Details of the Target
General Information of Target
| Target ID | LDTP02054 | |||||
|---|---|---|---|---|---|---|
| Target Name | Cytochrome P450 1A1 (CYP1A1) | |||||
| Gene Name | CYP1A1 | |||||
| Gene ID | 1543 | |||||
| Synonyms |
Cytochrome P450 1A1; CYPIA1; EC 1.14.14.1; Cytochrome P450 form 6; Cytochrome P450-C; Cytochrome P450-P1; Hydroperoxy icosatetraenoate dehydratase; EC 4.2.1.152 |
|||||
| 3D Structure | ||||||
| Sequence |
MLFPISMSATEFLLASVIFCLVFWVIRASRPQVPKGLKNPPGPWGWPLIGHMLTLGKNPH
LALSRMSQQYGDVLQIRIGSTPVVVLSGLDTIRQALVRQGDDFKGRPDLYTFTLISNGQS MSFSPDSGPVWAARRRLAQNGLKSFSIASDPASSTSCYLEEHVSKEAEVLISTLQELMAG PGHFNPYRYVVVSVTNVICAICFGRRYDHNHQELLSLVNLNNNFGEVVGSGNPADFIPIL RYLPNPSLNAFKDLNEKFYSFMQKMVKEHYKTFEKGHIRDITDSLIEHCQEKQLDENANV QLSDEKIINIVLDLFGAGFDTVTTAISWSLMYLVMNPRVQRKIQEELDTVIGRSRRPRLS DRSHLPYMEAFILETFRHSSFVPFTIPHSTTRDTSLKGFYIPKGRCVFVNQWQINHDQKL WVNPSEFLPERFLTPDGAIDKVLSEKVIIFGMGKRKCIGETIARWEVFLFLAILLQRVEF SVPLGVKVDMTPIYGLTMKHACCEHFQMQLRS |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Cytochrome P450 family
|
|||||
| Subcellular location |
Endoplasmic reticulum membrane
|
|||||
| Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids, steroid hormones and vitamins. Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase). Catalyzes the hydroxylation of carbon-hydrogen bonds. Exhibits high catalytic activity for the formation of hydroxyestrogens from estrone (E1) and 17beta-estradiol (E2), namely 2-hydroxy E1 and E2, as well as D-ring hydroxylated E1 and E2 at the C15-alpha and C16-alpha positions. Displays different regioselectivities for polyunsaturated fatty acids (PUFA) hydroxylation. Catalyzes the epoxidation of double bonds of certain PUFA. Converts arachidonic acid toward epoxyeicosatrienoic acid (EET) regioisomers, 8,9-, 11,12-, and 14,15-EET, that function as lipid mediators in the vascular system. Displays an absolute stereoselectivity in the epoxidation of eicosapentaenoic acid (EPA) producing the 17(R),18(S) enantiomer. May play an important role in all-trans retinoic acid biosynthesis in extrahepatic tissues. Catalyzes two successive oxidative transformation of all-trans retinol to all-trans retinal and then to the active form all-trans retinoic acid. May also participate in eicosanoids metabolism by converting hydroperoxide species into oxo metabolites (lipoxygenase-like reaction, NADPH-independent).
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
| ChEMBL ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
YN-1 Probe Info |
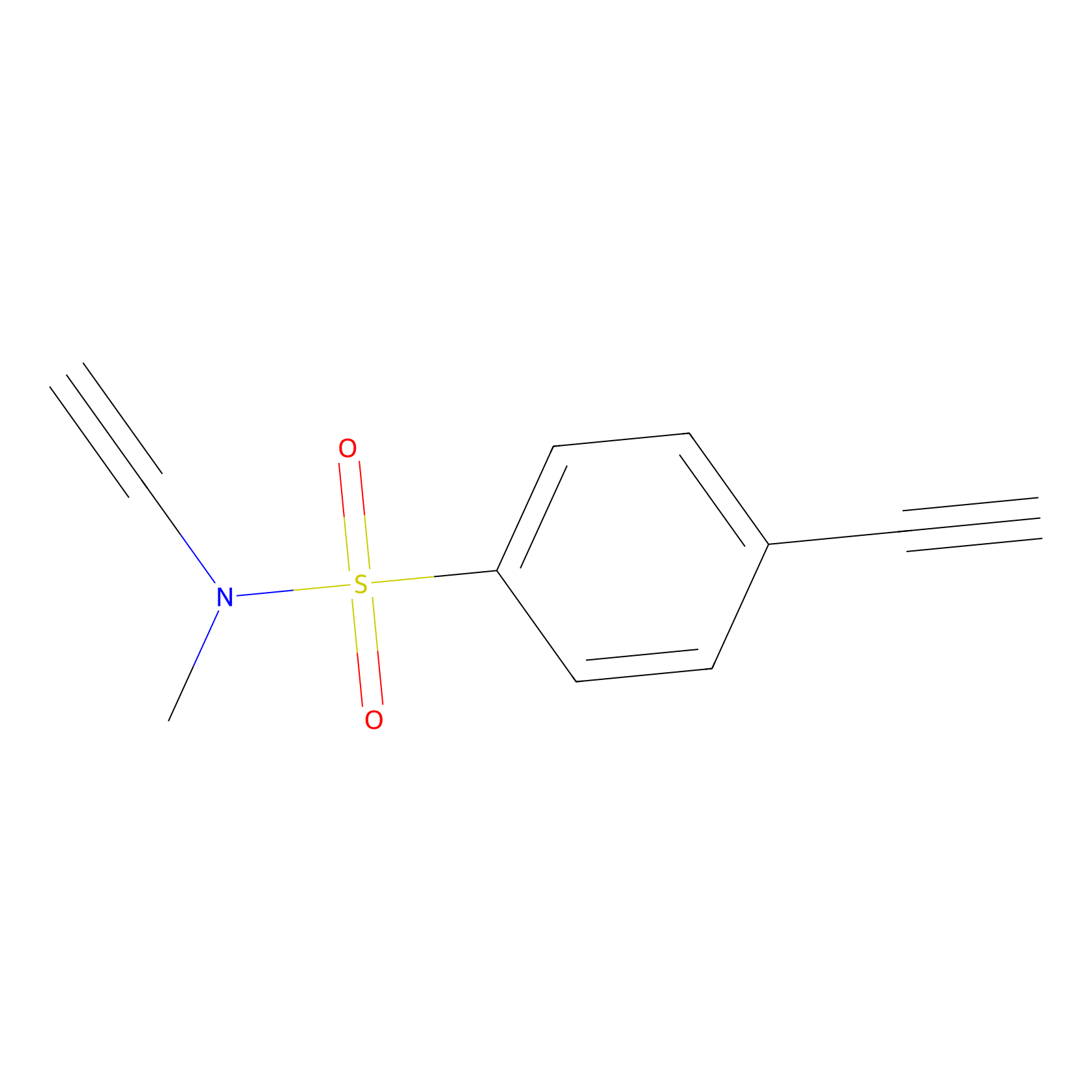 |
100.00 | LDD0444 | [1] | |
|
YN-4 Probe Info |
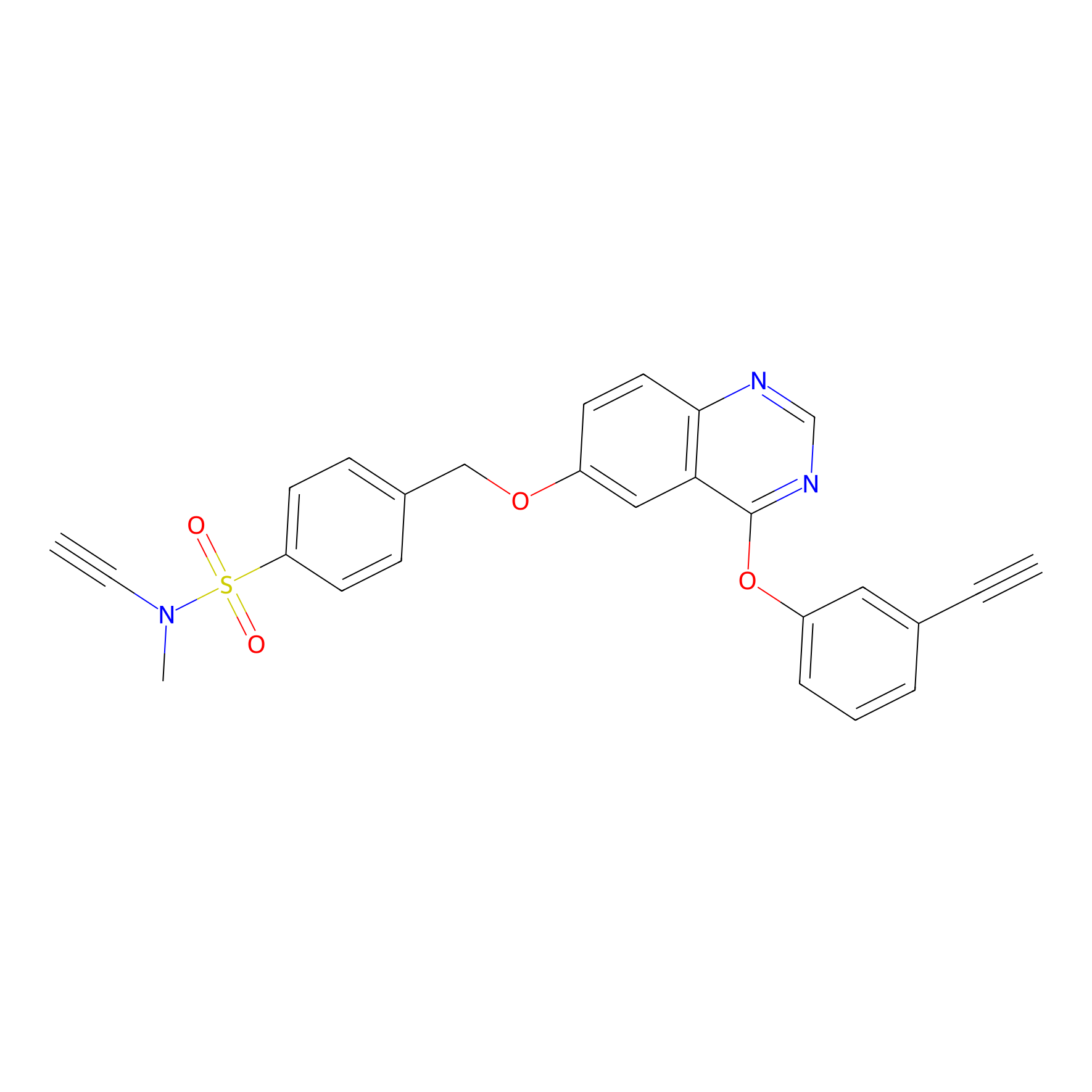 |
100.00 | LDD0445 | [1] | |
|
DBIA Probe Info |
 |
C289(1.15) | LDD3493 | [2] | |
|
ART-yne Probe Info |
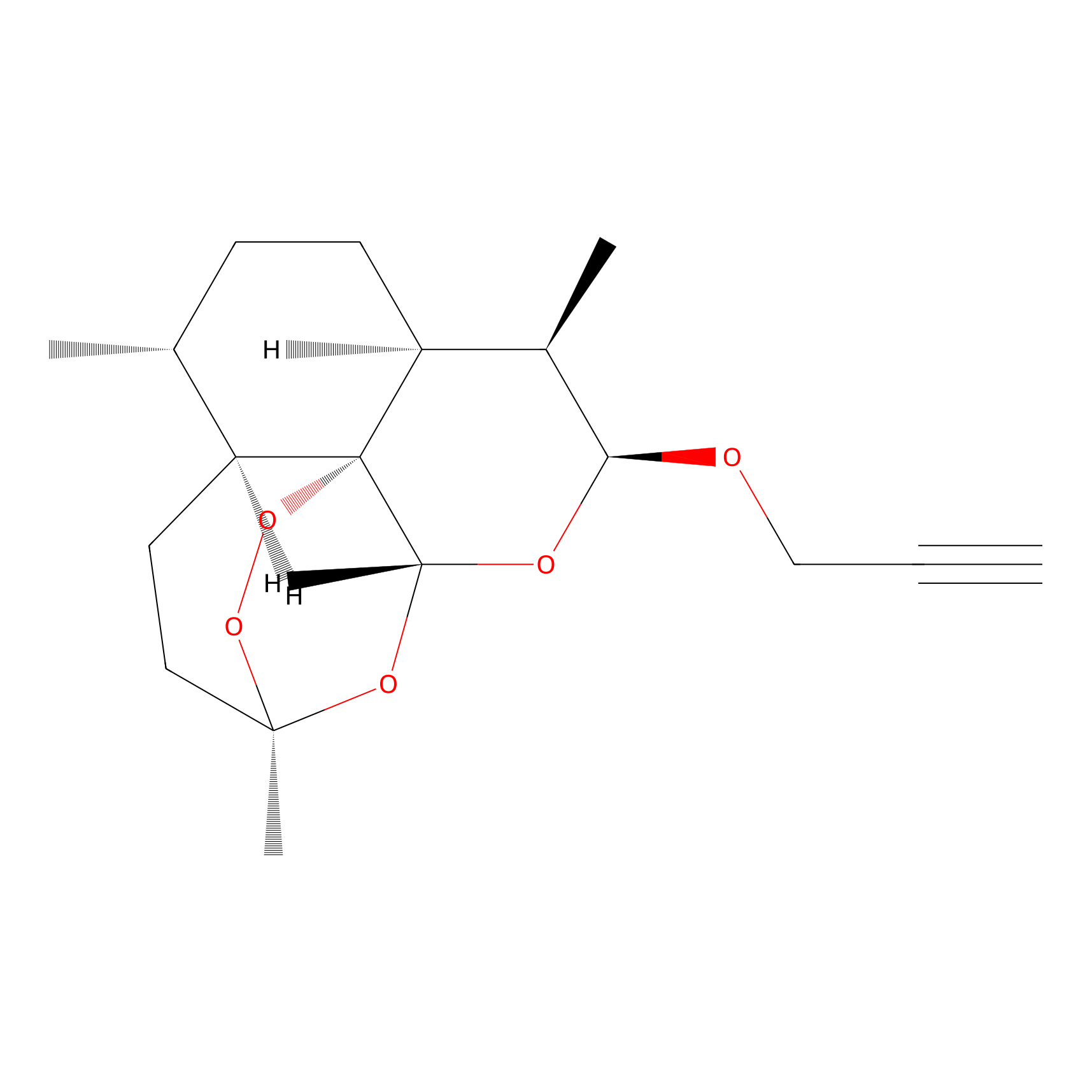 |
1.68 | LDD0234 | [3] | |
Competitor(s) Related to This Target
The Interaction Atlas With This Target
The Protein(s) Related To This Target
Cytokine and receptor
| Protein name | Family | Uniprot ID | |||
|---|---|---|---|---|---|
| CKLF-like MARVEL transmembrane domain-containing protein 5 (CMTM5) | Chemokine-like factor family | Q96DZ9 | |||
The Drug(s) Related To This Target
Approved
Investigative
Discontinued
| Drug Name | Drug Type | External ID | |||
|---|---|---|---|---|---|
| Phenacetin | Small molecular drug | DB03783 | |||
References
