Details of the Target
General Information of Target
Target Site Mutations in Different Cell Lines
| Cell line | Mutation details | Probe for labeling this protein in this cell | |||
|---|---|---|---|---|---|
| CMK | SNV: p.P429L | DBIA Probe Info | |||
| DU145 | SNV: p.V272M | . | |||
| HEC1B | SNV: p.R134C | . | |||
| HT115 | SNV: p.E321Ter | . | |||
| JHH7 | SNV: p.W291C | . | |||
| NCIH716 | SNV: p.S228F | . | |||
| SKHEP1 | SNV: p.D206E | . | |||
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
BTD Probe Info |
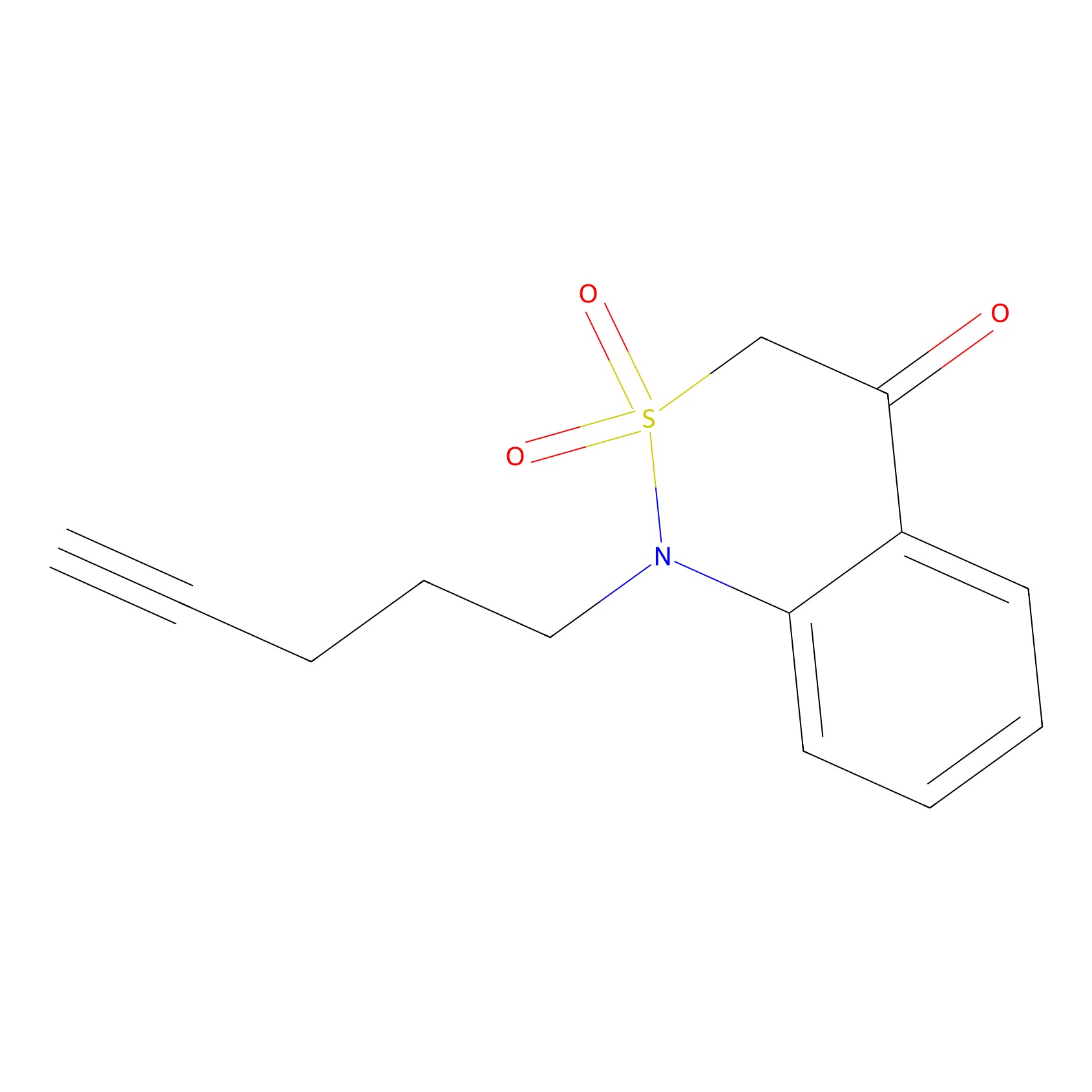 |
C110(1.03) | LDD2104 | [1] | |
|
DBIA Probe Info |
 |
C40(9.45) | LDD0209 | [2] | |
|
4-Iodoacetamidophenylacetylene Probe Info |
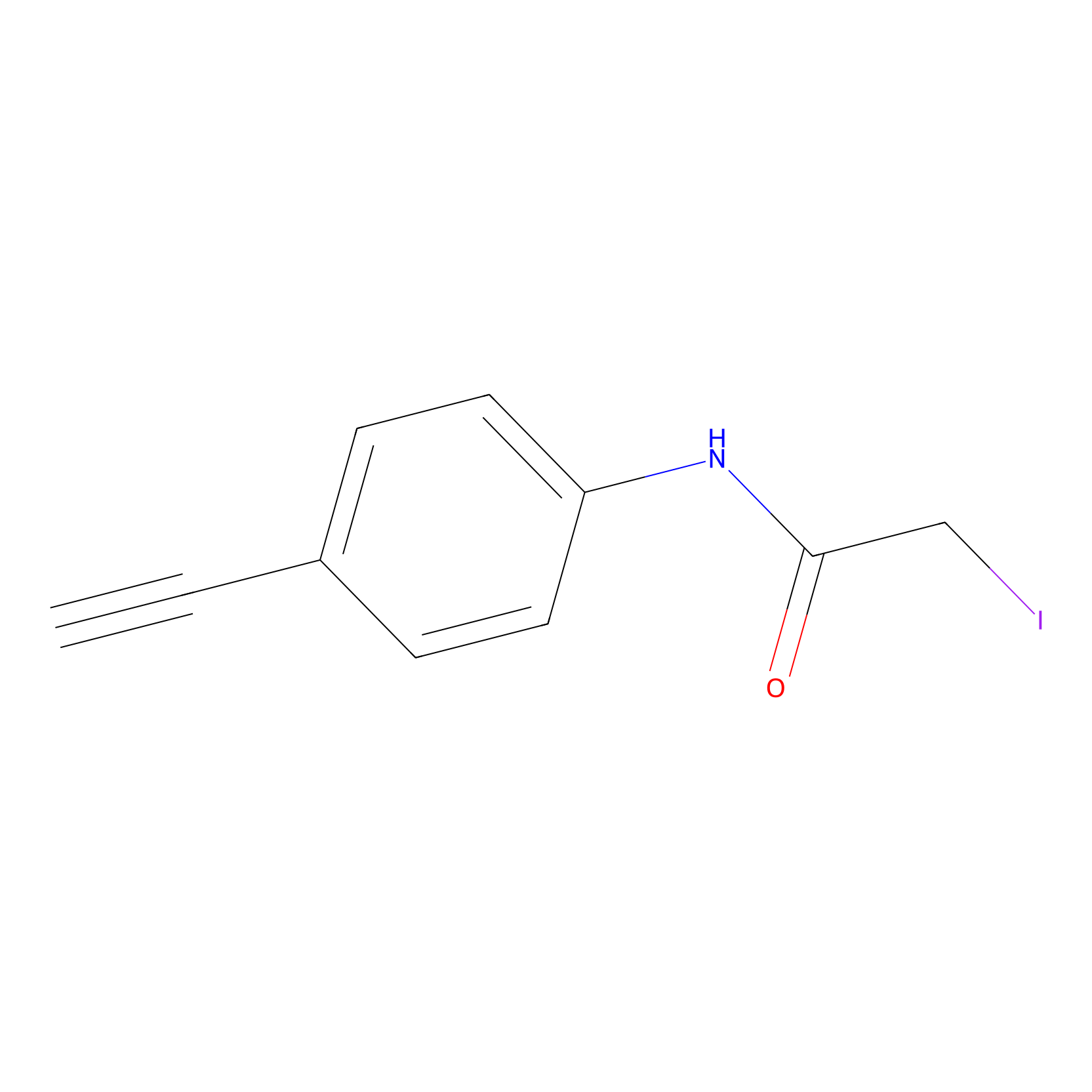 |
C243(0.00); C40(0.00); C422(0.00); C287(0.00) | LDD0038 | [3] | |
|
IA-alkyne Probe Info |
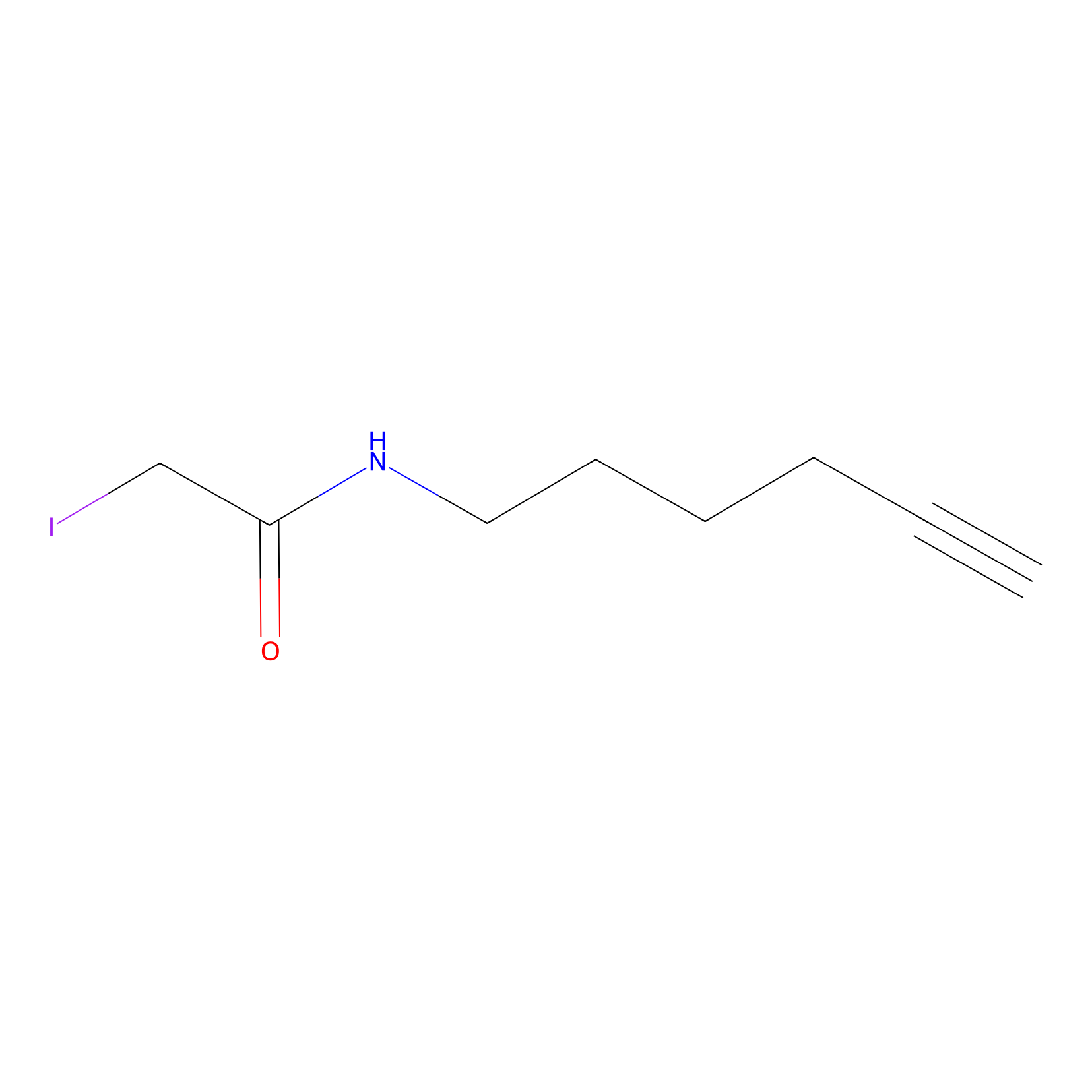 |
C243(0.00); C226(0.00); C40(0.00); C422(0.00) | LDD0036 | [3] | |
|
Lodoacetamide azide Probe Info |
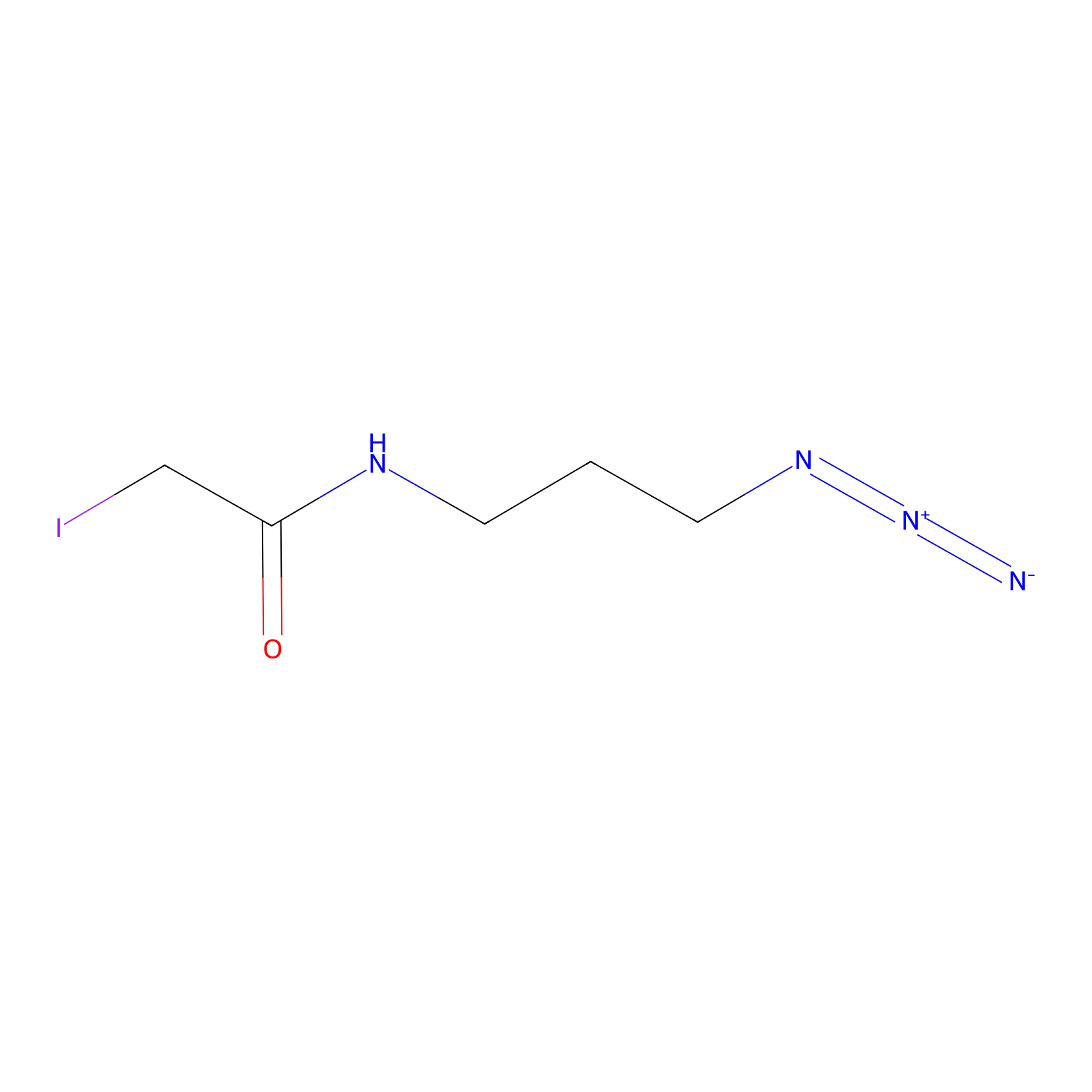 |
C243(0.00); C226(0.00); C40(0.00); C422(0.00) | LDD0037 | [3] | |
|
Compound 10 Probe Info |
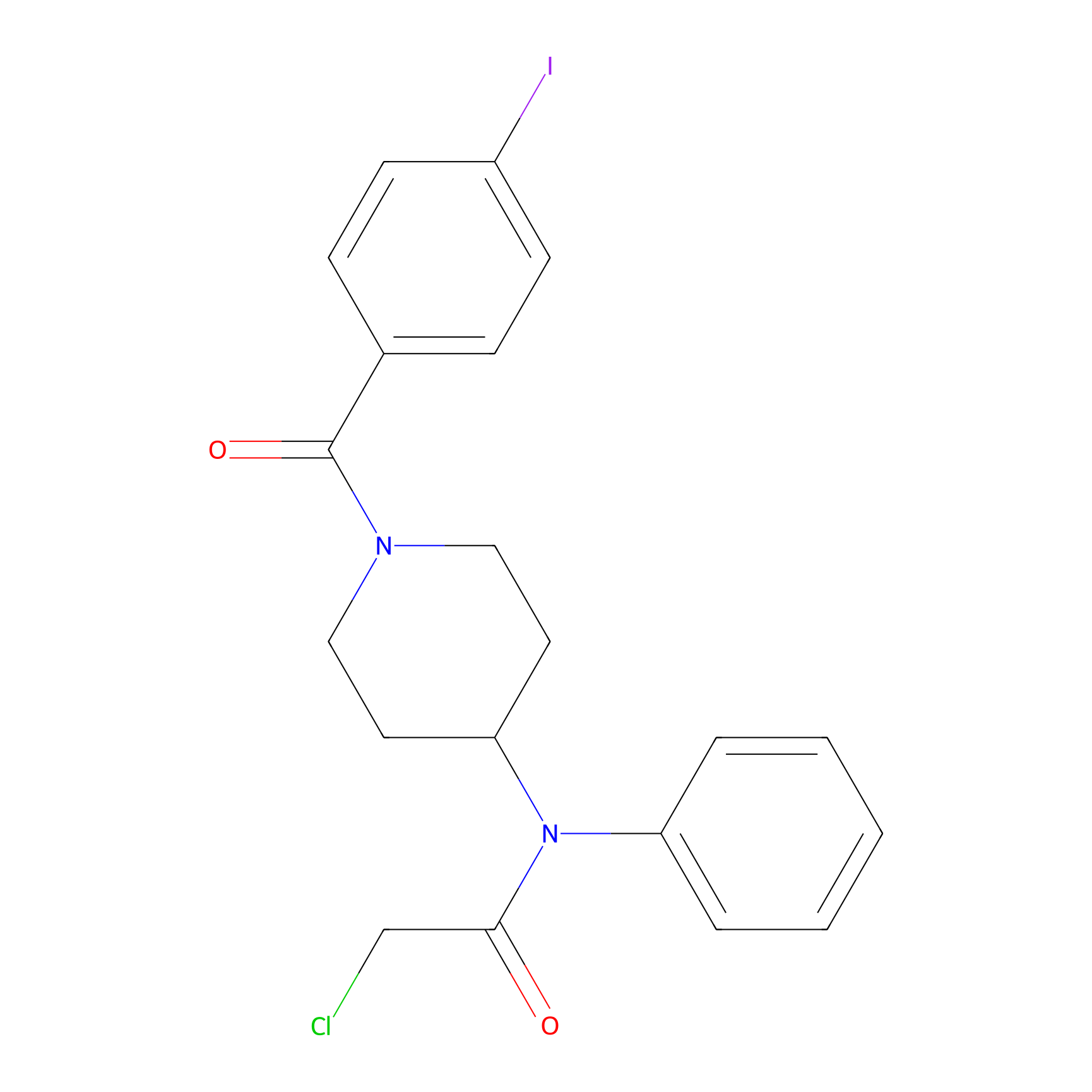 |
N.A. | LDD2216 | [4] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0548 | 1-(4-(Benzo[d][1,3]dioxol-5-ylmethyl)piperazin-1-yl)-2-nitroethan-1-one | MDA-MB-231 | C110(0.92) | LDD2142 | [1] |
| LDCM0519 | 1-(6-methoxy-3,4-dihydroquinolin-1(2H)-yl)-2-nitroethan-1-one | MDA-MB-231 | C110(1.08) | LDD2112 | [1] |
| LDCM0022 | KB02 | 22RV1 | C189(0.96) | LDD2243 | [5] |
| LDCM0023 | KB03 | Jurkat | C40(9.45) | LDD0209 | [2] |
| LDCM0024 | KB05 | COLO792 | C189(1.19) | LDD3310 | [5] |
| LDCM0511 | Nucleophilic fragment 18b | MDA-MB-231 | C110(1.03) | LDD2104 | [1] |
| LDCM0512 | Nucleophilic fragment 19a | MDA-MB-231 | C110(1.01) | LDD2105 | [1] |
| LDCM0526 | Nucleophilic fragment 26a | MDA-MB-231 | C110(1.50) | LDD2119 | [1] |
| LDCM0536 | Nucleophilic fragment 31 | MDA-MB-231 | C110(1.20) | LDD2129 | [1] |
| LDCM0547 | Nucleophilic fragment 41 | MDA-MB-231 | C110(0.79) | LDD2141 | [1] |
| LDCM0556 | Nucleophilic fragment 8a | MDA-MB-231 | C110(0.62) | LDD2150 | [1] |
References
