Details of the Target
General Information of Target
| Target ID | LDTP11394 | |||||
|---|---|---|---|---|---|---|
| Target Name | Serine/threonine-protein kinase PINK1, mitochondrial (PINK1) | |||||
| Gene Name | PINK1 | |||||
| Gene ID | 65018 | |||||
| Synonyms |
Serine/threonine-protein kinase PINK1, mitochondrial; EC 2.7.11.1; BRPK; PTEN-induced putative kinase protein 1 |
|||||
| 3D Structure | ||||||
| Sequence |
MEARSRSAEELRRAELVEIIVETEAQTGVSGINVAGGGKEGIFVRELREDSPAARSLSLQ
EGDQLLSARVFFENFKYEDALRLLQCAEPYKVSFCLKRTVPTGDLALRPGTVSGYEIKGP RAKVAKLNIQSLSPVKKKKMVPGALGVPADLAPVDVEFSFPKFSRLRRGLKAEAVKGPVP AAPARRRLQLPRLRVREVAEEAQAARLAAAAPPPRKAKVEAEVAAGARFTAPQVELVGPR LPGAEVGVPQVSAPKAAPSAEAAGGFALHLPTLGLGAPAPPAVEAPAVGIQVPQVELPAL PSLPTLPTLPCLETREGAVSVVVPTLDVAAPTVGVDLALPGAEVEARGEAPEVALKMPRL SFPRFGARAKEVAEAKVAKVSPEARVKGPRLRMPTFGLSLLEPRPAAPEVVESKLKLPTI KMPSLGIGVSGPEVKVPKGPEVKLPKAPEVKLPKVPEAALPEVRLPEVELPKVSEMKLPK VPEMAVPEVRLPEVELPKVSEMKLPKVPEMAVPEVRLPEVQLLKVSEMKLPKVPEMAVPE VRLPEVQLPKVSEMKLPEVSEVAVPEVRLPEVQLPKVPEMKVPEMKLPKVPEMKLPEMKL PEVQLPKVPEMAVPDVHLPEVQLPKVPEMKLPEMKLPEVKLPKVPEMAVPDVHLPEVQLP KVPEMKLPKMPEMAVPEVRLPEVQLPKVSEMKLPKVPEMAVPDVHLPEVQLPKVCEMKVP DMKLPEIKLPKVPEMAVPDVHLPEVQLPKVSEIRLPEMQVPKVPDVHLPKAPEVKLPRAP EVQLKATKAEQAEGMEFGFKMPKMTMPKLGRAESPSRGKPGEAGAEVSGKLVTLPCLQPE VDGEAHVGVPSLTLPSVELDLPGALGLQGQVPAAKMGKGERVEGPEVAAGVREVGFRVPS VEIVTPQLPAVEIEEGRLEMIETKVKPSSKFSLPKFGLSGPKVAKAEAEGAGRATKLKVS KFAISLPKARVGAEAEAKGAGEAGLLPALDLSIPQLSLDAHLPSGKVEVAGADLKFKGPR FALPKFGVRGRDTEAAELVPGVAELEGKGWGWDGRVKMPKLKMPSFGLARGKEAEVQGDR ASPGEKAESTAVQLKIPEVELVTLGAQEEGRAEGAVAVSGMQLSGLKVSTAGQVVTEGHD AGLRMPPLGISLPQVELTGFGEAGTPGQQAQSTVPSAEGTAGYRVQVPQVTLSLPGAQVA GGELLVGEGVFKMPTVTVPQLELDVGLSREAQAGEAATGEGGLRLKLPTLGARARVGGEG AEEQPPGAERTFCLSLPDVELSPSGGNHAEYQVAEGEGEAGHKLKVRLPRFGLVRAKEGA EEGEKAKSPKLRLPRVGFSQSEMVTGEGSPSPEEEEEEEEEGSGEGASGRRGRVRVRLPR VGLAAPSKASRGQEGDAAPKSPVREKSPKFRFPRVSLSPKARSGSGDQEEGGLRVRLPSV GFSETGAPGPARMEGAQAAAV |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Protein kinase superfamily, Ser/Thr protein kinase family
|
|||||
| Subcellular location |
Mitochondrion outer membrane
|
|||||
| Function |
Serine/threonine-protein kinase which protects against mitochondrial dysfunction during cellular stress by phosphorylating mitochondrial proteins such as PRKN and DNM1L, to coordinate mitochondrial quality control mechanisms that remove and replace dysfunctional mitochondrial components. Depending on the severity of mitochondrial damage and/or dysfunction, activity ranges from preventing apoptosis and stimulating mitochondrial biogenesis to regulating mitochondrial dynamics and eliminating severely damaged mitochondria via mitophagy . Mediates the translocation and activation of PRKN at the outer membrane (OMM) of dysfunctional/depolarized mitochondria. At the OMM of damaged mitochondria, phosphorylates pre-existing polyubiquitin chains at 'Ser-65', the PINK1-phosphorylated polyubiquitin then recruits PRKN from the cytosol to the OMM where PRKN is fully activated by phosphorylation at 'Ser-65' by PINK1. In damaged mitochondria, mediates the decision between mitophagy or preventing apoptosis by promoting PRKN-dependent poly- or monoubiquitination of VDAC1; polyubiquitination of VDAC1 by PRKN promotes mitophagy, while monoubiquitination of VDAC1 by PRKN decreases mitochondrial calcium influx which ultimately inhibits apoptosis. When cellular stress results in irreversible mitochondrial damage, functions with PRKN to promote clearance of damaged mitochondria via selective autophagy (mitophagy) . The PINK1-PRKN pathway also promotes fission of damaged mitochondria by phosphorylating and thus promoting the PRKN-dependent degradation of mitochondrial proteins involved in fission such as MFN2. This prevents the refusion of unhealthy mitochondria with the mitochondrial network or initiates mitochondrial fragmentation facilitating their later engulfment by autophagosomes. Also promotes mitochondrial fission independently of PRKN and ATG7-mediated mitophagy, via the phosphorylation and activation of DNM1L. Regulates motility of damaged mitochondria by promoting the ubiquitination and subsequent degradation of MIRO1 and MIRO2; in motor neurons, this likely inhibits mitochondrial intracellular anterograde transport along the axons which probably increases the chance of the mitochondria undergoing mitophagy in the soma. Required for ubiquinone reduction by mitochondrial complex I by mediating phosphorylation of complex I subunit NDUFA10. Phosphorylates LETM1, positively regulating its mitochondrial calcium transport activity.
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
| ChEMBL ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
IA-alkyne Probe Info |
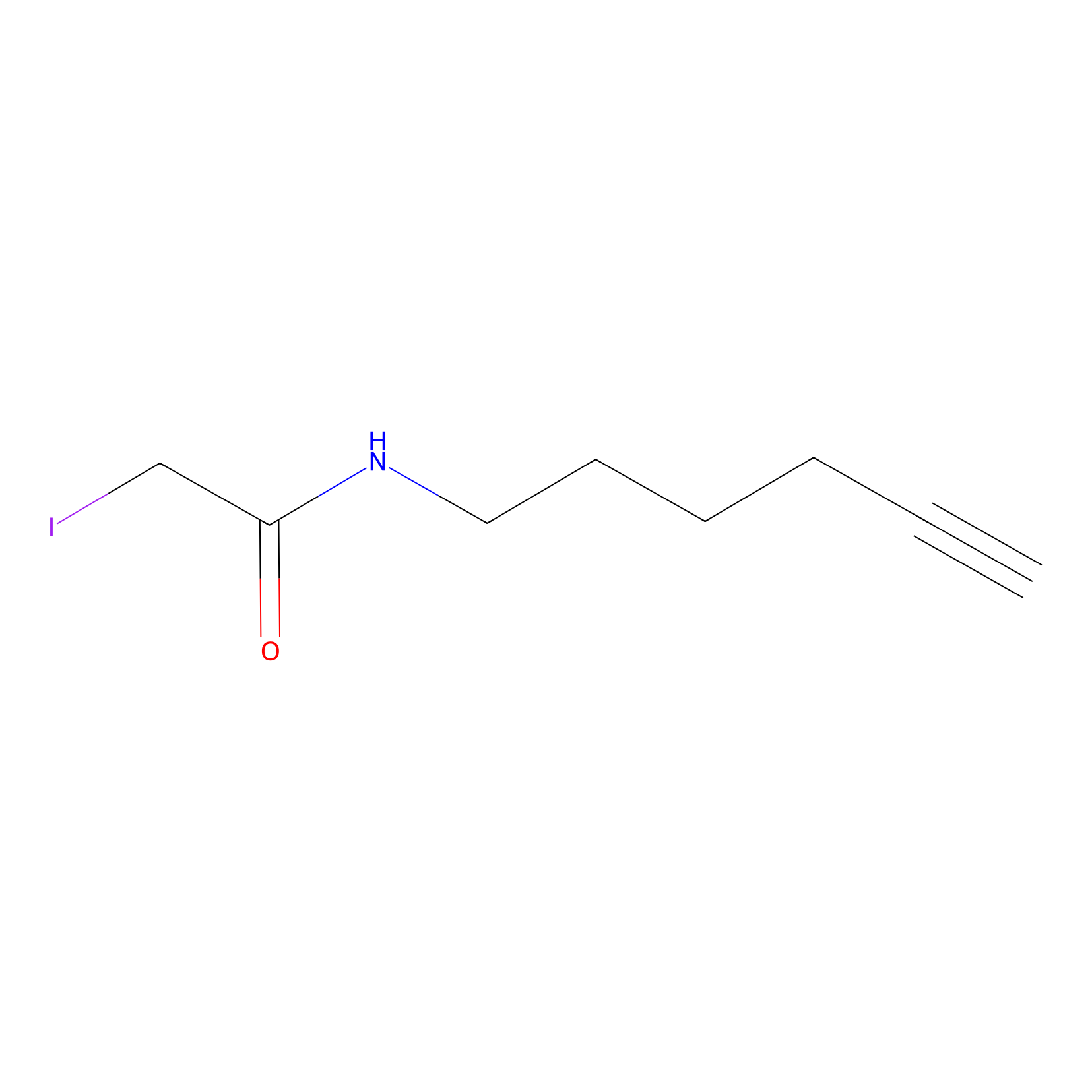 |
N.A. | LDD0166 | [1] | |
|
IPM Probe Info |
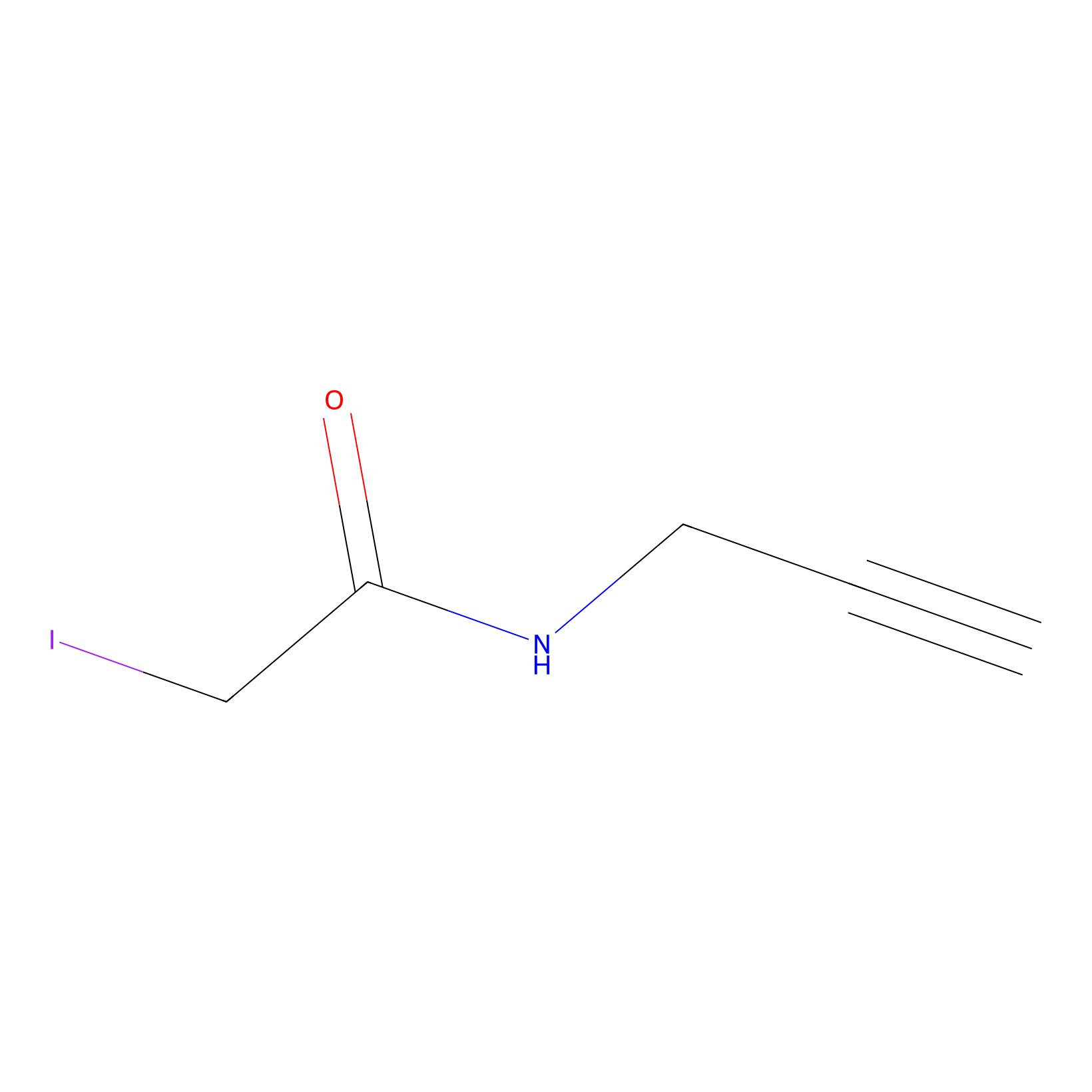 |
N.A. | LDD0147 | [2] | |
The Interaction Atlas With This Target
The Protein(s) Related To This Target
Enzyme
Transporter and channel
| Protein name | Family | Uniprot ID | |||
|---|---|---|---|---|---|
| Amyloid-beta precursor protein (APP) | APP family | P05067 | |||
| Huntingtin (HTT) | Huntingtin family | P42858 | |||
Transcription factor
| Protein name | Family | Uniprot ID | |||
|---|---|---|---|---|---|
| Zinc finger protein 366 (ZNF366) | . | Q8N895 | |||
Other
References
