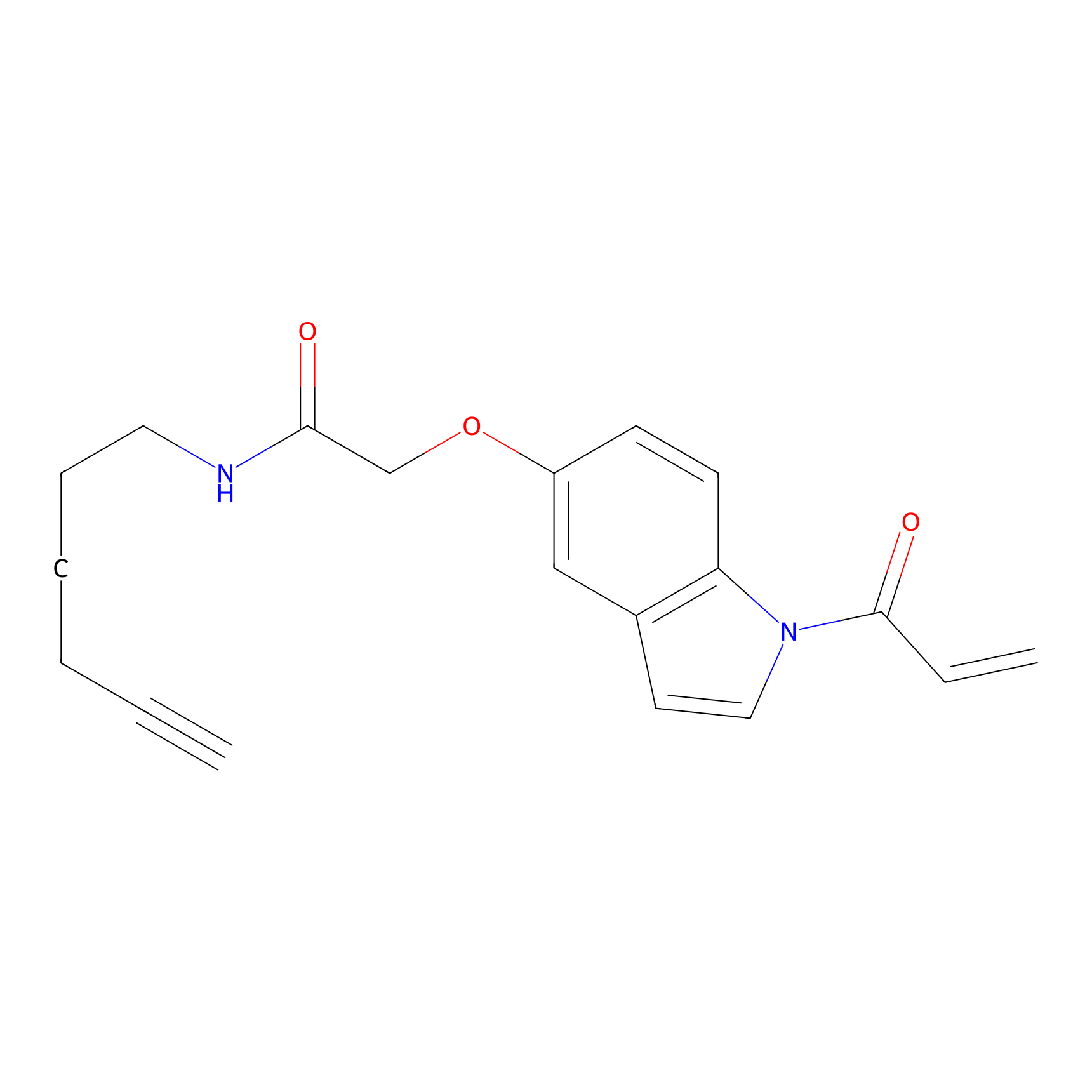 |
N.A. | LDD2223 | [7] | |
PAL-AfBPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
DA-2 Probe Info |
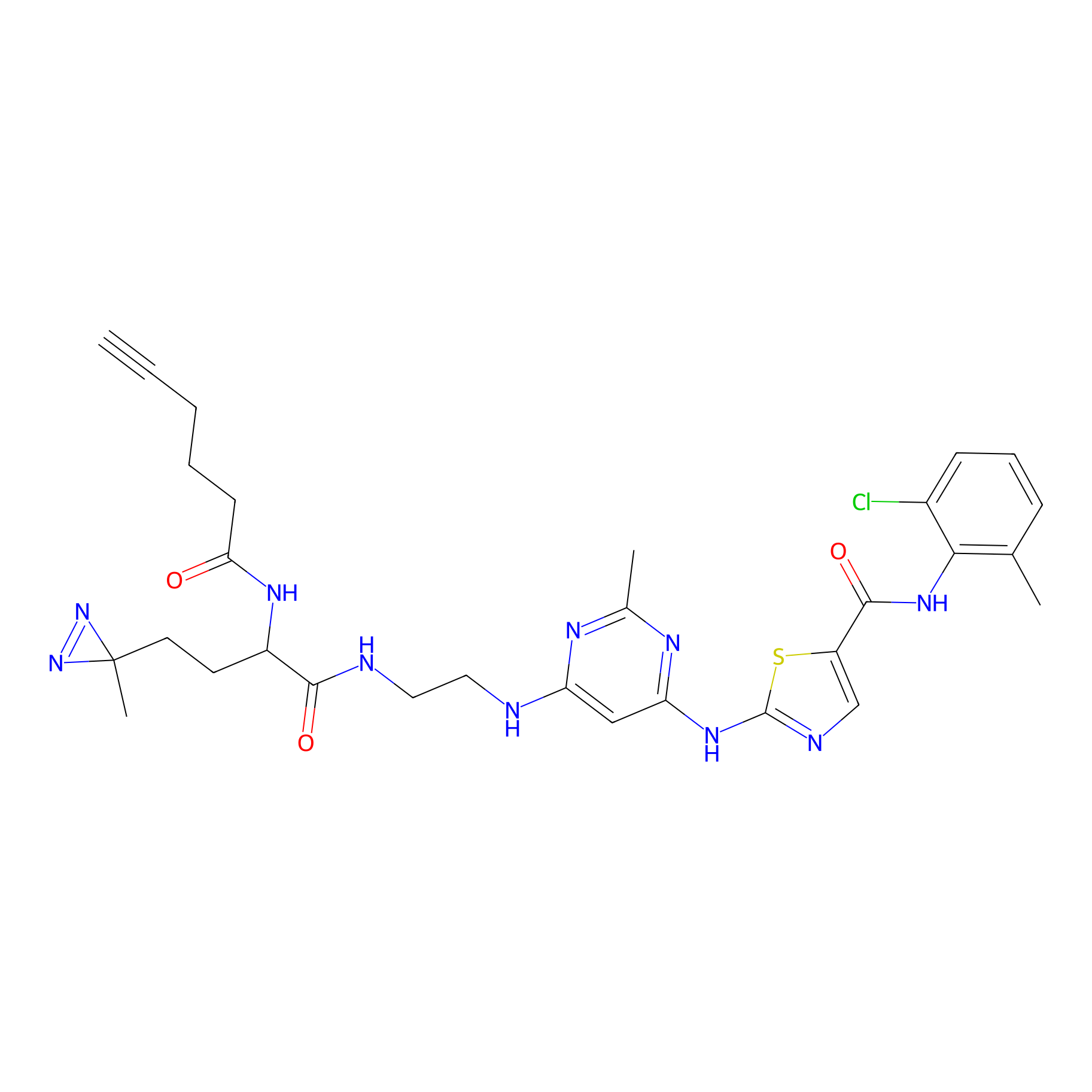 |
N.A. | LDD0073 | [8] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0108 | Chloroacetamide | HeLa | H14(0.00); C206(0.00) | LDD0222 | [6] |
| LDCM0107 | IAA | HeLa | H14(0.00); C206(0.00) | LDD0221 | [6] |
| LDCM0022 | KB02 | 769-P | C242(0.83) | LDD2246 | [3] |
| LDCM0023 | KB03 | 769-P | C242(0.82) | LDD2663 | [3] |
| LDCM0024 | KB05 | NCI-N87 | C242(0.90) | LDD3364 | [3] |
| LDCM0109 | NEM | HeLa | N.A. | LDD0223 | [6] |
The Interaction Atlas With This Target
The Drug(s) Related To This Target
Approved
References
