Details of the Target
General Information of Target
| Target ID | LDTP08474 | |||||
|---|---|---|---|---|---|---|
| Target Name | 5-phosphohydroxy-L-lysine phospho-lyase (PHYKPL) | |||||
| Gene Name | PHYKPL | |||||
| Gene ID | 85007 | |||||
| Synonyms |
AGXT2L2; 5-phosphohydroxy-L-lysine phospho-lyase; EC 4.2.3.134; Alanine--glyoxylate aminotransferase 2-like 2 |
|||||
| 3D Structure | ||||||
| Sequence |
MAADQRPKADTLALRQRLISSSCRLFFPEDPVKIVRAQGQYMYDEQGAEYIDCISNVAHV
GHCHPLVVQAAHEQNQVLNTNSRYLHDNIVDYAQRLSETLPEQLCVFYFLNSGSEANDLA LRLARHYTGHQDVVVLDHAYHGHLSSLIDISPYKFRNLDGQKEWVHVAPLPDTYRGPYRE DHPNPAMAYANEVKRVVSSAQEKGRKIAAFFAESLPSVGGQIIPPAGYFSQVAEHIRKAG GVFVADEIQVGFGRVGKHFWAFQLQGKDFVPDIVTMGKSIGNGHPVACVAATQPVARAFE ATGVEYFNTFGGSPVSCAVGLAVLNVLEKEQLQDHATSVGSFLMQLLGQQKIKHPIVGDV RGVGLFIGVDLIKDEATRTPATEEAAYLVSRLKENYVLLSTDGPGRNILKFKPPMCFSLD NARQVVAKLDAILTDMEEKVRSCETLRLQP |
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Class-III pyridoxal-phosphate-dependent aminotransferase family
|
|||||
| Subcellular location |
Mitochondrion
|
|||||
| Function | Catalyzes the pyridoxal-phosphate-dependent breakdown of 5-phosphohydroxy-L-lysine, converting it to ammonia, inorganic phosphate and 2-aminoadipate semialdehyde. | |||||
| Uniprot ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
Probe 1 Probe Info |
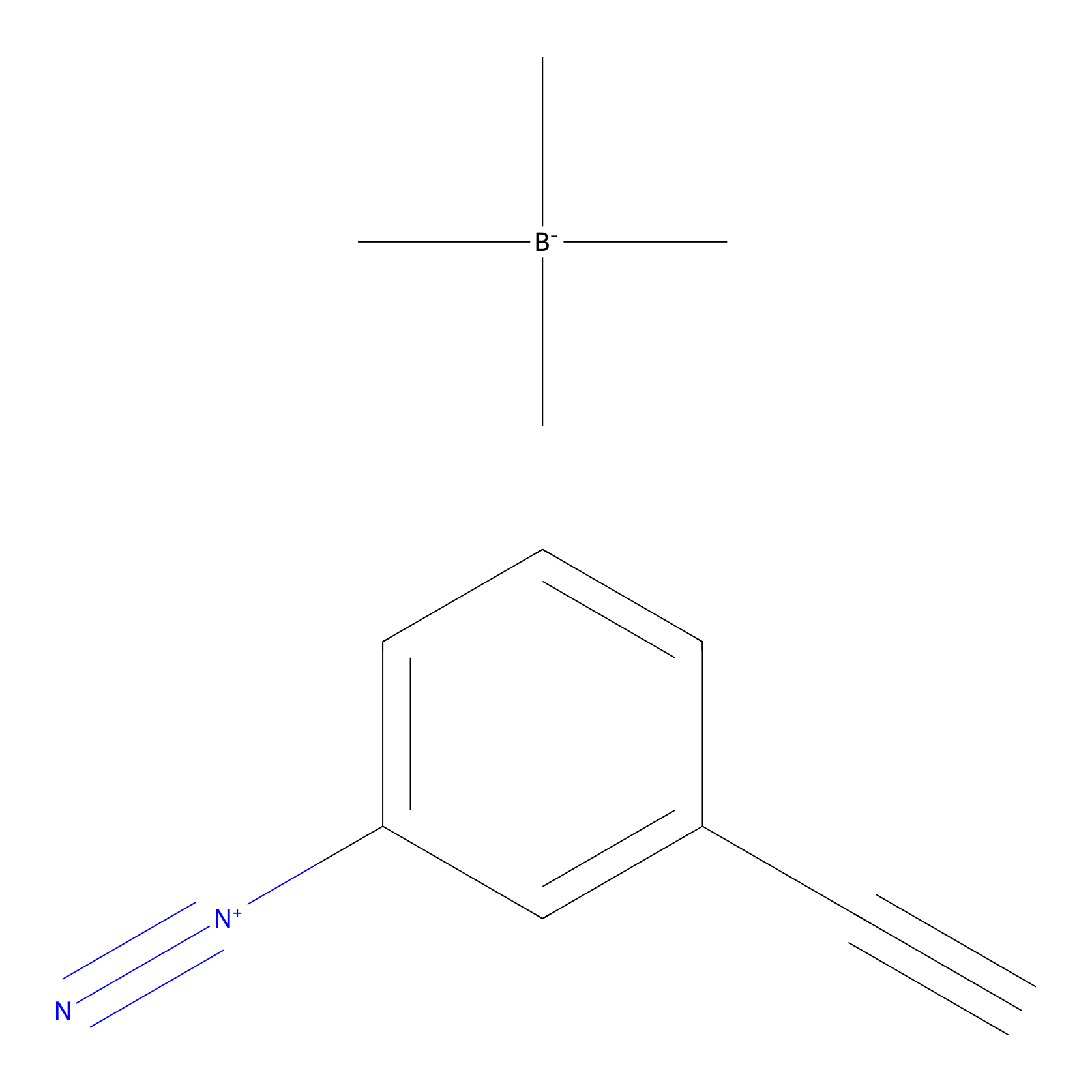 |
Y387(19.95); Y396(23.69) | LDD3495 | [1] | |
|
DBIA Probe Info |
 |
C443(0.85) | LDD3336 | [2] | |
|
NAIA_5 Probe Info |
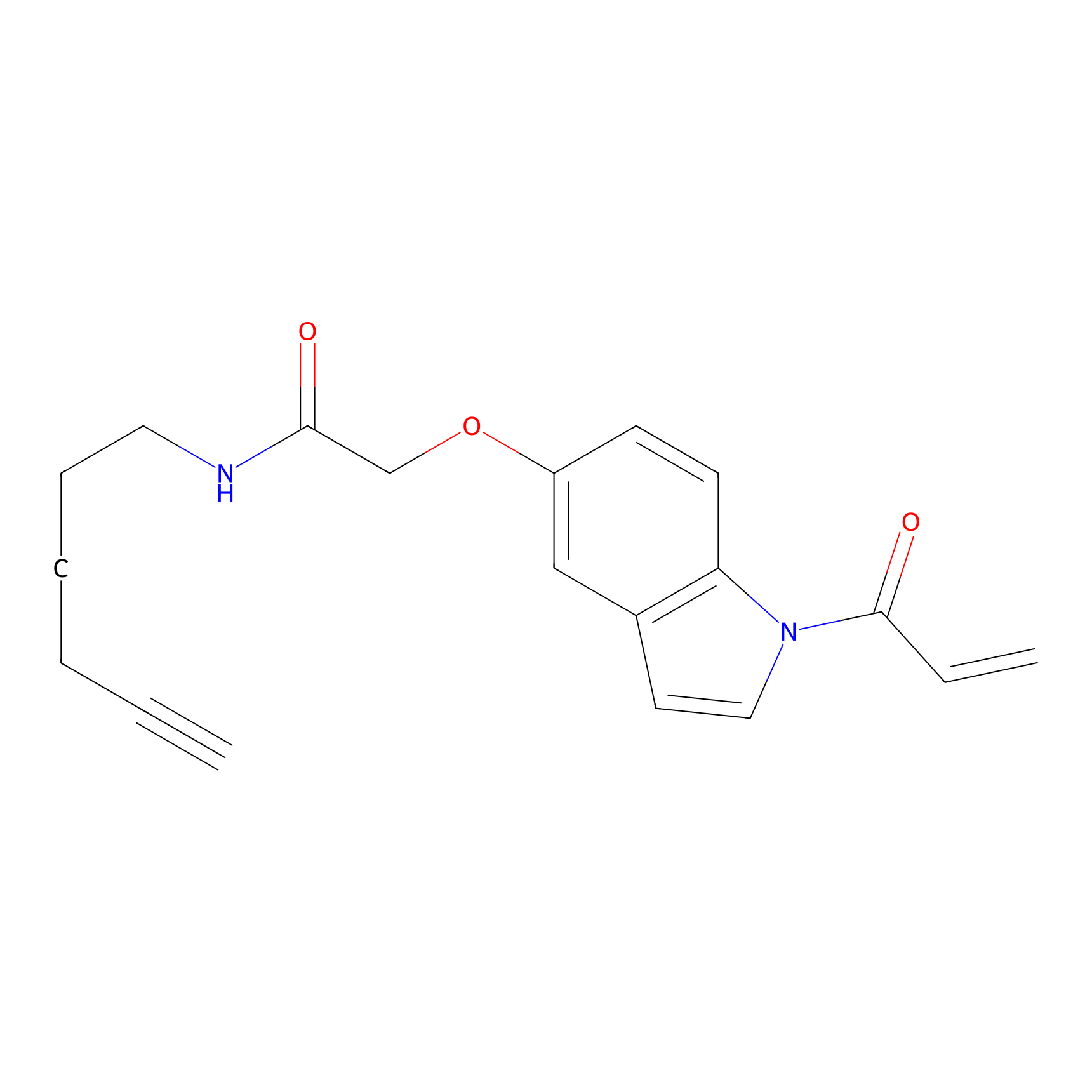 |
C416(0.00); C288(0.00) | LDD2223 | [3] | |
Competitor(s) Related to This Target
References
