Details of the Target
General Information of Target
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
STPyne Probe Info |
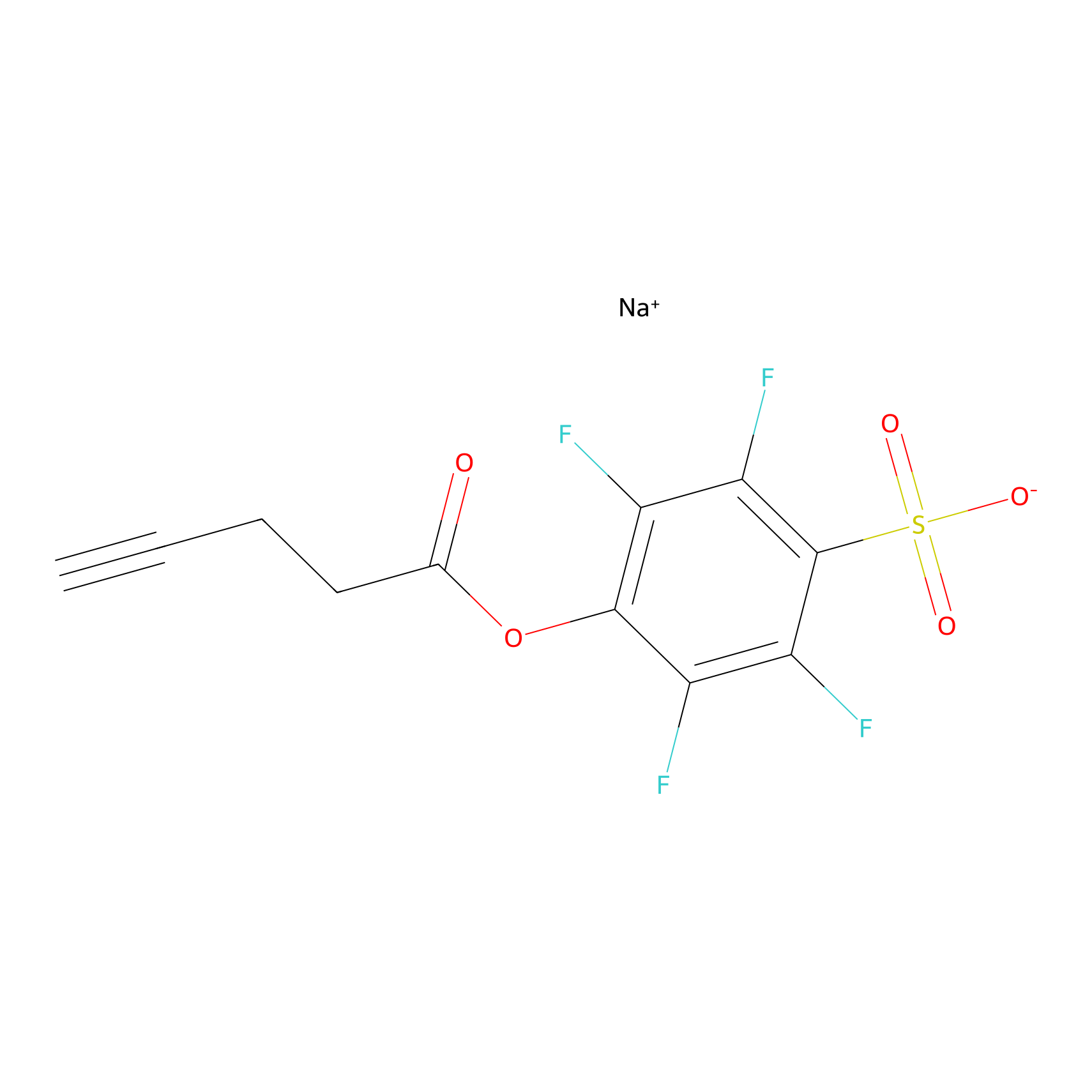 |
K202(16.23) | LDD2217 | [1] | |
|
DBIA Probe Info |
 |
C357(0.75) | LDD3310 | [2] | |
|
BTD Probe Info |
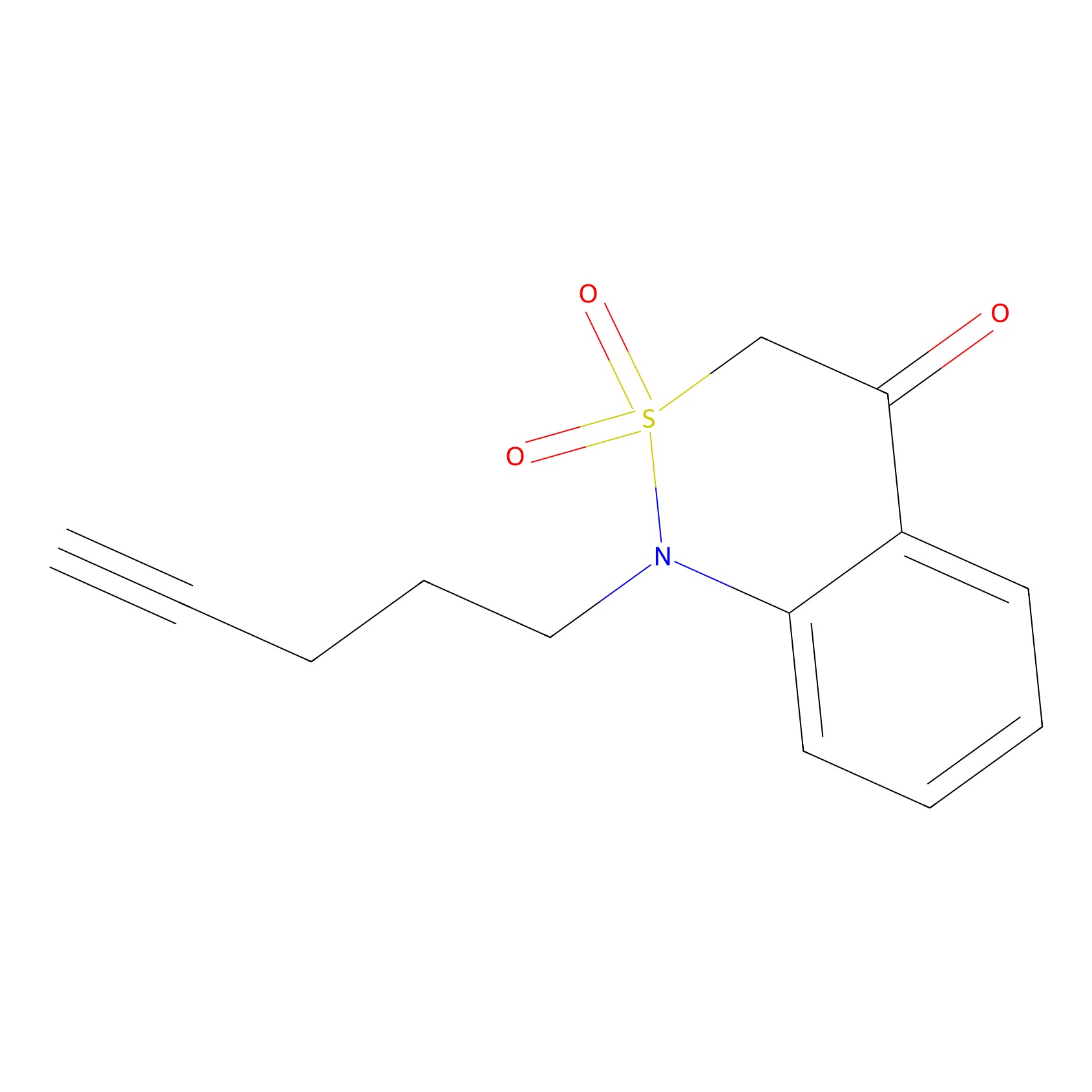 |
C339(0.78) | LDD2093 | [3] | |
|
HHS-475 Probe Info |
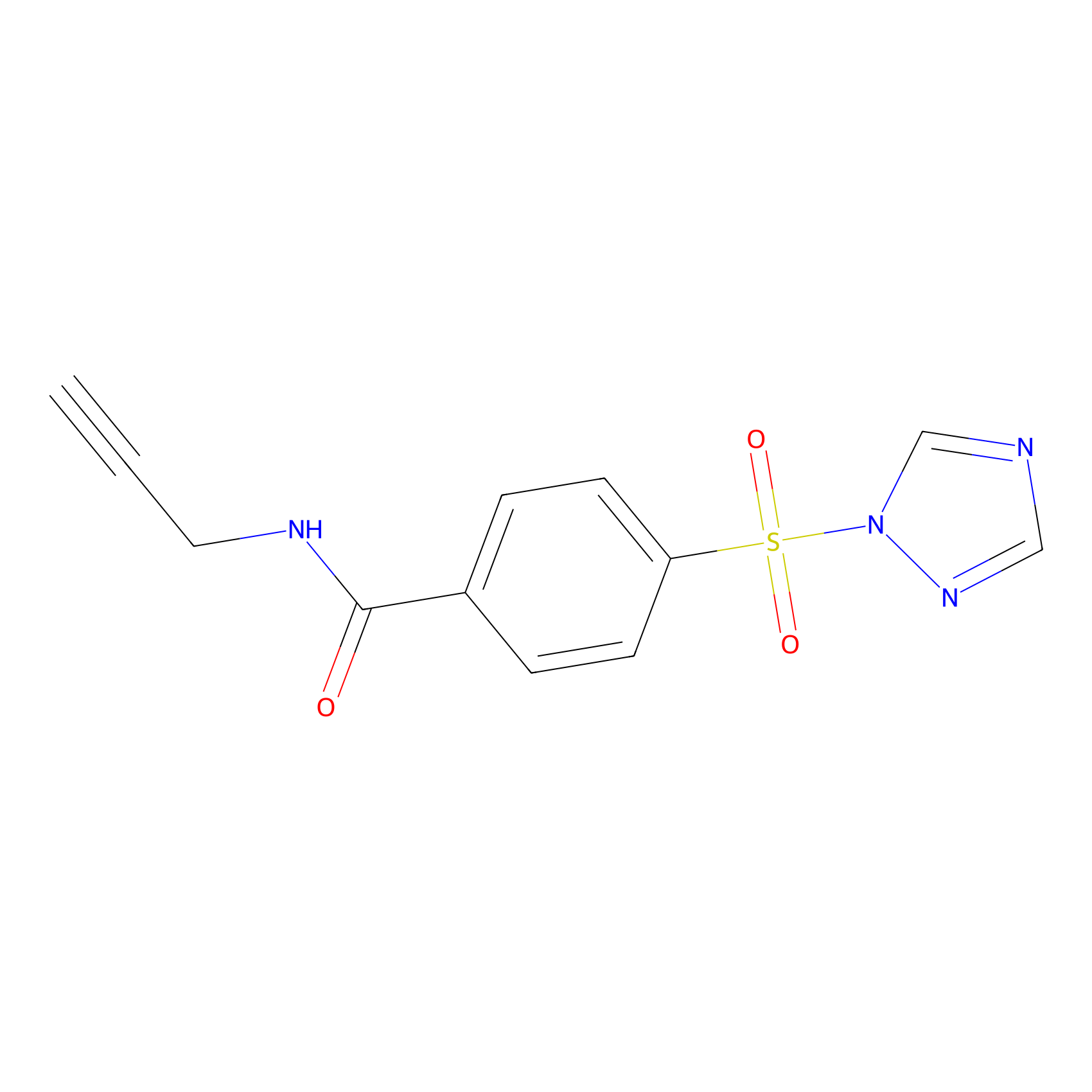 |
Y44(0.93) | LDD0264 | [4] | |
|
HHS-465 Probe Info |
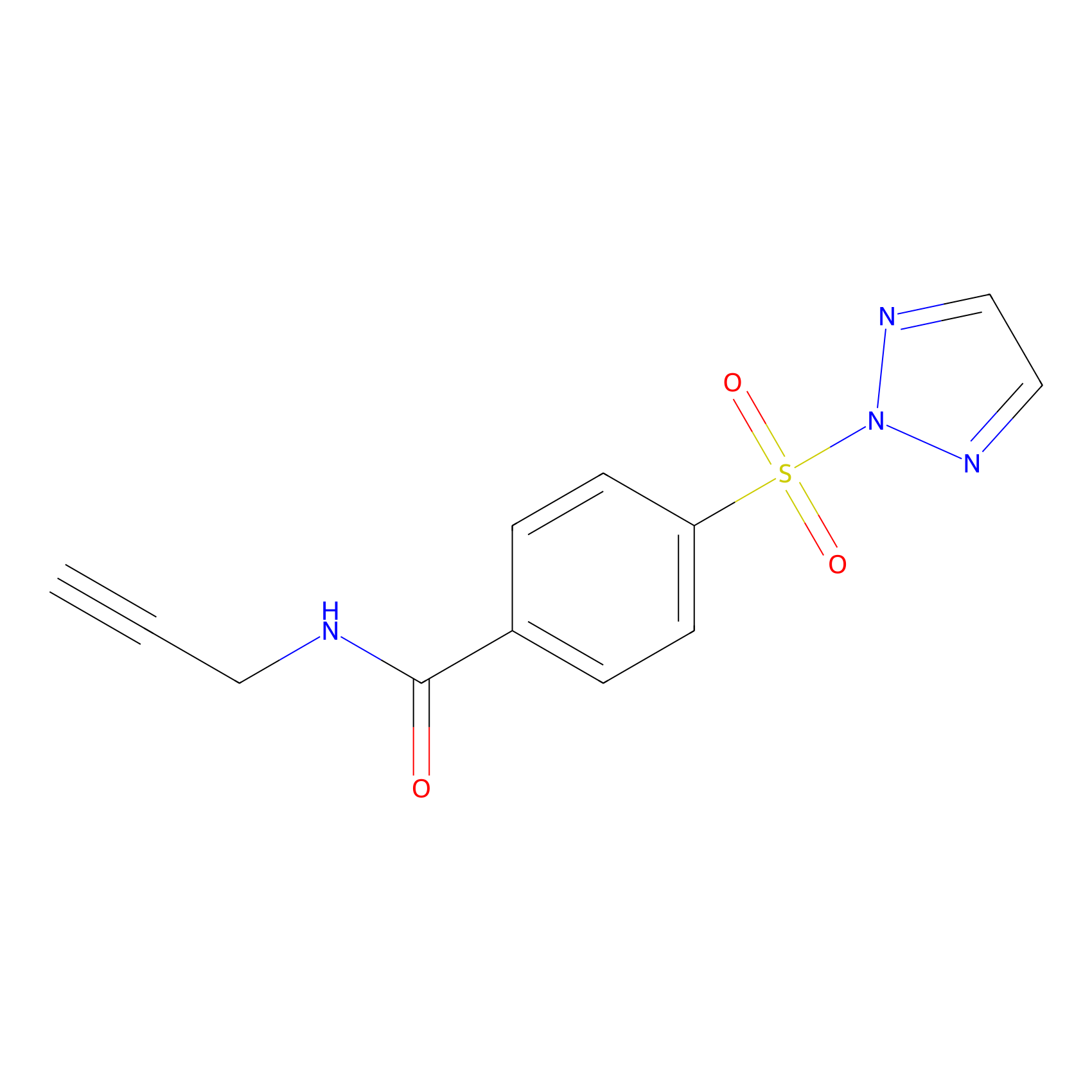 |
Y131(8.62); Y257(10.00); Y44(7.91) | LDD2237 | [5] | |
|
Acrolein Probe Info |
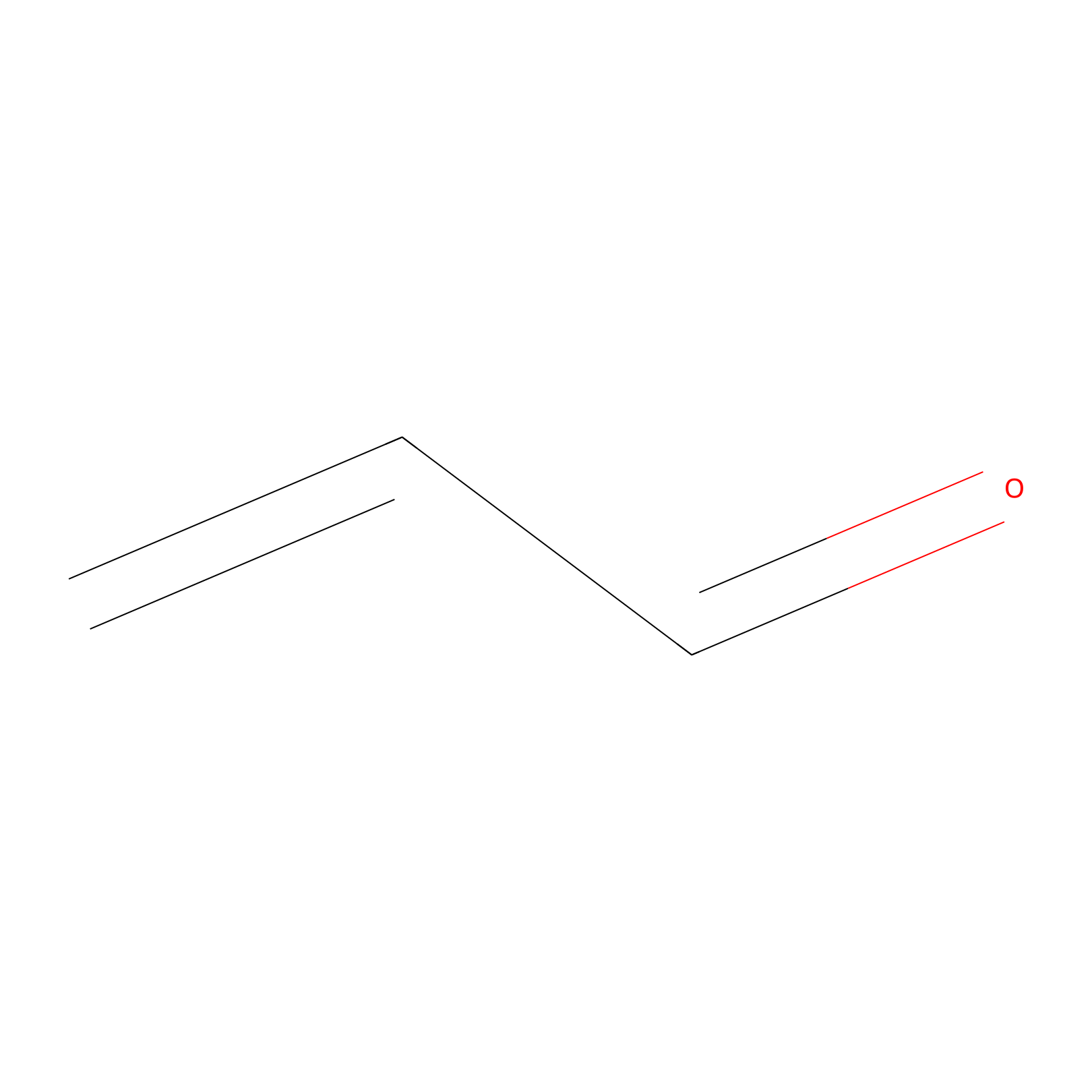 |
H25(0.00); C339(0.00) | LDD0221 | [6] | |
|
IPIAA_L Probe Info |
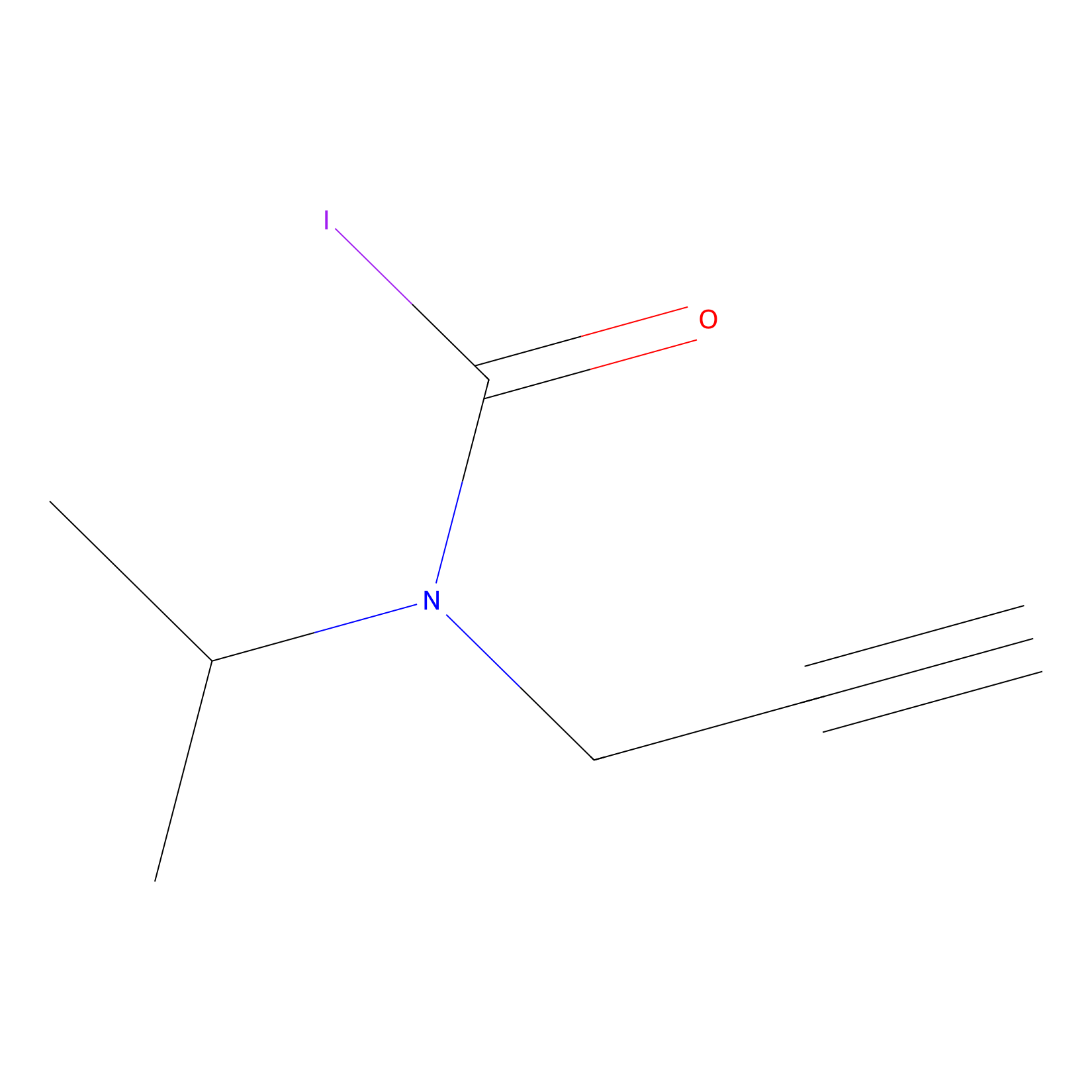 |
N.A. | LDD0031 | [7] | |
|
WYneN Probe Info |
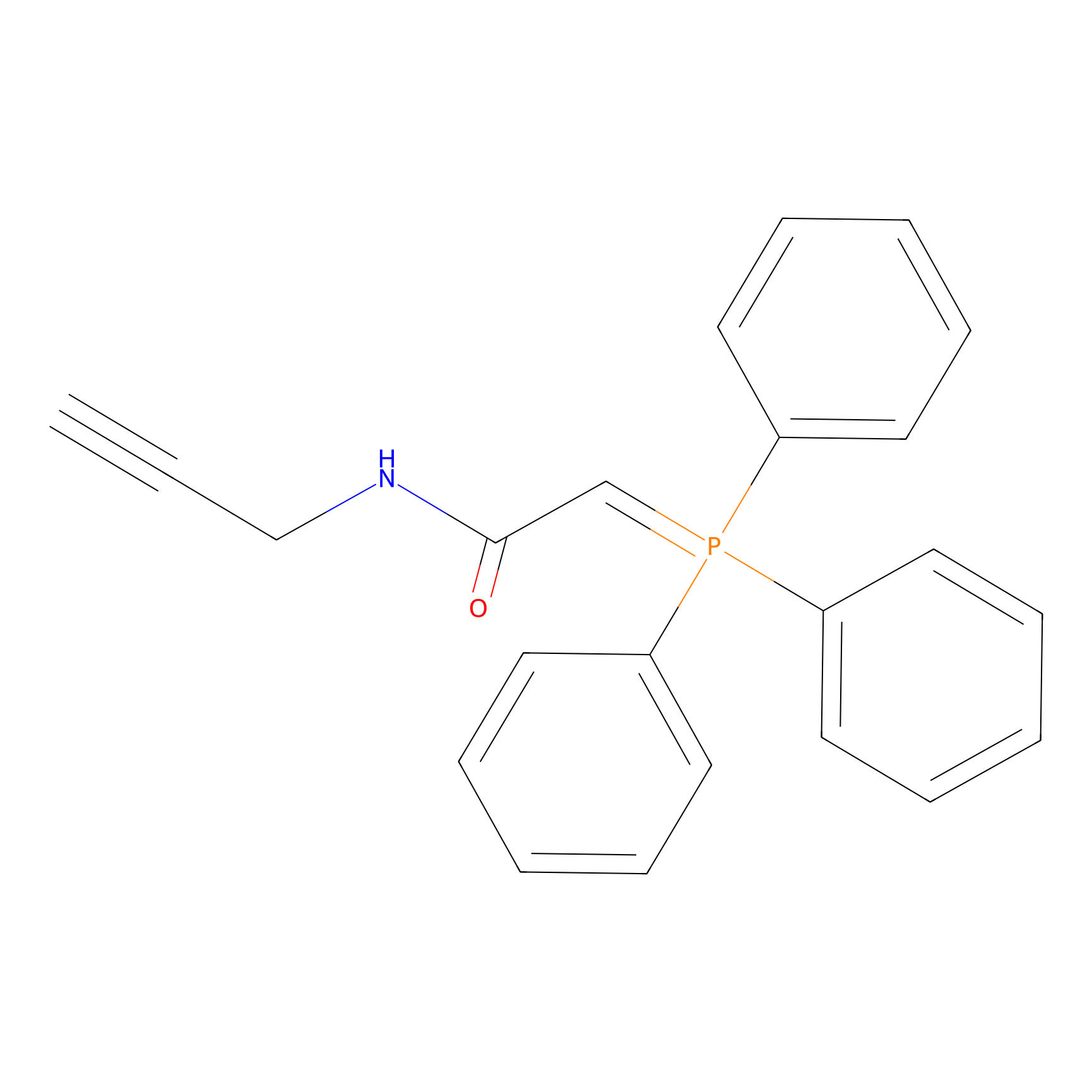 |
N.A. | LDD0021 | [8] | |
|
ENE Probe Info |
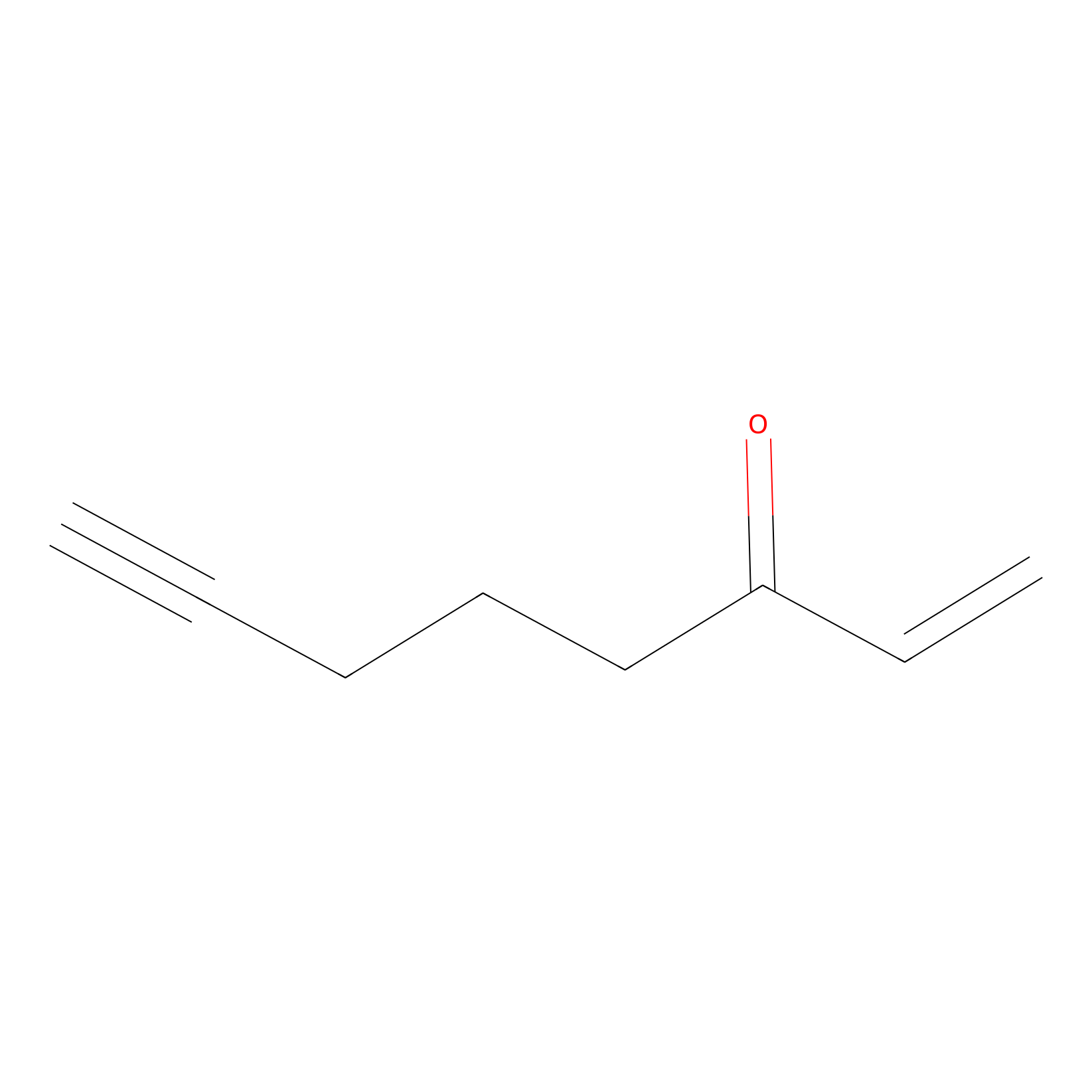 |
C357(0.00); C399(0.00) | LDD0006 | [8] | |
|
IA-alkyne Probe Info |
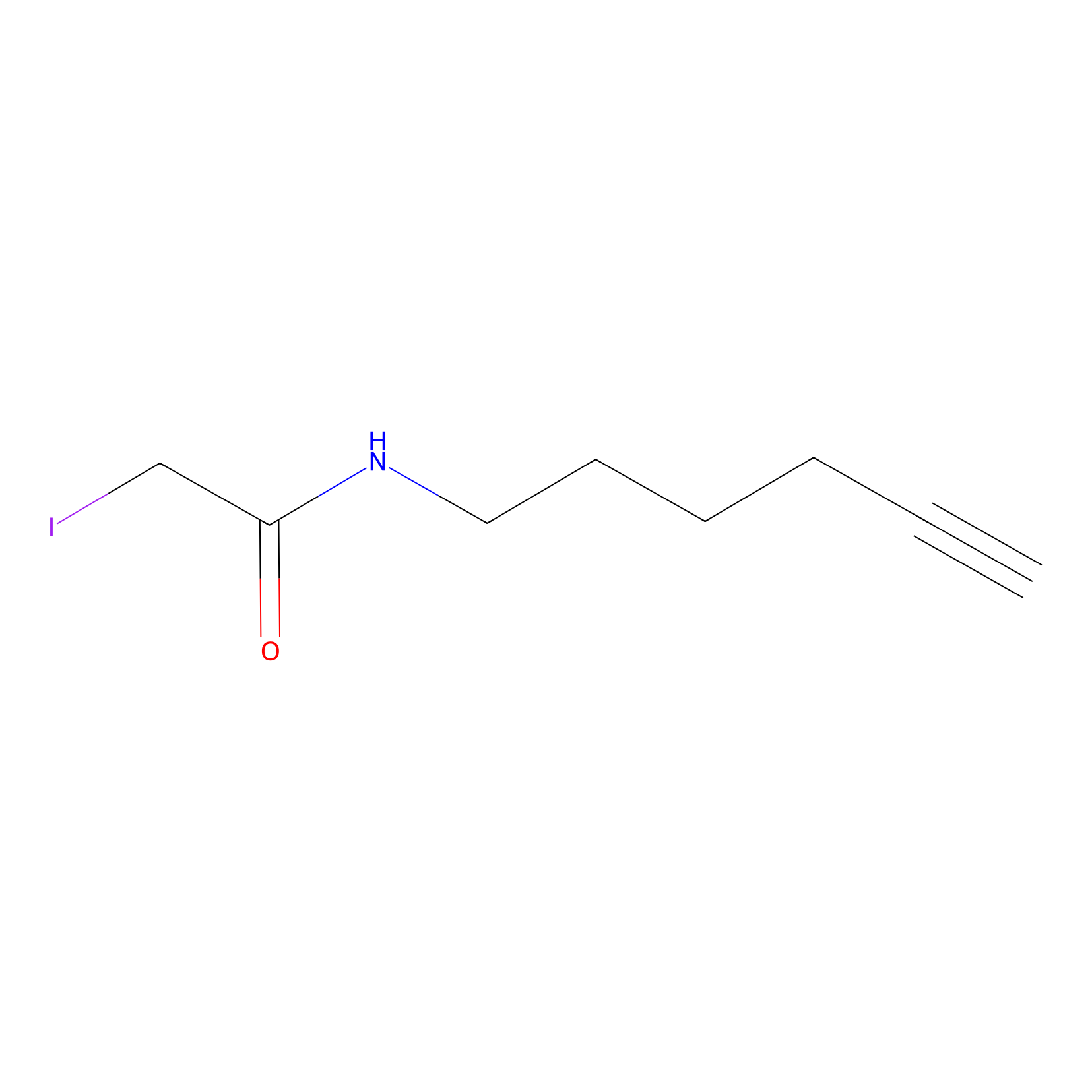 |
N.A. | LDD0149 | [9] | |
|
Ox-W18 Probe Info |
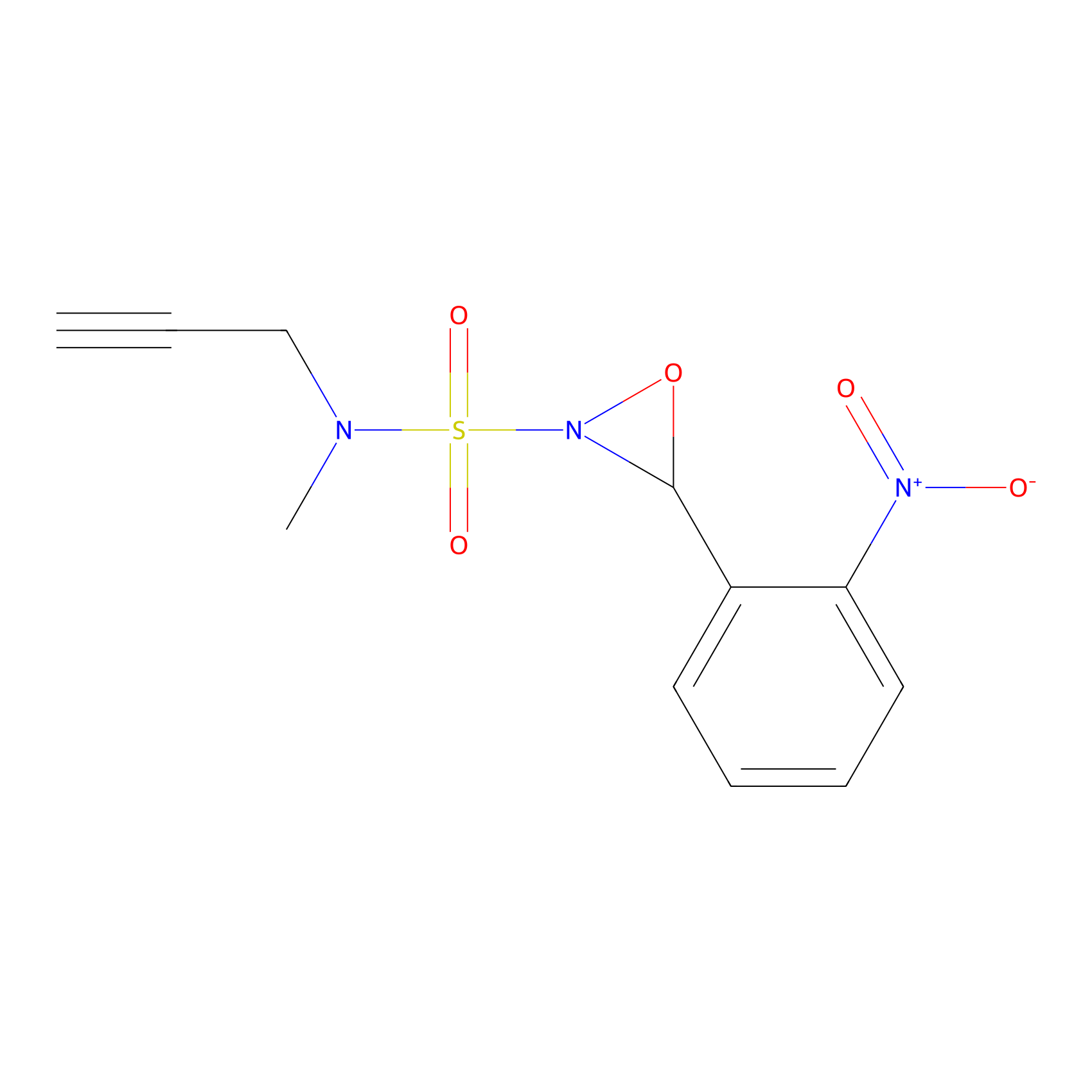 |
N.A. | LDD2175 | [10] | |
PAL-AfBPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
STS-2 Probe Info |
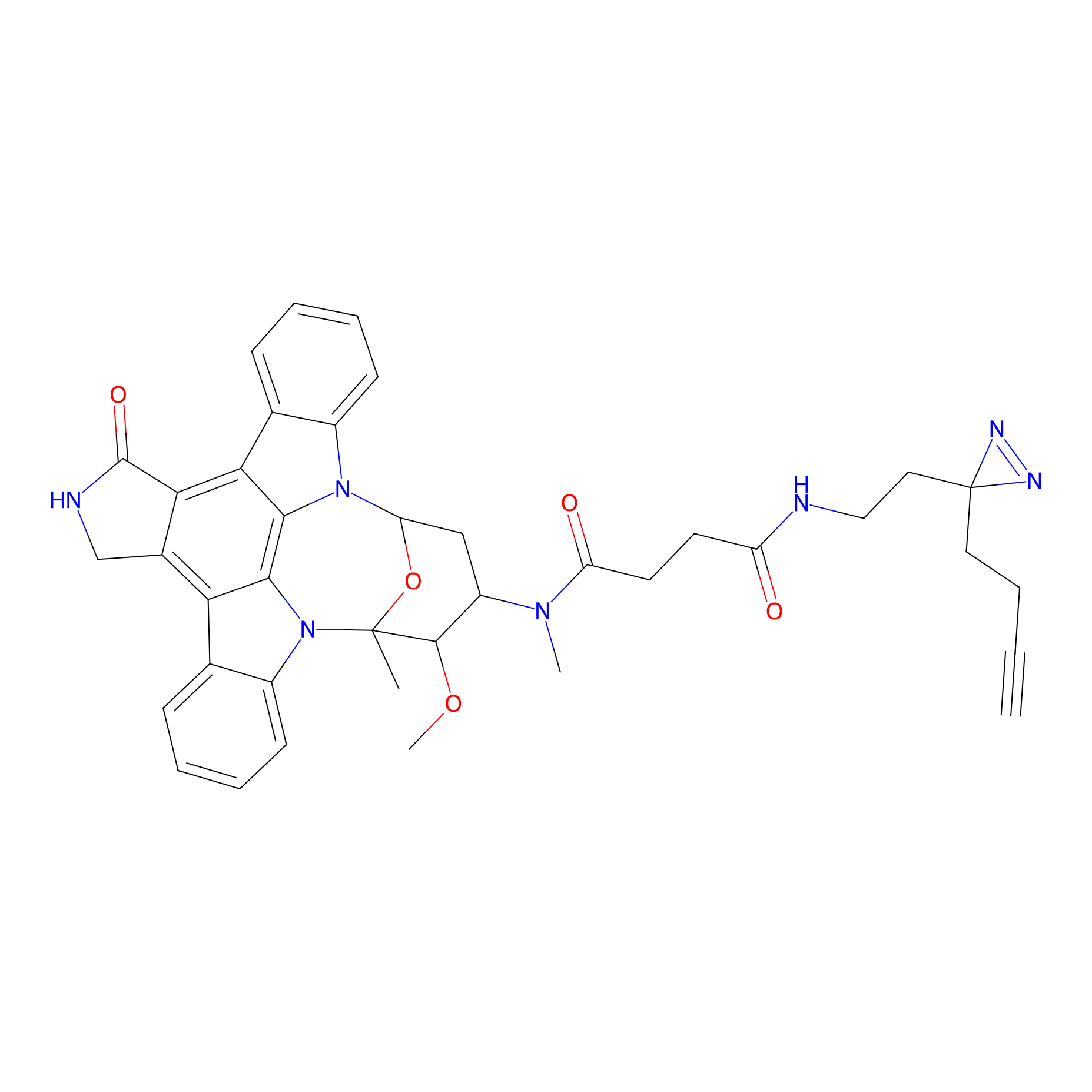 |
1.00 | LDD0139 | [11] | |
|
STS-1 Probe Info |
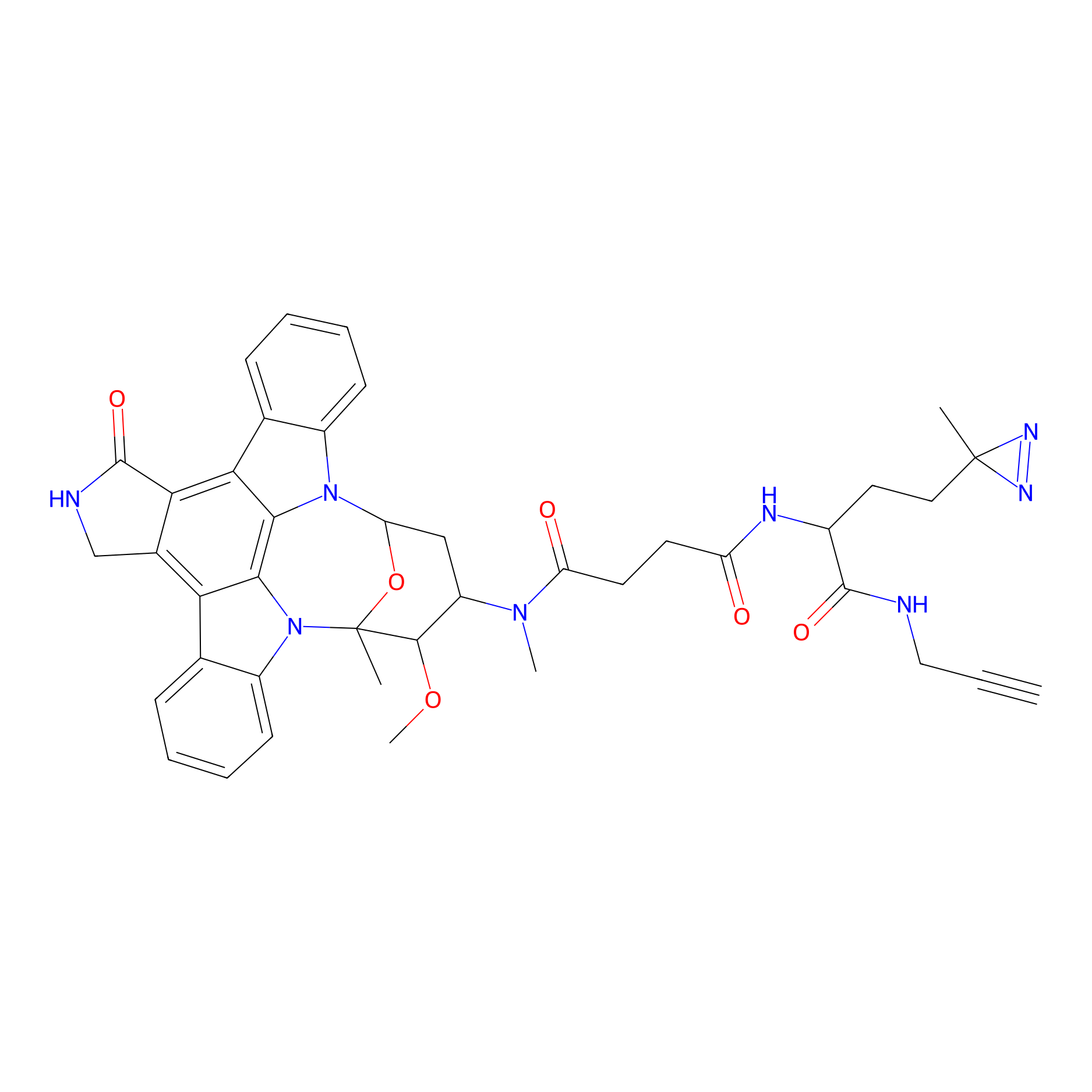 |
N.A. | LDD0069 | [12] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0558 | 2-Cyano-N-phenylacetamide | MDA-MB-231 | C339(1.20) | LDD2152 | [3] |
| LDCM0025 | 4SU-RNA | HEK-293T | C399(2.47) | LDD0371 | [13] |
| LDCM0020 | ARS-1620 | HCC44 | C357(1.22) | LDD2171 | [14] |
| LDCM0116 | HHS-0101 | DM93 | Y44(0.93) | LDD0264 | [4] |
| LDCM0117 | HHS-0201 | DM93 | Y44(0.86) | LDD0265 | [4] |
| LDCM0118 | HHS-0301 | DM93 | Y44(0.79) | LDD0266 | [4] |
| LDCM0119 | HHS-0401 | DM93 | Y44(0.84) | LDD0267 | [4] |
| LDCM0120 | HHS-0701 | DM93 | Y44(0.93) | LDD0268 | [4] |
| LDCM0107 | IAA | HeLa | H25(0.00); C339(0.00) | LDD0221 | [6] |
| LDCM0022 | KB02 | 22RV1 | C357(1.33) | LDD2243 | [2] |
| LDCM0023 | KB03 | 22RV1 | C357(1.70) | LDD2660 | [2] |
| LDCM0024 | KB05 | COLO792 | C357(0.75) | LDD3310 | [2] |
| LDCM0109 | NEM | HeLa | N.A. | LDD0224 | [6] |
| LDCM0500 | Nucleophilic fragment 13a | MDA-MB-231 | C339(0.78) | LDD2093 | [3] |
| LDCM0506 | Nucleophilic fragment 16a | MDA-MB-231 | C339(2.24) | LDD2099 | [3] |
| LDCM0508 | Nucleophilic fragment 17a | MDA-MB-231 | C339(2.23) | LDD2101 | [3] |
| LDCM0516 | Nucleophilic fragment 21a | MDA-MB-231 | C339(0.99) | LDD2109 | [3] |
| LDCM0518 | Nucleophilic fragment 22a | MDA-MB-231 | C339(0.92) | LDD2111 | [3] |
| LDCM0522 | Nucleophilic fragment 24a | MDA-MB-231 | C339(0.60) | LDD2115 | [3] |
| LDCM0532 | Nucleophilic fragment 29a | MDA-MB-231 | C339(1.44) | LDD2125 | [3] |
| LDCM0534 | Nucleophilic fragment 30a | MDA-MB-231 | C339(0.72) | LDD2127 | [3] |
| LDCM0536 | Nucleophilic fragment 31 | MDA-MB-231 | C339(0.77) | LDD2129 | [3] |
| LDCM0540 | Nucleophilic fragment 35 | MDA-MB-231 | C339(1.27) | LDD2133 | [3] |
| LDCM0542 | Nucleophilic fragment 37 | MDA-MB-231 | C339(0.62) | LDD2135 | [3] |
| LDCM0546 | Nucleophilic fragment 40 | MDA-MB-231 | C337(1.11) | LDD2140 | [3] |
| LDCM0549 | Nucleophilic fragment 43 | MDA-MB-231 | C337(0.91) | LDD2143 | [3] |
| LDCM0550 | Nucleophilic fragment 5a | MDA-MB-231 | C339(1.37) | LDD2144 | [3] |
| LDCM0021 | THZ1 | HCT 116 | C357(1.22) | LDD2173 | [14] |
The Interaction Atlas With This Target
References
