Details of the Target
General Information of Target
| Target ID | LDTP05563 | |||||
|---|---|---|---|---|---|---|
| Target Name | Cytochrome P450 4F3 (CYP4F3) | |||||
| Gene Name | CYP4F3 | |||||
| Gene ID | 4051 | |||||
| Synonyms |
LTB4H; Cytochrome P450 4F3; EC 1.14.14.1; 20-hydroxyeicosatetraenoic acid synthase; 20-HETE synthase; CYPIVF3; Cytochrome P450-LTB-omega; Docosahexaenoic acid omega-hydroxylase CYP4F3; EC 1.14.14.79; Leukotriene-B(4) 20-monooxygenase 2; Leukotriene-B(4) omega-hydroxylase 2; EC 1.14.14.94
|
|||||
| 3D Structure | ||||||
| Sequence |
MPQLSLSSLGLWPMAASPWLLLLLVGASWLLARILAWTYTFYDNCCRLRCFPQPPKRNWF
LGHLGLIHSSEEGLLYTQSLACTFGDMCCWWVGPWHAIVRIFHPTYIKPVLFAPAAIVPK DKVFYSFLKPWLGDGLLLSAGEKWSRHRRMLTPAFHFNILKPYMKIFNESVNIMHAKWQL LASEGSARLDMFEHISLMTLDSLQKCVFSFDSHCQEKPSEYIAAILELSALVTKRHQQIL LYIDFLYYLTPDGQRFRRACRLVHDFTDAVIQERRRTLPSQGVDDFLQAKAKSKTLDFID VLLLSKDEDGKKLSDEDIRAEADTFMFEGHDTTASGLSWVLYHLAKHPEYQERCRQEVQE LLKDREPKEIEWDDLAQLPFLTMCIKESLRLHPPVPAVSRCCTQDIVLPDGRVIPKGIIC LISVFGTHHNPAVWPDPEVYDPFRFDPKNIKERSPLAFIPFSAGPRNCIGQAFAMAEMKV VLGLTLLRFRVLPDHTEPRRKPELVLRAEGGLWLRVEPLS |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Cytochrome P450 family
|
|||||
| Subcellular location |
Endoplasmic reticulum membrane
|
|||||
| Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids and their oxygenated derivatives (oxylipins). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (CPR; NADPH-ferrihemoprotein reductase). May play a role in inactivation of pro-inflammatory and anti-inflammatory oxylipins during the resolution of inflammation .; [Isoform CYP4F3A]: Catalyzes predominantly the oxidation of the terminal carbon (omega-oxidation) of oxylipins in myeloid cells, displaying higher affinity for arachidonate metabolite leukotriene B4 (LTB4). Inactivates LTB4 via three successive oxidative transformations to 20-hydroxy-LTB4, then to 20-oxo-LTB4 and to 20-carboxy-LTB4. Has omega-hydroxylase activity toward long-chain fatty acid epoxides with preference for 8,9-epoxy-(5Z,11Z,14Z)-eicosatrienoate (EET) and 9,10-epoxyoctadecanoate. Omega-hydroxylates monohydroxy polyunsaturated fatty acids (PUFAs), including hydroxyeicosatetraenoates (HETEs) and hydroxyeicosapentaenoates (HEPEs), to dihydroxy compounds. Contributes to the degradation of saturated very long-chain fatty acids (VLCFAs) such as docosanoic acid, by catalyzing successive omega-oxidations to the corresponding dicarboxylic acid, thereby initiating chain shortening. Has low hydroxylase activity toward PUFAs.; [Isoform CYP4F3B]: Catalyzes predominantly the oxidation of the terminal carbon (omega-oxidation) of polyunsaturated fatty acids (PUFAs). Participates in the conversion of arachidonic acid to 20-hydroxyeicosatetraenoic acid (20-HETE), a signaling molecule acting both as vasoconstrictive and natriuretic with overall effect on arterial blood pressure. Has high omega-hydroxylase activity toward other PUFAs, including eicosatrienoic acid (ETA), eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Can also catalyze the oxidation of the penultimate carbon (omega-1 oxidation) of PUFAs with lower efficiency. Contributes to the degradation of saturated very long-chain fatty acids (VLCFAs) such as docosanoic acid and hexacosanoic acid, by catalyzing successive omega-oxidations to the corresponding dicarboxylic acids, thereby initiating chain shortening. Omega-hydroxylates long-chain 3-hydroxy fatty acids, likely initiating the oxidative conversion to the corresponding 3-hydroxydicarboxylic fatty acids. Has omega-hydroxylase activity toward long-chain fatty acid epoxides with preference for 8,9-epoxy-(5Z,11Z,14Z)-eicosatrienoate (EET) and 9,10-epoxyoctadecanoate.
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
| ChEMBL ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
YN-4 Probe Info |
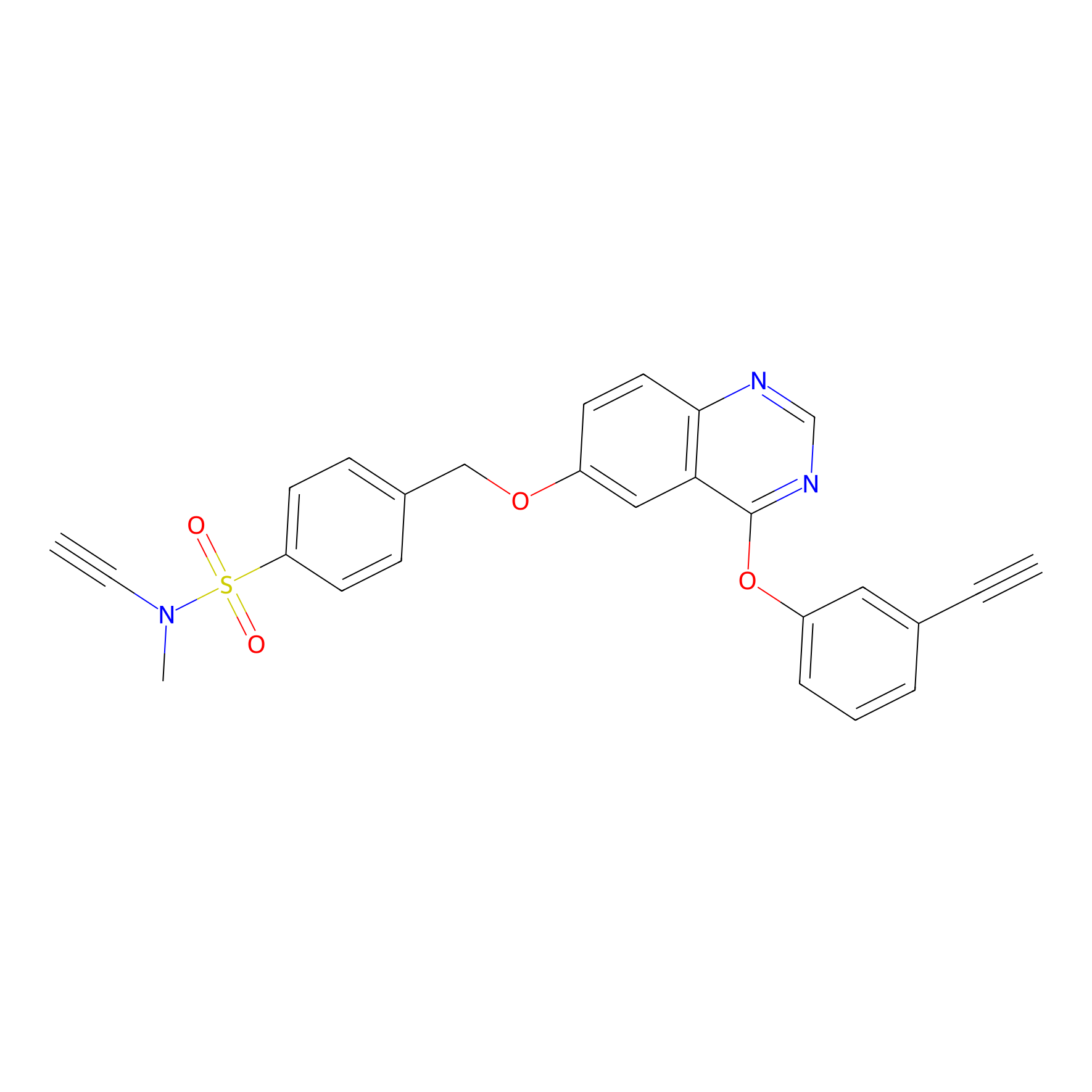 |
100.00 | LDD0445 | [1] | |
|
N1 Probe Info |
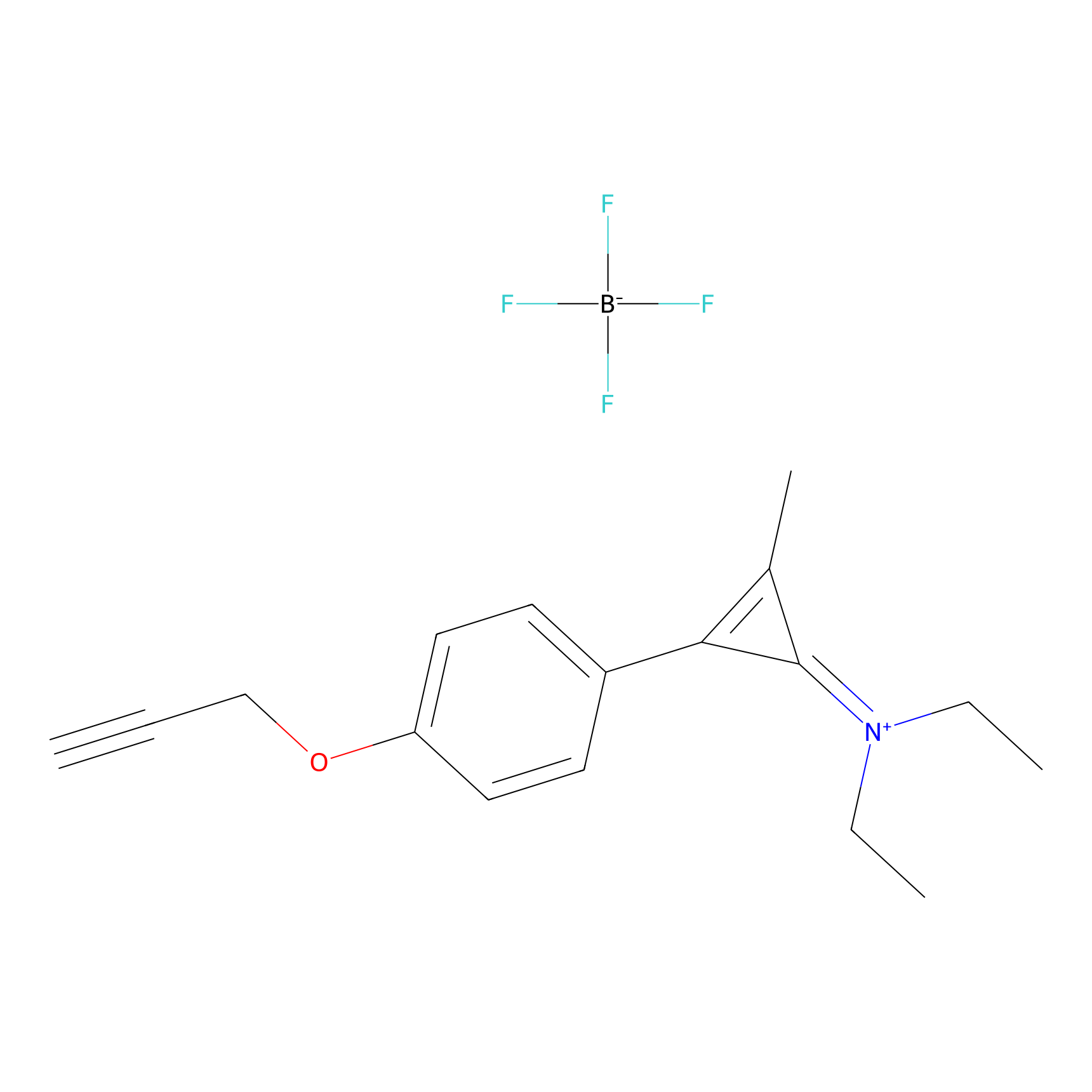 |
N.A. | LDD0245 | [2] | |
|
IA-alkyne Probe Info |
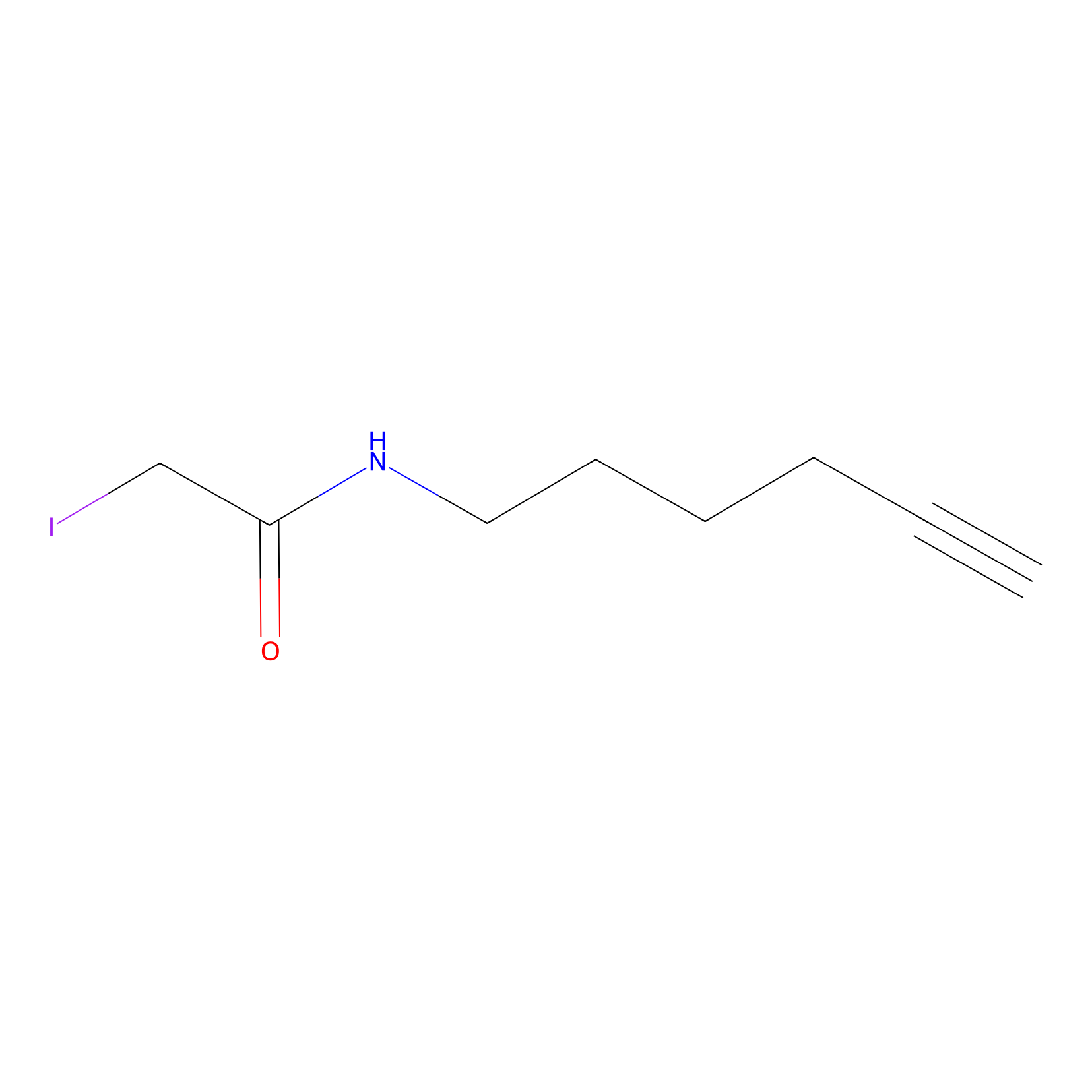 |
C354(0.00); C384(0.00); C420(0.00); C468(0.00) | LDD0162 | [3] | |
The Interaction Atlas With This Target
References
