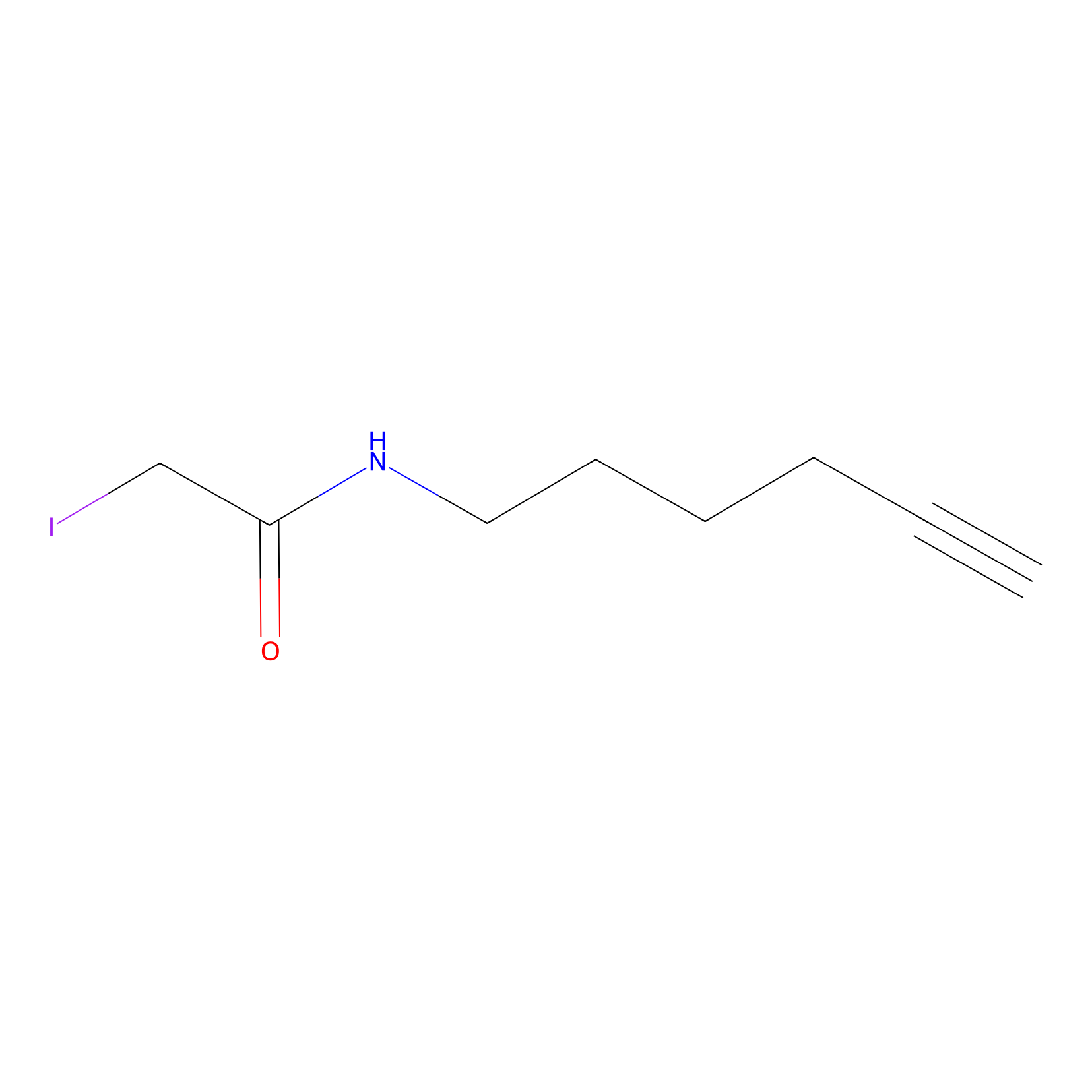
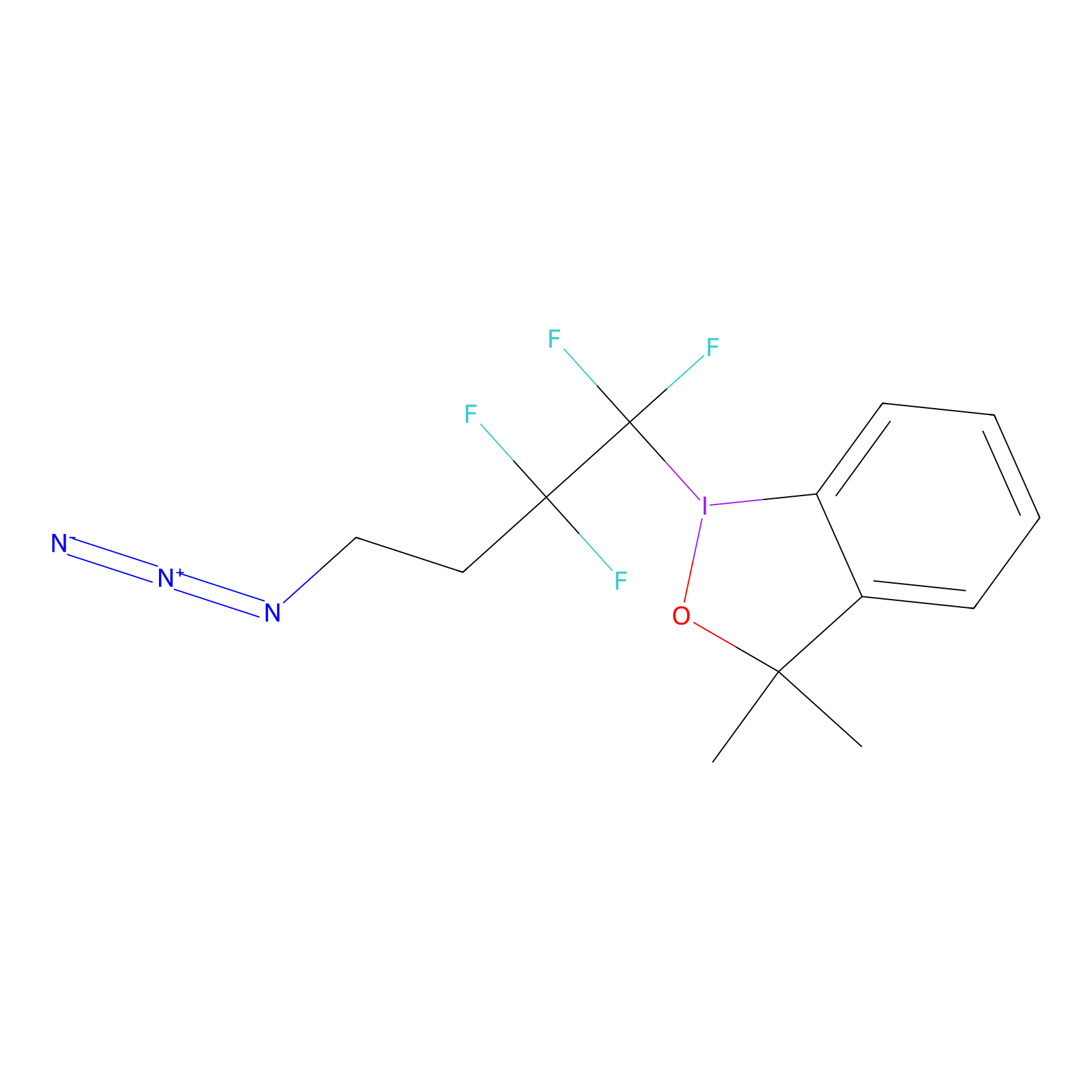
| Drug Name |
Drug Type |
External ID |
| (E)-8-biphenyl-4-yl-1-oxazol-2-yl-oct-7-en-1-one |
Small molecular drug |
D0KE3I
|
| 2-(Methylsulfonylthio)Ethyl 2-propylpentanoate |
Small molecular drug |
D0H1FZ
|
| 4-benzoylamino-n-hydroxy-benzamide |
Small molecular drug |
D09ZNU
|
| 4-butyrylamino-n-hydroxy-benzamide |
Small molecular drug |
D05LLX
|
| 4-chloro-n-(5-hydroxycarbamoyl-pentyl)-benzamide |
Small molecular drug |
D0A5LS
|
| 4-dimethylamino-n-(6-mercapto-hexyl)-benzamide |
Small molecular drug |
D06XNP
|
| 4-hydroxy-n-(5-hydroxycarbamoyl-pentyl)-benzamide |
Small molecular drug |
D04NSP
|
| 5-(4-chloro-phenyl)-pentanoic Acid Hydroxyamide |
Small molecular drug |
D0V2ZO
|
| 5-(4-hydroxyphenyl)-3h-1,2-dithiole-3-thione |
Small molecular drug |
D06IQB
|
| 5-mercapto-pentanoic Acid Phenylamide |
Small molecular drug |
D0D2KE
|
| 6-(2-bromo-acetylamino)-hexanoic Acid Phenylamide |
Small molecular drug |
D02MQM
|
| 6-benzenesulfinylhexanoic Acid Hydroxamide |
Small molecular drug |
D00ZSD
|
| 6-benzenesulfonylhexanoic Acid Hydroxamide |
Small molecular drug |
D05AHZ
|
| 6-mercapto-hexanoic Acid Phenylamide |
Small molecular drug |
D0B3ZM
|
| 6-phenoxy-hexane-1-thiol |
Small molecular drug |
D09YBN
|
| 6-phenylsulfanylhexanoic Acid Hydroxamide |
Small molecular drug |
D0S9JF
|
| 7-(Biphenyl-3-yloxy)-1-oxazol-2-yl-heptan-1-one |
Small molecular drug |
D0B2ZJ
|
| 7-(Biphenyl-4-yloxy)-1,1,1-trifluoro-heptan-2-one |
Small molecular drug |
D0Z3BM
|
| 7-(Biphenyl-4-yloxy)-1-oxazol-2-yl-heptan-1-one |
Small molecular drug |
D02WZK
|
| 7-(Naphthalen-2-yloxy)-1-oxazol-2-yl-heptan-1-one |
Small molecular drug |
D05VFP
|
| 7-mercapto-heptanoic Acid Benzothiazol-2-ylamide |
Small molecular drug |
D0Z5KX
|
| 7-mercapto-heptanoic Acid Biphenyl-3-ylamide |
Small molecular drug |
D05XAN
|
| 7-mercapto-heptanoic Acid Biphenyl-4-ylamide |
Small molecular drug |
D01XWZ
|
| 7-mercapto-heptanoic Acid Phenylamide |
Small molecular drug |
D00SEE
|
| 7-mercapto-heptanoic Acid Pyridin-3-ylamide |
Small molecular drug |
D07FJU
|
| 7-mercapto-heptanoic Acid Quinolin-3-ylamide |
Small molecular drug |
D0T4IY
|
| 8-(Biphenyl-4-yloxy)-1,1,1-trifluoro-octan-2-one |
Small molecular drug |
D0AX7B
|
| 8-mercapto-octanoic Acid Phenylamide |
Small molecular drug |
D0XZ9Q
|
| 8-oxo-8-phenyl-octanoic Acid |
Small molecular drug |
D0RU1X
|
| 9,9,9-trifluoro-8-oxo-nonanoic Acid Phenylamide |
Small molecular drug |
D00KFF
|
| 9-(Biphenyl-4-yloxy)-1,1,1-trifluoro-nonan-2-one |
Small molecular drug |
D05FEJ
|
| Abexinostat |
Small molecular drug |
DB12565
|
| Cyclostellettamine Derivative |
Small molecular drug |
D0C0VP
|
| N-(2-mercapto-ethyl)-n'-phenyl-oxalamide |
Small molecular drug |
D0F0KS
|
| N-(2-mercapto-ethyl)-n'-phenyl-succinamide |
Small molecular drug |
D01MFV
|
| N-(5-hydroxycarbamoyl-pentyl)-4-nitro-benzamide |
Small molecular drug |
D0C2KD
|
| N-(6-hydroxycarbamoyl-hexyl)-benzamide |
Small molecular drug |
D0YY9M
|
| N-(6-mercapto-hexyl)-benzamide |
Small molecular drug |
D04HSS
|
| N-hydroxy-4-((R)-2-phenyl-butyrylamino)-benzamide |
Small molecular drug |
D04ZDI
|
| N-hydroxy-4-((S)-2-phenyl-butyrylamino)-benzamide |
Small molecular drug |
D0QO0L
|
| N-hydroxy-4-(2-phenyl-butyrylamino)-benzamide |
Small molecular drug |
D06VBN
|
| N-hydroxy-4-(4-phenyl-butyrylamino)-benzamide |
Small molecular drug |
D0S7WQ
|
| N-hydroxy-4-(5-phenyl-pentanoylamino)-benzamide |
Small molecular drug |
D0R5VG
|
| N-hydroxy-4-(Pentanoylamino-methyl)-benzamide |
Small molecular drug |
D0Z7YS
|
| N-hydroxy-4-(Phenylacetylamino-methyl)-benzamide |
Small molecular drug |
D02UTC
|
| N-hydroxy-4-phenylacetylamino-benzamide |
Small molecular drug |
D01XXN
|
| Octanedioic Acid Hydroxyamide Pyridin-2-ylamide |
Small molecular drug |
D0E8KR
|
| Octanedioic Acid Hydroxyamide Pyridin-4-ylamide |
Small molecular drug |
D04GGW
|
| Psammaplin A |
Small molecular drug |
D0N0TS
|
| St-2986 |
Small molecular drug |
D0N9AK
|
| St-2987 |
Small molecular drug |
D0T0SK
|
| St-3050 |
Small molecular drug |
D01YBR
|
| Thioacetic Acid S-(6-phenylcarbamoyl-hexyl) Ester |
Small molecular drug |
D05GVY
|
| N-hydroxy-4-(3-phenyl-propionylamino)-benzamide |
. |
D0R4BS
|