Details of the Target
General Information of Target
| Target ID | LDTP10875 | |||||
|---|---|---|---|---|---|---|
| Target Name | D-aspartate oxidase (DDO) | |||||
| Gene Name | DDO | |||||
| Gene ID | 8528 | |||||
| Synonyms |
D-aspartate oxidase; DASOX; DASPO; DDO; EC 1.4.3.1 |
|||||
| 3D Structure | ||||||
| Sequence |
MGVNQSVGFPPVTGPHLVGCGDVMEGQNLQGSFFRLFYPCQKAEETMEQPLWIPRYEYCT
GLAEYLQFNKRCGGLLFNLAVGSCRLPVSWNGPFKTKDSGYPLIIFSHGLGAFRTLYSAF CMELASRGFVVAVPEHRDRSAATTYFCKQAPEENQPTNESLQEEWIPFRRVEEGEKEFHV RNPQVHQRVSECLRVLKILQEVTAGQTVFNILPGGLDLMTLKGNIDMSRVAVMGHSFGGA TAILALAKETQFRCAVALDAWMFPLERDFYPKARGPVFFINTEKFQTMESVNLMKKICAQ HEQSRIITVLGSVHRSQTDFAFVTGNLIGKFFSTETRGSLDPYEGQEVMVRAMLAFLQKH LDLKEDYNQWNNLIEGIGPSLTPGAPHHLSSL |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
DAMOX/DASOX family
|
|||||
| Subcellular location |
Peroxisome
|
|||||
| Function |
Selectively catalyzes the oxidative deamination of acidic amino acids. Suppresses the level of D-aspartate in the brain, an amino acid that can act as an agonist for glutamate receptors. Protects the organism from the toxicity of D-amino acids. May also function in the intestine.
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
| ChEMBL ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
DBIA Probe Info |
 |
C356(2.51) | LDD3439 | [1] | |
|
BTD Probe Info |
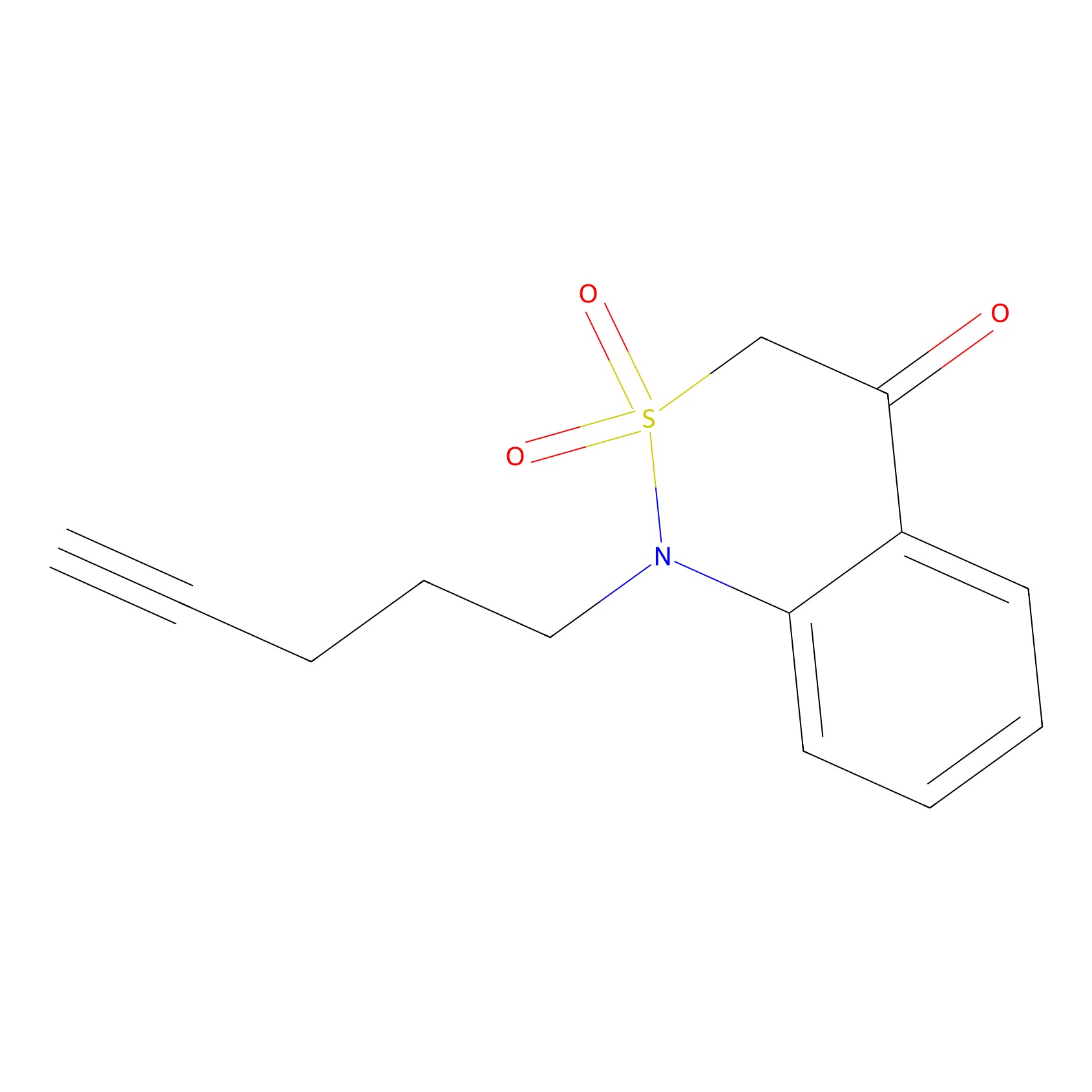 |
C269(1.93) | LDD1700 | [2] | |
|
IPM Probe Info |
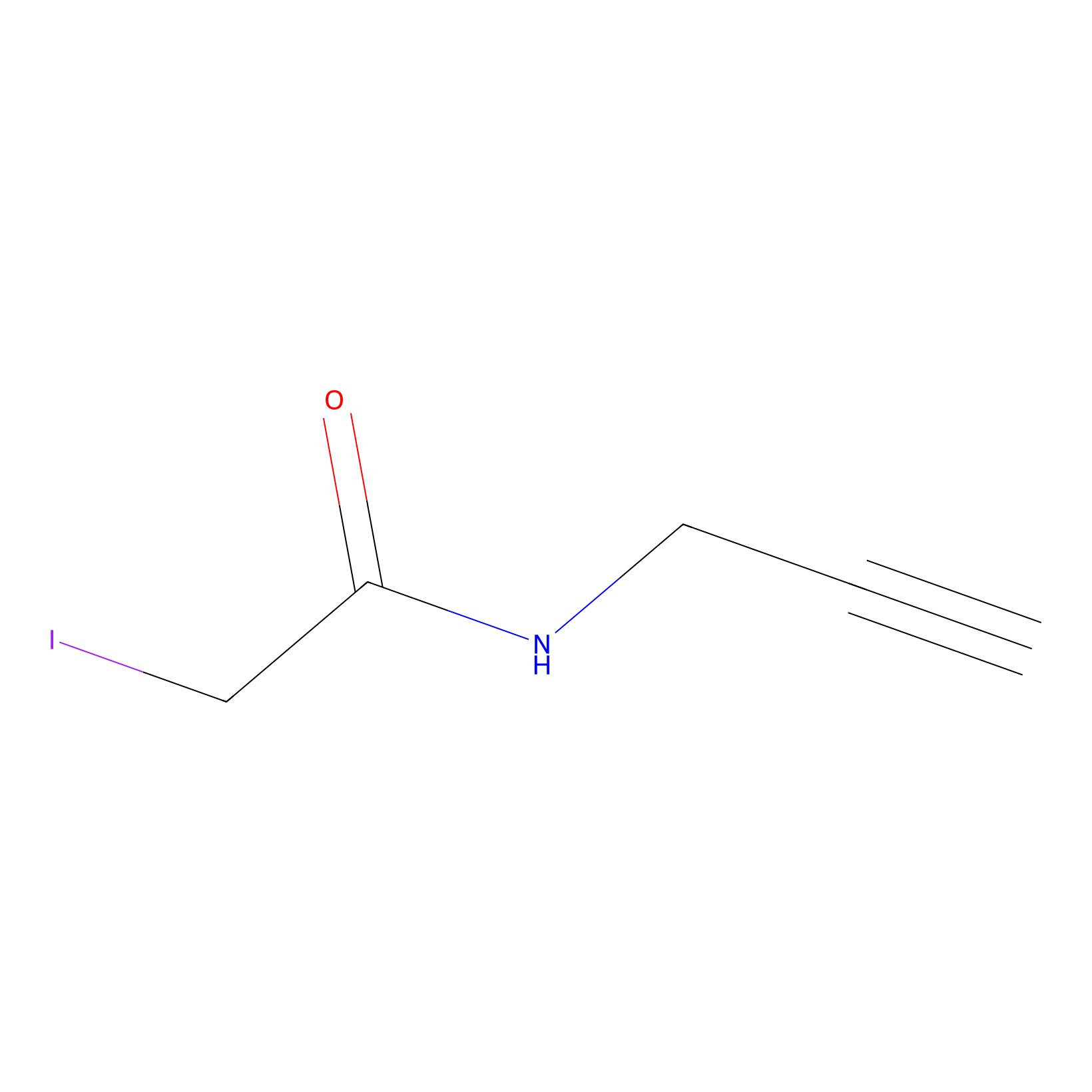 |
C269(28.63) | LDD1701 | [2] | |
|
Acrolein Probe Info |
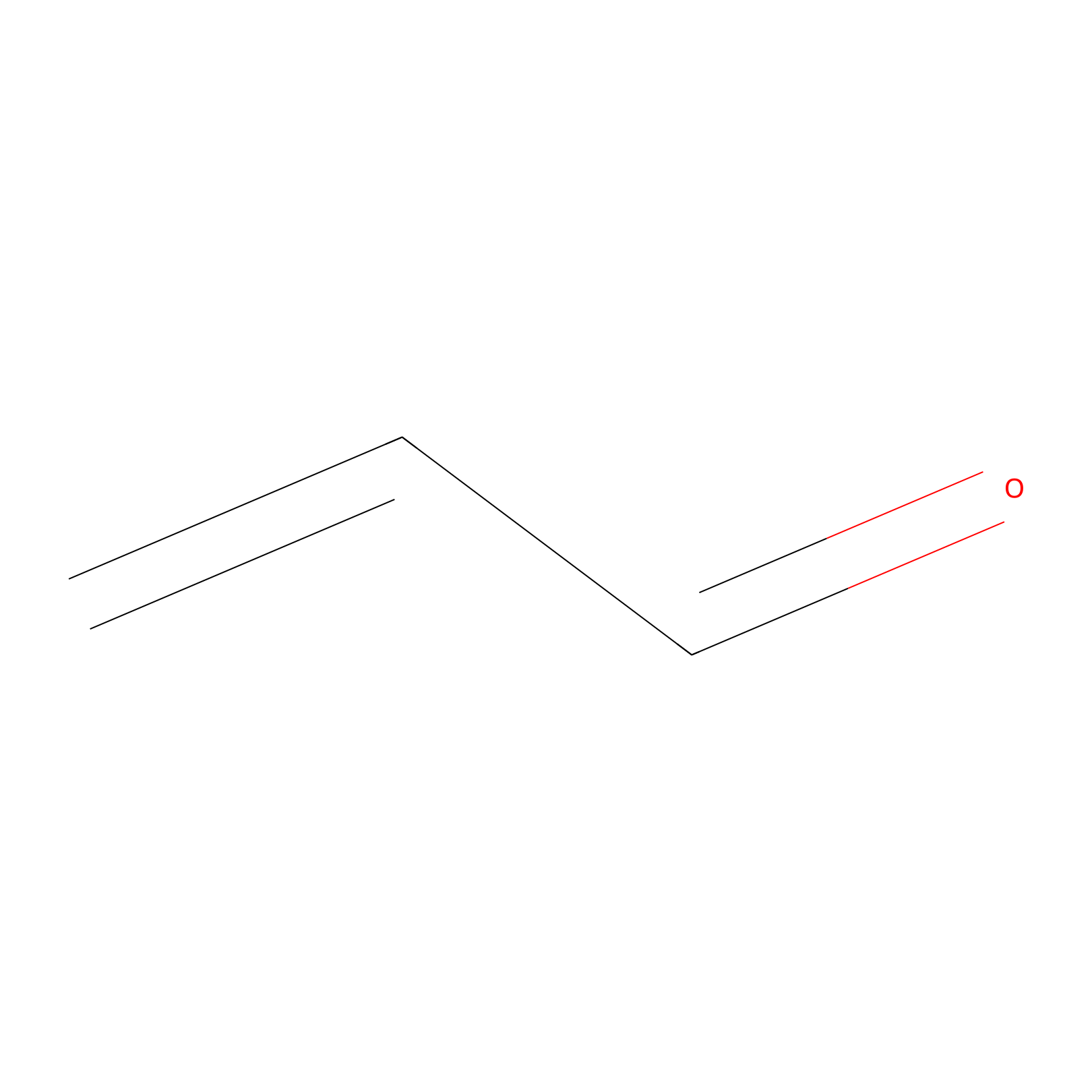 |
C328(0.00); H330(0.00) | LDD0221 | [3] | |
|
TFBX Probe Info |
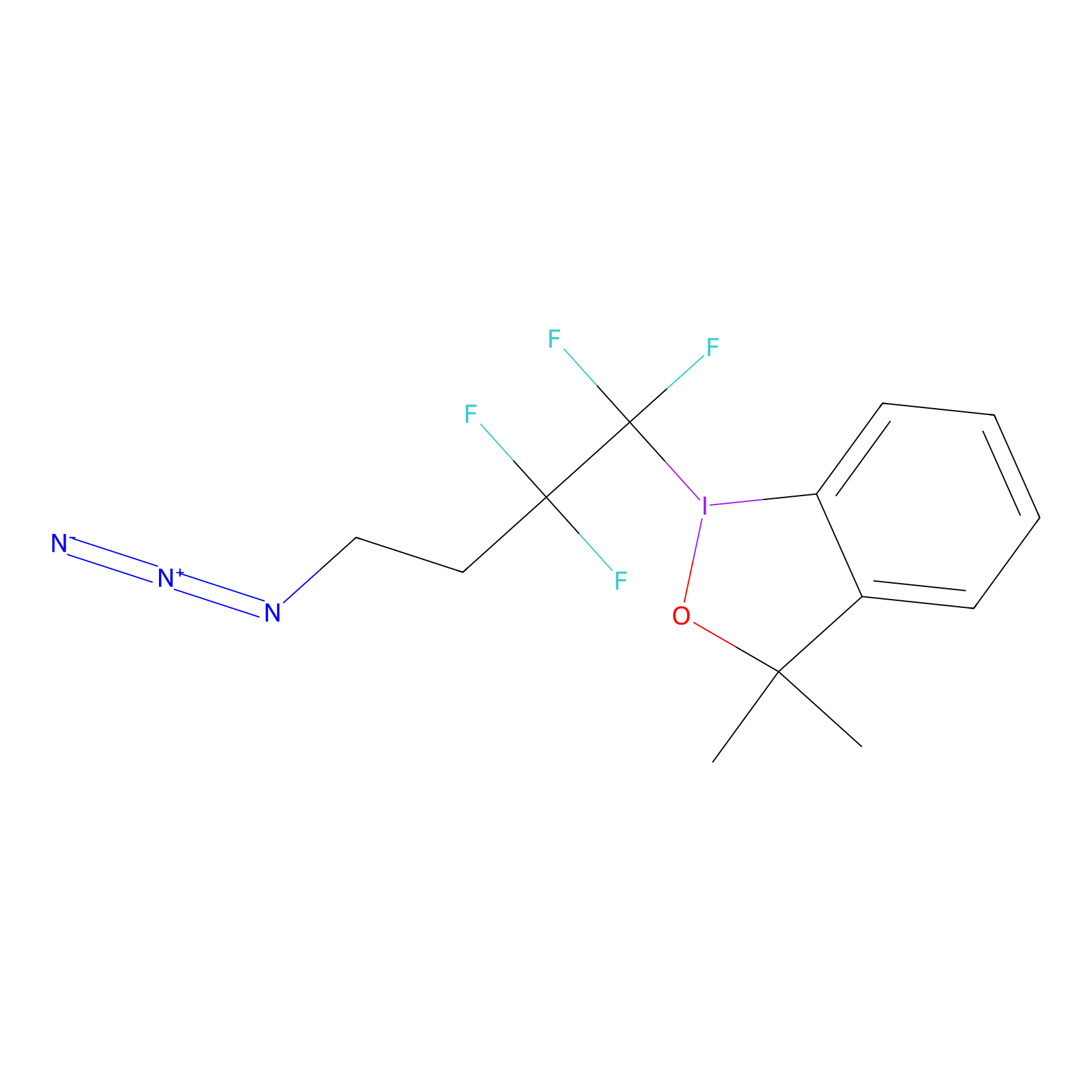 |
N.A. | LDD0148 | [4] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0519 | 1-(6-methoxy-3,4-dihydroquinolin-1(2H)-yl)-2-nitroethan-1-one | MDA-MB-231 | C269(0.95) | LDD2112 | [2] |
| LDCM0502 | 1-(Cyanoacetyl)piperidine | MDA-MB-231 | C269(0.65) | LDD2095 | [2] |
| LDCM0537 | 2-Cyano-N,N-dimethylacetamide | MDA-MB-231 | C269(0.95) | LDD2130 | [2] |
| LDCM0524 | 2-Cyano-N-(2-morpholin-4-yl-ethyl)-acetamide | MDA-MB-231 | C269(1.16) | LDD2117 | [2] |
| LDCM0539 | 3-(4-Isopropylpiperazin-1-yl)-3-oxopropanenitrile | MDA-MB-231 | C269(0.50) | LDD2132 | [2] |
| LDCM0545 | Acetamide | MDA-MB-231 | C269(0.61) | LDD2138 | [2] |
| LDCM0213 | Electrophilic fragment 2 | MDA-MB-231 | C269(7.40) | LDD1702 | [2] |
| LDCM0107 | IAA | HeLa | C328(0.00); H330(0.00) | LDD0221 | [3] |
| LDCM0022 | KB02 | EGI-1 | C356(1.22) | LDD2323 | [1] |
| LDCM0023 | KB03 | MDA-MB-231 | C269(28.63) | LDD1701 | [2] |
| LDCM0024 | KB05 | SNU-1196 | C356(2.51) | LDD3439 | [1] |
| LDCM0528 | N-(4-bromophenyl)-2-cyano-N-phenylacetamide | MDA-MB-231 | C269(0.86) | LDD2121 | [2] |
| LDCM0109 | NEM | HeLa | C328(0.00); H330(0.00) | LDD0224 | [3] |
| LDCM0506 | Nucleophilic fragment 16a | MDA-MB-231 | C269(0.97) | LDD2099 | [2] |
| LDCM0511 | Nucleophilic fragment 18b | MDA-MB-231 | C269(0.81) | LDD2104 | [2] |
| LDCM0513 | Nucleophilic fragment 19b | MDA-MB-231 | C269(0.65) | LDD2106 | [2] |
| LDCM0516 | Nucleophilic fragment 21a | MDA-MB-231 | C269(0.97) | LDD2109 | [2] |
| LDCM0521 | Nucleophilic fragment 23b | MDA-MB-231 | C269(1.02) | LDD2114 | [2] |
| LDCM0527 | Nucleophilic fragment 26b | MDA-MB-231 | C269(0.66) | LDD2120 | [2] |
| LDCM0532 | Nucleophilic fragment 29a | MDA-MB-231 | C269(0.76) | LDD2125 | [2] |
| LDCM0534 | Nucleophilic fragment 30a | MDA-MB-231 | C269(1.05) | LDD2127 | [2] |
| LDCM0543 | Nucleophilic fragment 38 | MDA-MB-231 | C269(1.54) | LDD2136 | [2] |
| LDCM0211 | Nucleophilic fragment 3b | MDA-MB-231 | C269(1.93) | LDD1700 | [2] |
| LDCM0546 | Nucleophilic fragment 40 | MDA-MB-231 | C269(0.68) | LDD2140 | [2] |
| LDCM0550 | Nucleophilic fragment 5a | MDA-MB-231 | C269(2.67) | LDD2144 | [2] |
| LDCM0559 | Nucleophilic fragment 9b | MDA-MB-231 | C269(2.64) | LDD2153 | [2] |
References
