Details of the Target
General Information of Target
| Target ID | LDTP09318 | |||||
|---|---|---|---|---|---|---|
| Target Name | Cytochrome P450 2W1 (CYP2W1) | |||||
| Gene Name | CYP2W1 | |||||
| Gene ID | 54905 | |||||
| Synonyms |
Cytochrome P450 2W1; EC 1.14.14.-; CYPIIW1 |
|||||
| 3D Structure | ||||||
| Sequence |
MALLLLLFLGLLGLWGLLCACAQDPSPAARWPPGPRPLPLVGNLHLLRLSQQDRSLMELS
ERYGPVFTVHLGRQKTVVLTGFEAVKEALAGPGQELADRPPIAIFQLIQRGGGIFFSSGA RWRAARQFTVRALHSLGVGREPVADKILQELKCLSGQLDGYRGRPFPLALLGWAPSNITF ALLFGRRFDYRDPVFVSLLGLIDEVMVLLGSPGLQLFNVYPWLGALLQLHRPVLRKIEEV RAILRTLLEARRPHVCPGDPVCSYVDALIQQGQGDDPEGLFAEANAVACTLDMVMAGTET TSATLQWAALLMGRHPDVQGRVQEELDRVLGPGRTPRLEDQQALPYTSAVLHEVQRFITL LPHVPRCTAADTQLGGFLLPKGTPVIPLLTSVLLDETQWQTPGQFNPGHFLDANGHFVKR EAFLPFSAGRRVCVGERLARTELFLLFAGLLQRYRLLPPPGVSPASLDTTPARAFTMRPR AQALCAVPRP |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Cytochrome P450 family
|
|||||
| Subcellular location |
Endoplasmic reticulum lumen
|
|||||
| Function |
A cytochrome P450 monooxygenase that may play a role in retinoid and phospholipid metabolism. Catalyzes the hydroxylation of saturated carbon hydrogen bonds. Hydroxylates all trans-retinoic acid (atRA) to 4-hydroxyretinoate and may regulate atRA clearance. Other retinoids such as all-trans retinol and all-trans retinal are potential endogenous substrates. Catalyzes both epoxidation of double bonds and hydroxylation of carbon hydrogen bonds of the fatty acyl chain of 1-acylphospholipids/2-lysophospholipids. Can metabolize various lysophospholipids classes including lysophosphatidylcholines (LPCs), lysophosphatidylinositols (LPIs), lysophosphatidylserines (LPSs), lysophosphatidylglycerols (LPGs), lysophosphatidylethanolamines (LPEs) and lysophosphatidic acids (LPAs). Has low or no activity toward 2-acylphospholipids/1-lysophospholipids, diacylphospholipids and free fatty acids. May play a role in tumorigenesis by activating procarcinogens such as aflatoxin B1, polycyclic aromatic hydrocarbon dihydrodiols and aromatic amines. Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (CPR; NADPH-ferrihemoprotein reductase).
|
|||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
| ChEMBL ID | ||||||
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
IPM Probe Info |
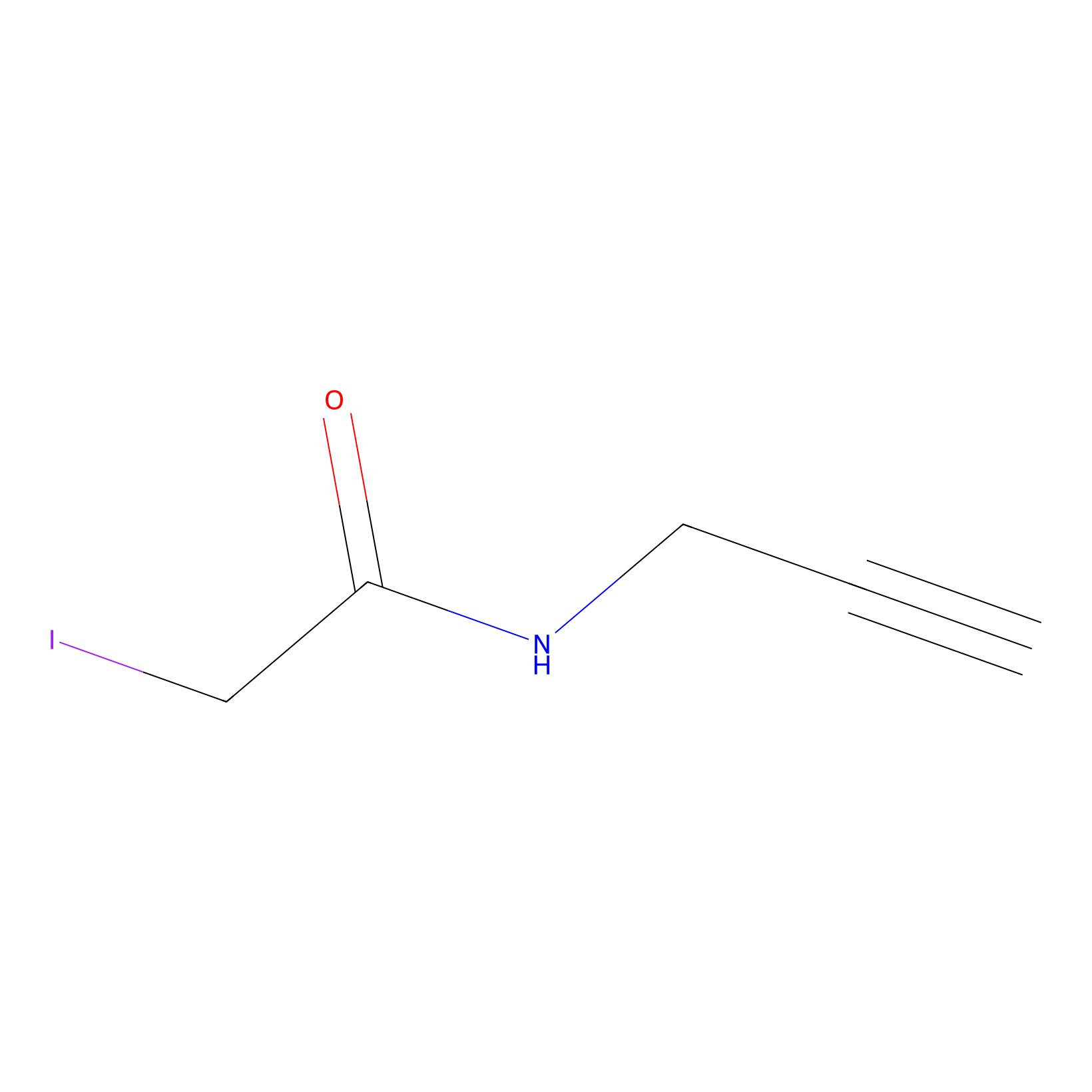 |
C153(0.00); C433(0.00) | LDD0241 | [1] | |
|
DBIA Probe Info |
 |
C153(1.87) | LDD3452 | [2] | |
|
W1 Probe Info |
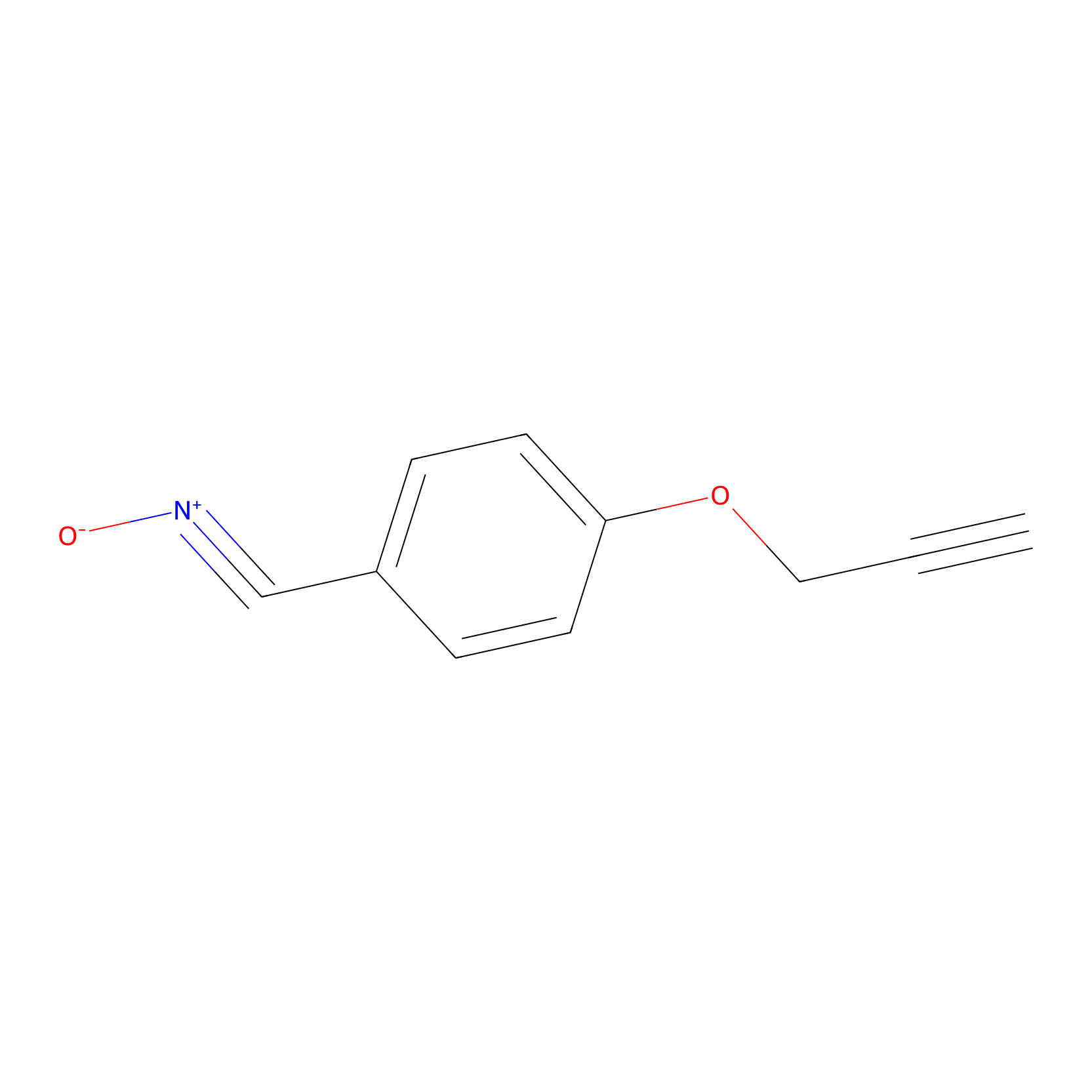 |
R366(0.00); H363(0.00) | LDD0236 | [1] | |
Competitor(s) Related to This Target
References
