Details of the Target
General Information of Target
| Target ID | LDTP02553 | |||||
|---|---|---|---|---|---|---|
| Target Name | Glycogen phosphorylase, muscle form (PYGM) | |||||
| Gene Name | PYGM | |||||
| Gene ID | 5837 | |||||
| Synonyms |
Glycogen phosphorylase, muscle form; EC 2.4.1.1; Myophosphorylase |
|||||
| 3D Structure | ||||||
| Sequence |
MSRPLSDQEKRKQISVRGLAGVENVTELKKNFNRHLHFTLVKDRNVATPRDYYFALAHTV
RDHLVGRWIRTQQHYYEKDPKRIYYLSLEFYMGRTLQNTMVNLALENACDEATYQLGLDM EELEEIEEDAGLGNGGLGRLAACFLDSMATLGLAAYGYGIRYEFGIFNQKISGGWQMEEA DDWLRYGNPWEKARPEFTLPVHFYGHVEHTSQGAKWVDTQVVLAMPYDTPVPGYRNNVVN TMRLWSAKAPNDFNLKDFNVGGYIQAVLDRNLAENISRVLYPNDNFFEGKELRLKQEYFV VAATLQDIIRRFKSSKFGCRDPVRTNFDAFPDKVAIQLNDTHPSLAIPELMRILVDLERM DWDKAWDVTVRTCAYTNHTVLPEALERWPVHLLETLLPRHLQIIYEINQRFLNRVAAAFP GDVDRLRRMSLVEEGAVKRINMAHLCIAGSHAVNGVARIHSEILKKTIFKDFYELEPHKF QNKTNGITPRRWLVLCNPGLAEVIAERIGEDFISDLDQLRKLLSFVDDEAFIRDVAKVKQ ENKLKFAAYLEREYKVHINPNSLFDIQVKRIHEYKRQLLNCLHVITLYNRIKREPNKFFV PRTVMIGGKAAPGYHMAKMIIRLVTAIGDVVNHDPAVGDRLRVIFLENYRVSLAEKVIPA ADLSEQISTAGTEASGTGNMKFMLNGALTIGTMDGANVEMAEEAGEENFFIFGMRVEDVD KLDQRGYNAQEYYDRIPELRQVIEQLSSGFFSPKQPDLFKDIVNMLMHHDRFKVFADYED YIKCQEKVSALYKNPREWTRMVIRNIATSGKFSSDRTIAQYAREIWGVEPSRQRLPAPDE AI |
|||||
| Target Type |
Patented-recorded
|
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Glycogen phosphorylase family
|
|||||
| Function |
Allosteric enzyme that catalyzes the rate-limiting step in glycogen catabolism, the phosphorolytic cleavage of glycogen to produce glucose-1-phosphate, and plays a central role in maintaining cellular and organismal glucose homeostasis.
|
|||||
| TTD ID | ||||||
| Uniprot ID | ||||||
| DrugMap ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
| ChEMBL ID | ||||||
Target Site Mutations in Different Cell Lines
| Cell line | Mutation details | Probe for labeling this protein in this cell | |||
|---|---|---|---|---|---|
| EFO27 | SNV: p.H615N | DBIA Probe Info | |||
| HCT116 | SNV: p.Y375H | . | |||
| HT | SNV: p.Y156H | . | |||
| KMCH1 | SNV: p.K364Ter | . | |||
| LS123 | SNV: p.E77D; p.G318A | DBIA Probe Info | |||
| LS180 | SNV: p.R520H | DBIA Probe Info | |||
| PF382 | SNV: p.S789G | . | |||
| RL952 | SNV: p.R70H | . | |||
| SW1783 | SNV: p.V323M | DBIA Probe Info | |||
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
YN-4 Probe Info |
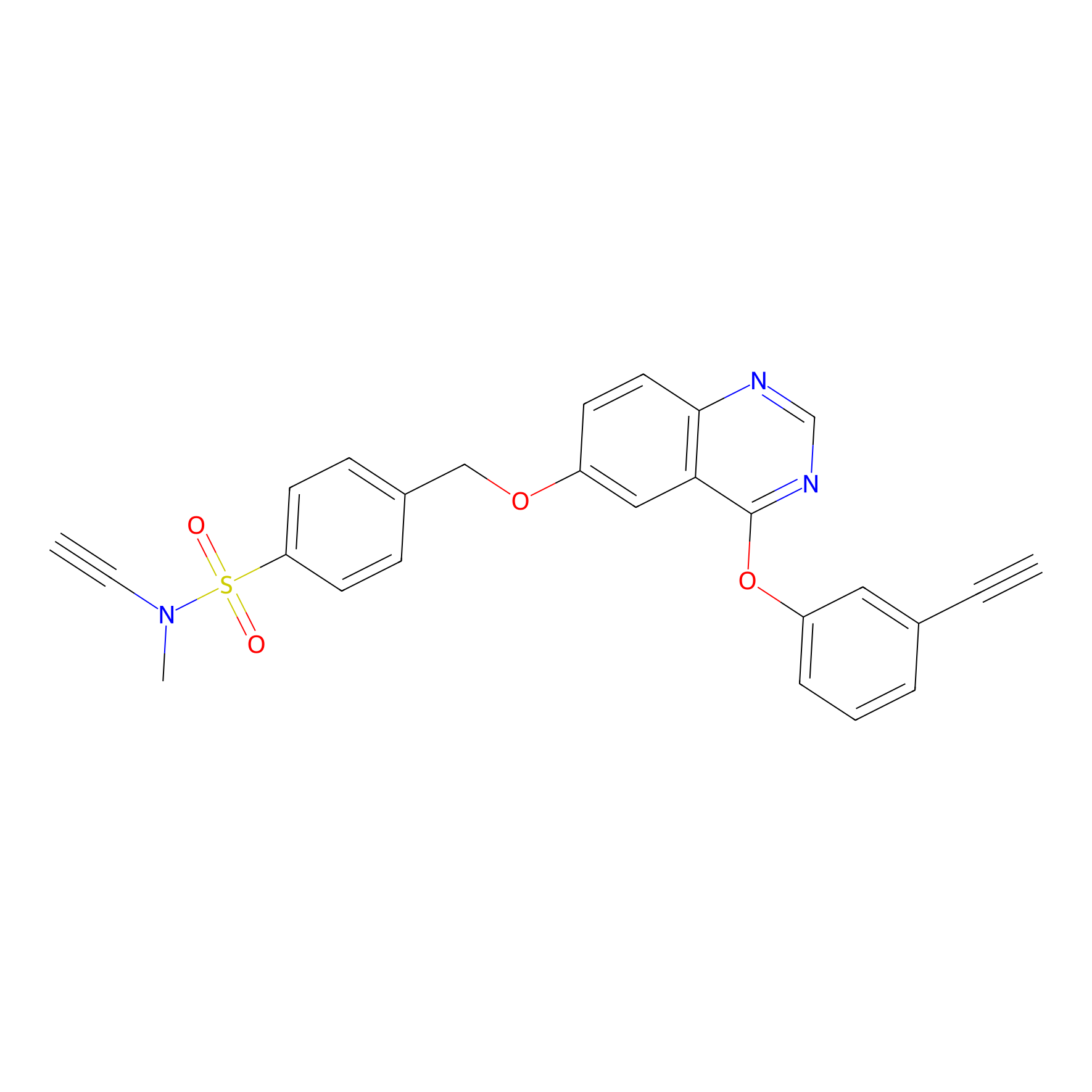 |
100.00 | LDD0445 | [1] | |
|
DBIA Probe Info |
 |
C373(1.55) | LDD3310 | [2] | |
|
AHL-Pu-1 Probe Info |
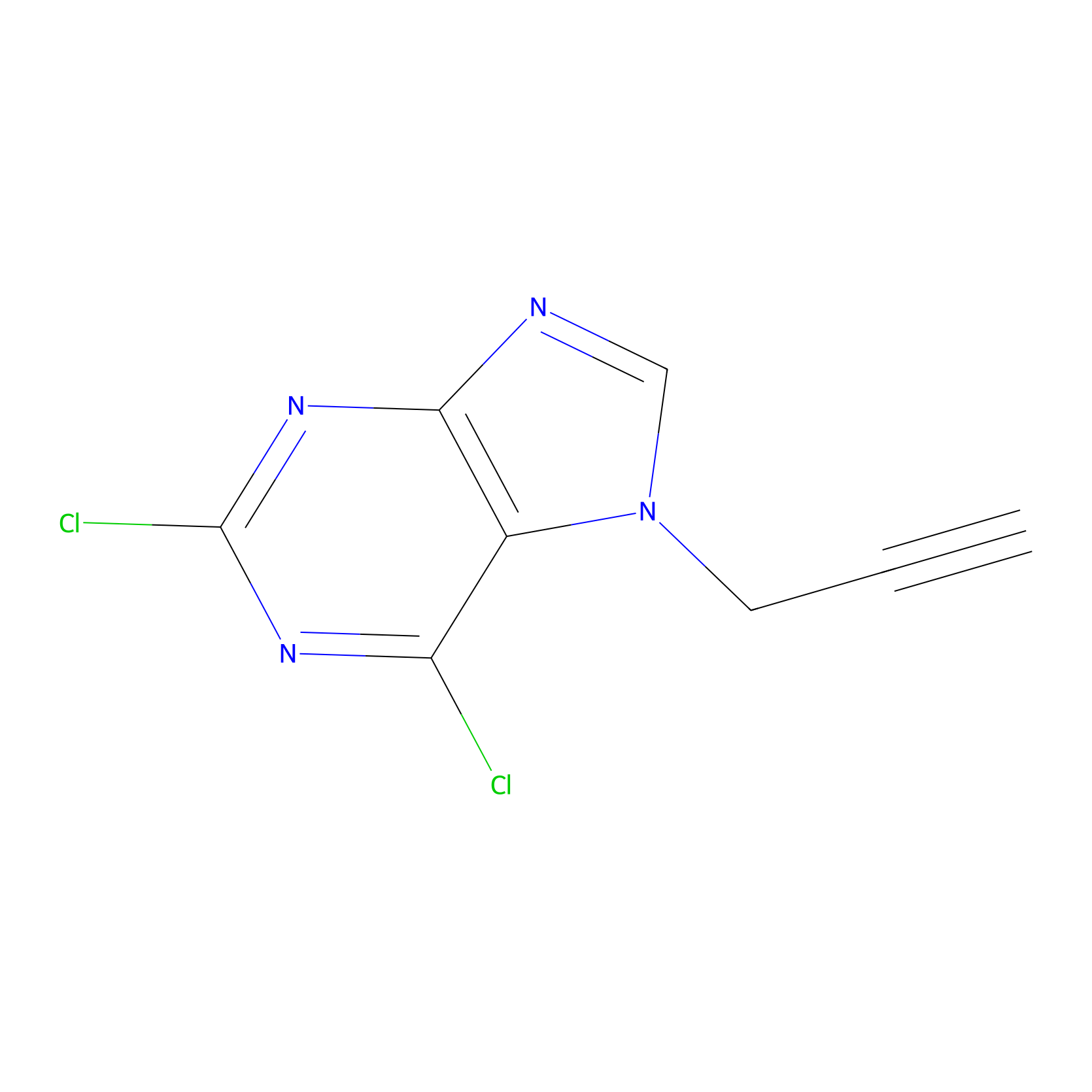 |
C319(2.74); C143(3.49) | LDD0170 | [3] | |
|
HHS-475 Probe Info |
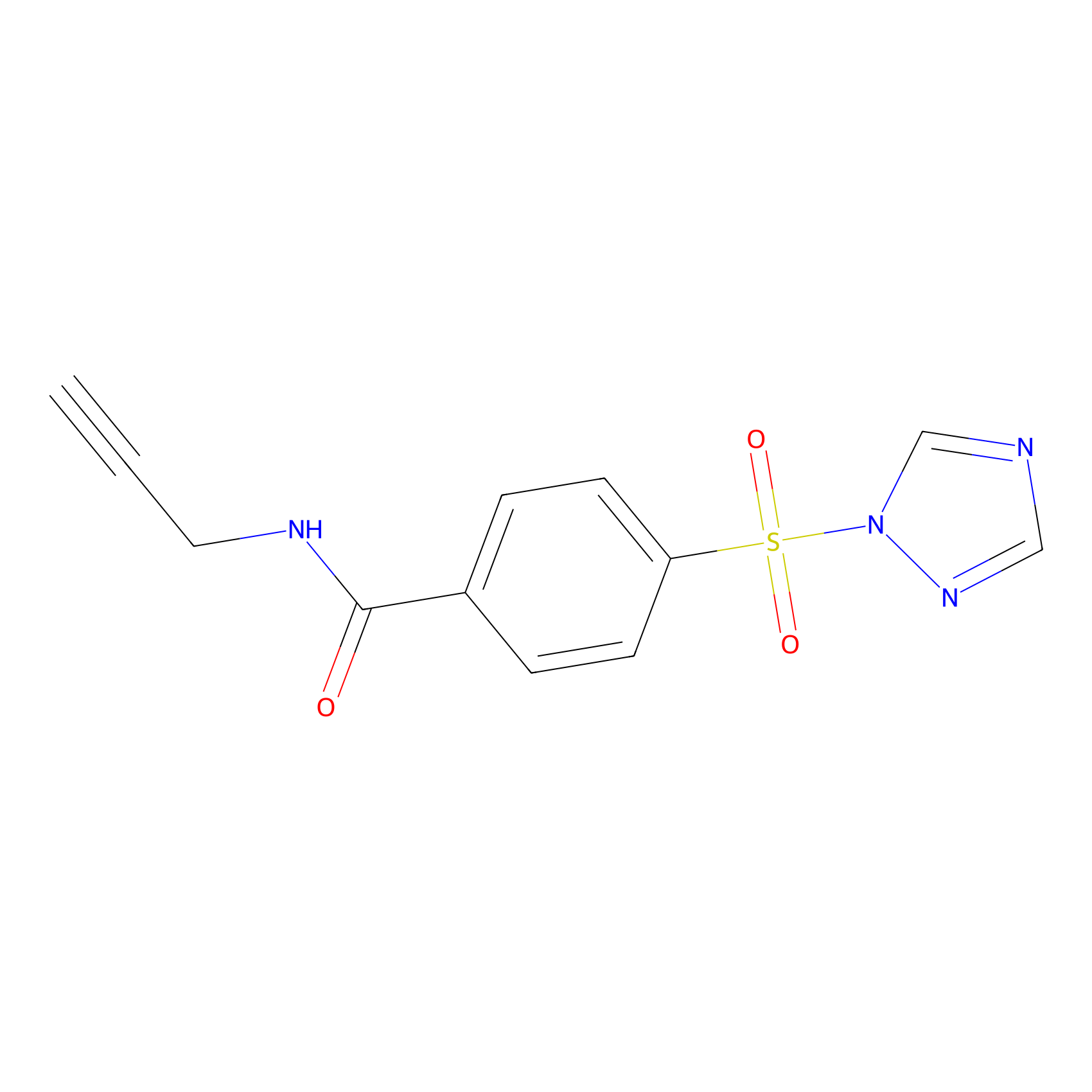 |
Y614(0.83); Y298(0.99); Y186(1.51) | LDD0264 | [4] | |
|
HHS-465 Probe Info |
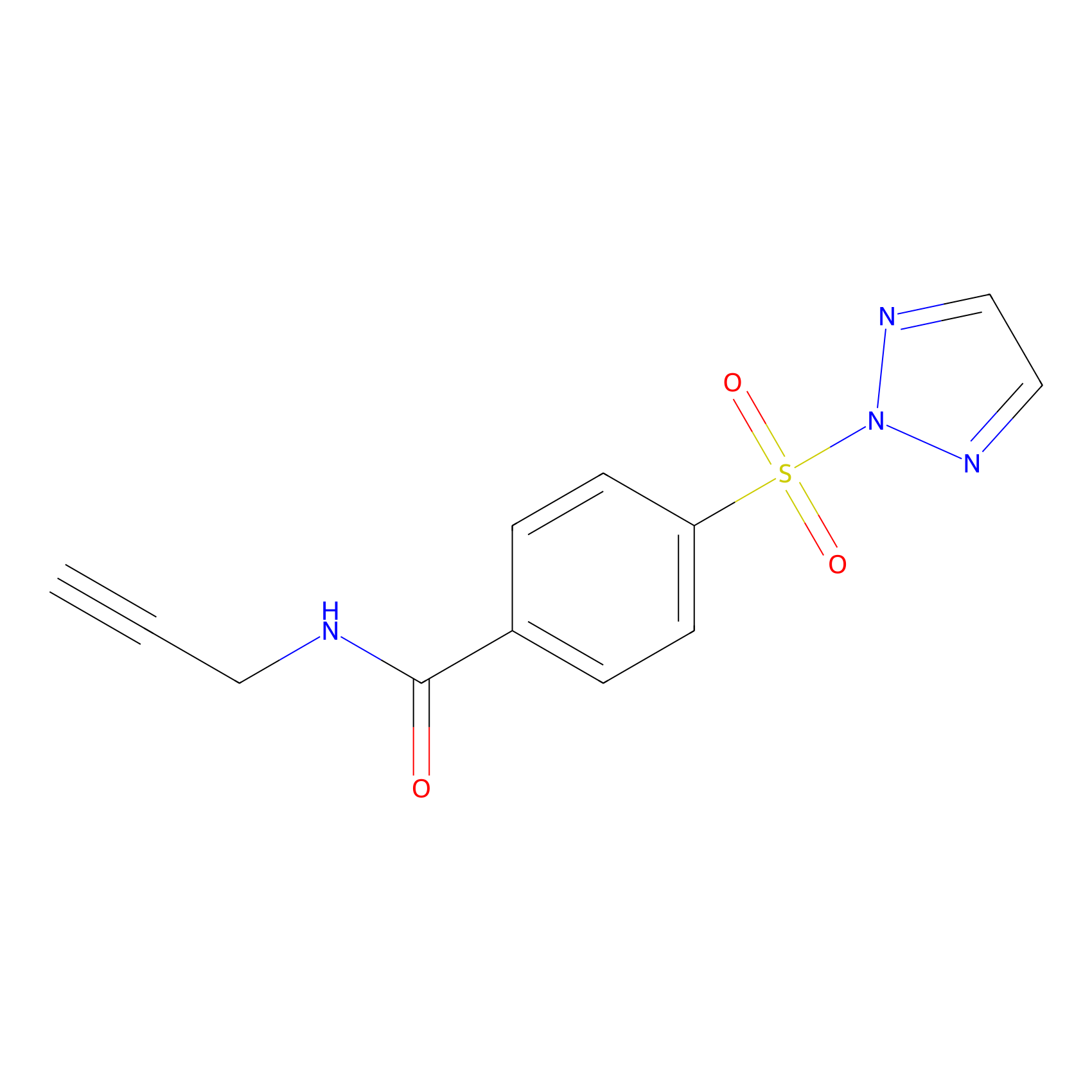 |
Y186(10.00); Y281(10.00); Y574(10.00); Y614(9.33) | LDD2237 | [5] | |
|
NAIA_4 Probe Info |
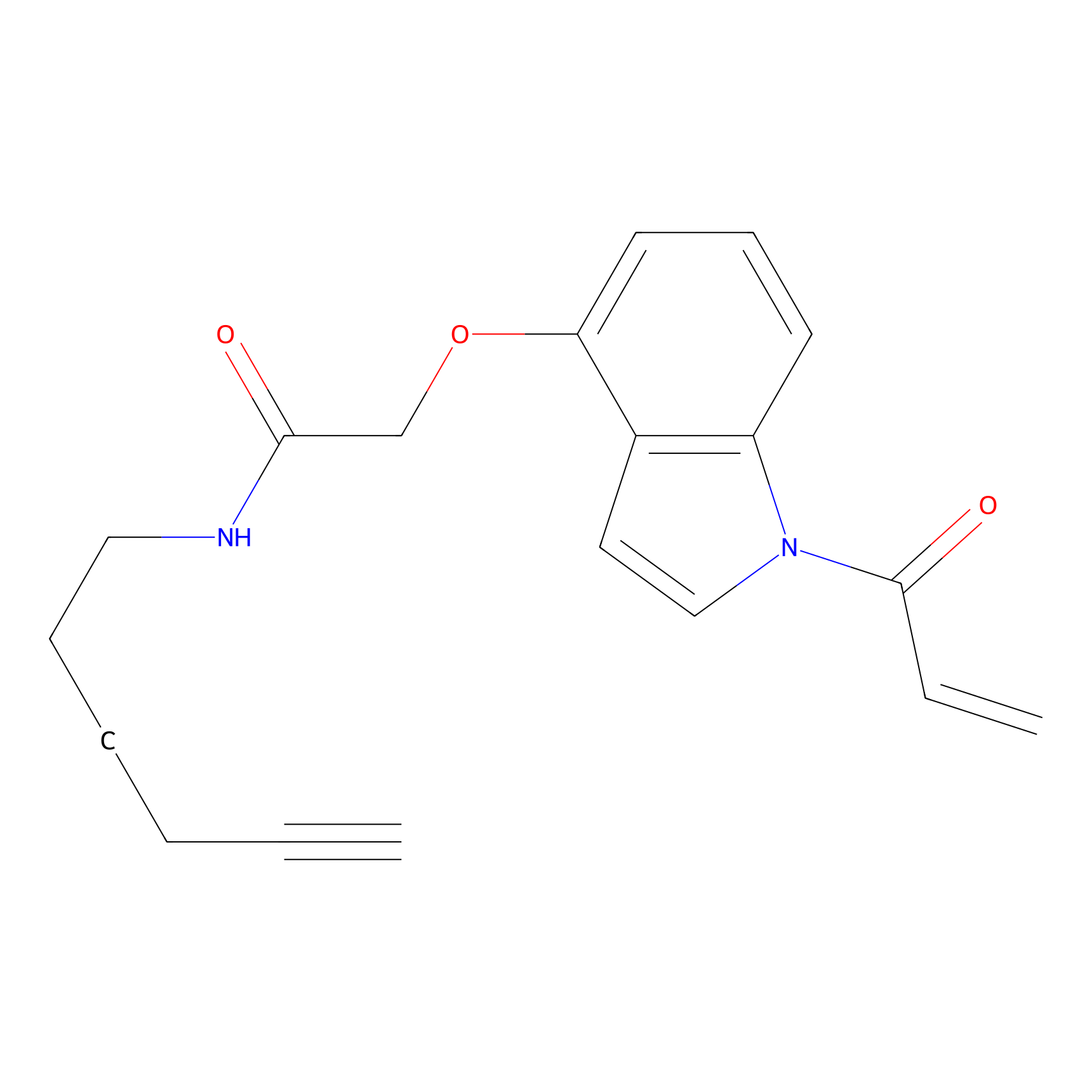 |
C109(0.00); C373(0.00) | LDD2226 | [6] | |
|
YN-1 Probe Info |
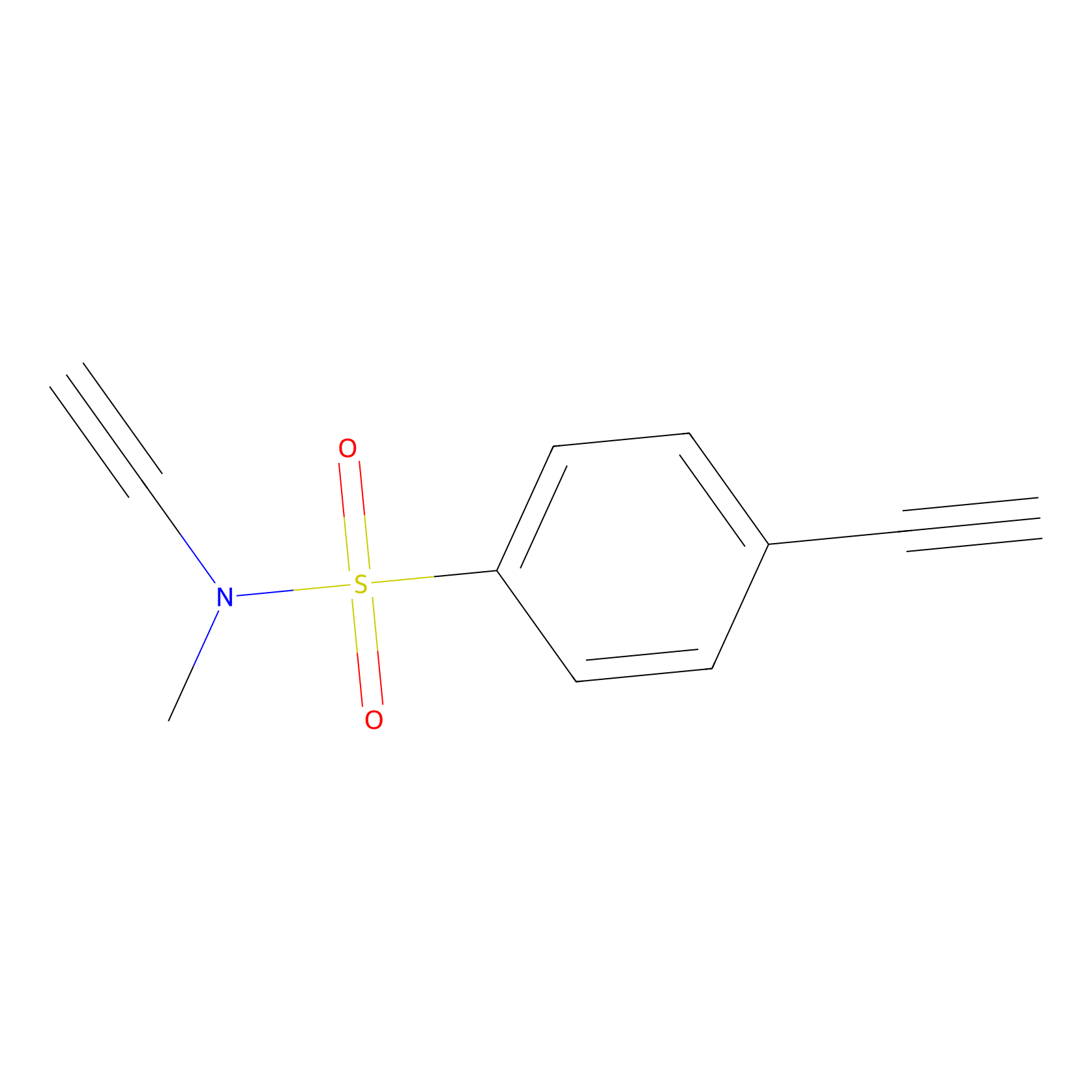 |
N.A. | LDD0447 | [1] | |
PAL-AfBPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
STS-1 Probe Info |
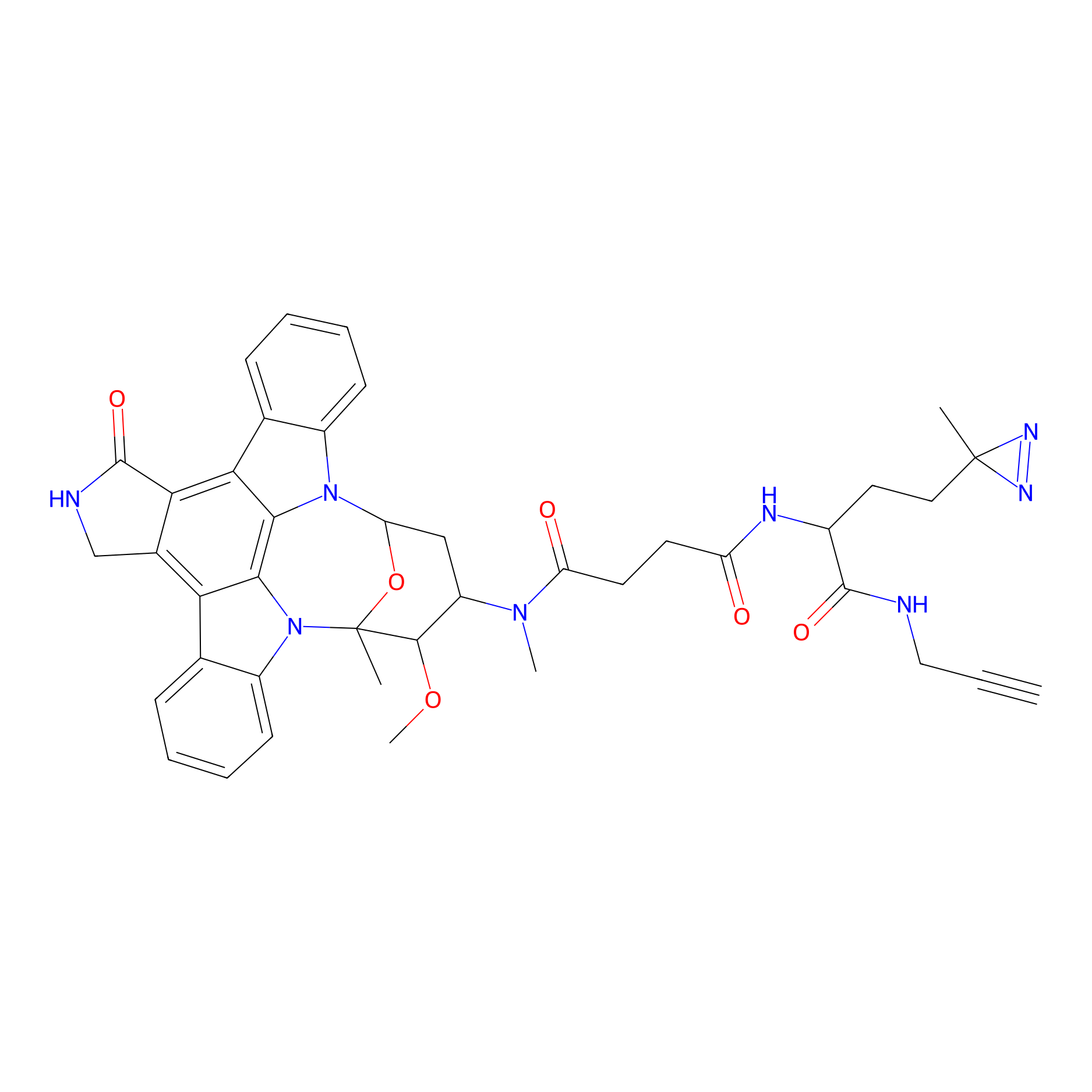 |
N.A. | LDD0136 | [7] | |
|
STS-2 Probe Info |
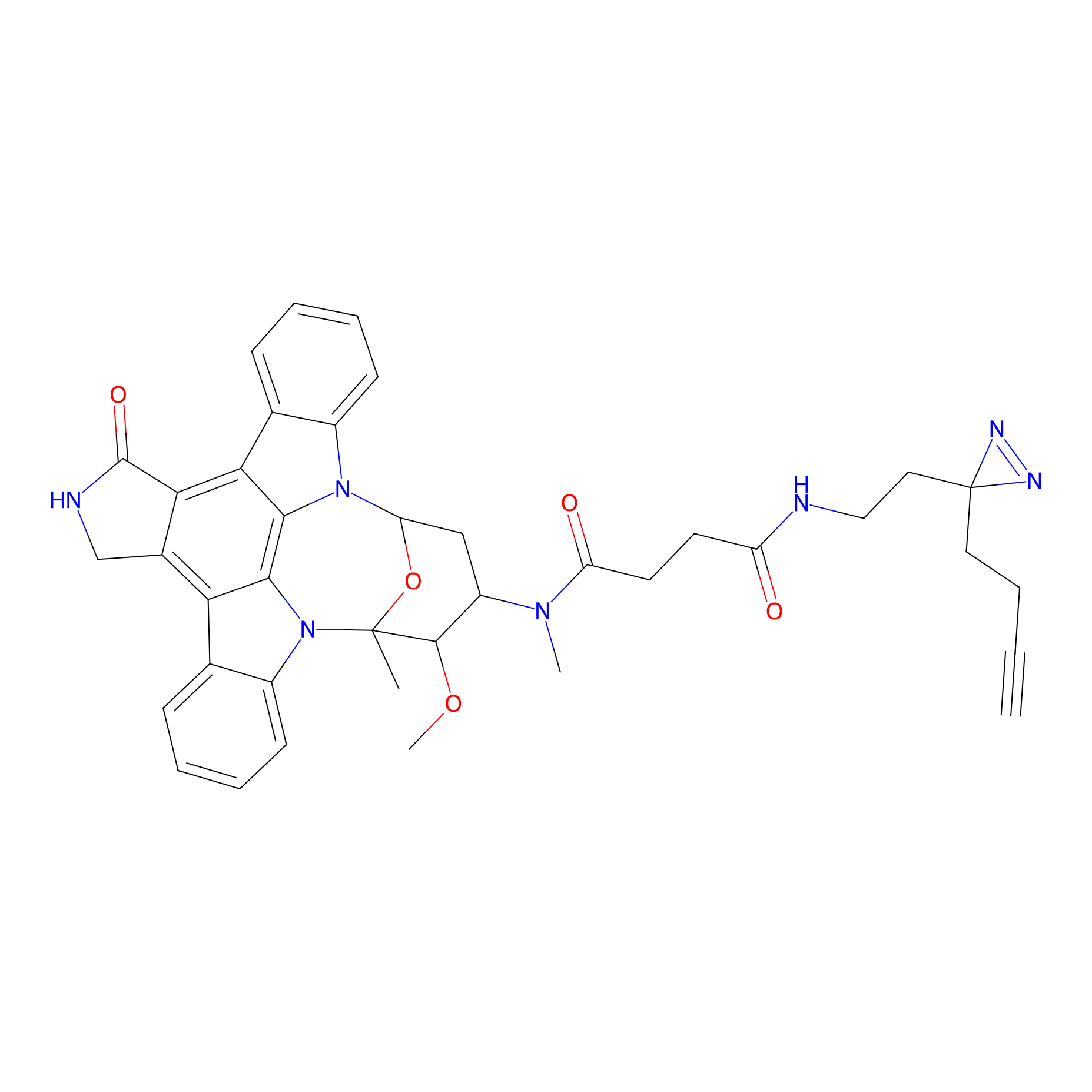 |
N.A. | LDD0139 | [7] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0025 | 4SU-RNA | DM93 | C319(2.74); C143(3.49) | LDD0170 | [3] |
| LDCM0026 | 4SU-RNA+native RNA | DM93 | C319(4.24) | LDD0171 | [3] |
| LDCM0116 | HHS-0101 | DM93 | Y614(0.83); Y298(0.99); Y186(1.51) | LDD0264 | [4] |
| LDCM0117 | HHS-0201 | DM93 | Y186(0.80); Y614(0.80); Y298(0.88) | LDD0265 | [4] |
| LDCM0118 | HHS-0301 | DM93 | Y614(0.70); Y298(0.72); Y186(1.13) | LDD0266 | [4] |
| LDCM0119 | HHS-0401 | DM93 | Y614(0.78); Y298(0.82); Y186(0.87) | LDD0267 | [4] |
| LDCM0120 | HHS-0701 | DM93 | Y614(0.46); Y298(0.55); Y186(0.68) | LDD0268 | [4] |
| LDCM0022 | KB02 | 42-MG-BA | C373(1.58) | LDD2244 | [2] |
| LDCM0023 | KB03 | 769-P | C373(1.54) | LDD2663 | [2] |
| LDCM0024 | KB05 | COLO792 | C373(1.55) | LDD3310 | [2] |
The Interaction Atlas With This Target
The Protein(s) Related To This Target
Enzyme
The Drug(s) Related To This Target
Approved
| Drug Name | Drug Type | External ID | |||
|---|---|---|---|---|---|
| Pyridoxal Phosphate | Small molecular drug | DB00114 | |||
Phase 2
| Drug Name | Drug Type | External ID | |||
|---|---|---|---|---|---|
| Ursolic Acid | Small molecular drug | D07LUJ | |||
Investigative
Discontinued
| Drug Name | Drug Type | External ID | |||
|---|---|---|---|---|---|
| Jtt-651 | . | D09HAL | |||
References
