Details of the Target
General Information of Target
Target Site Mutations in Different Cell Lines
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
OPA-S-S-alkyne Probe Info |
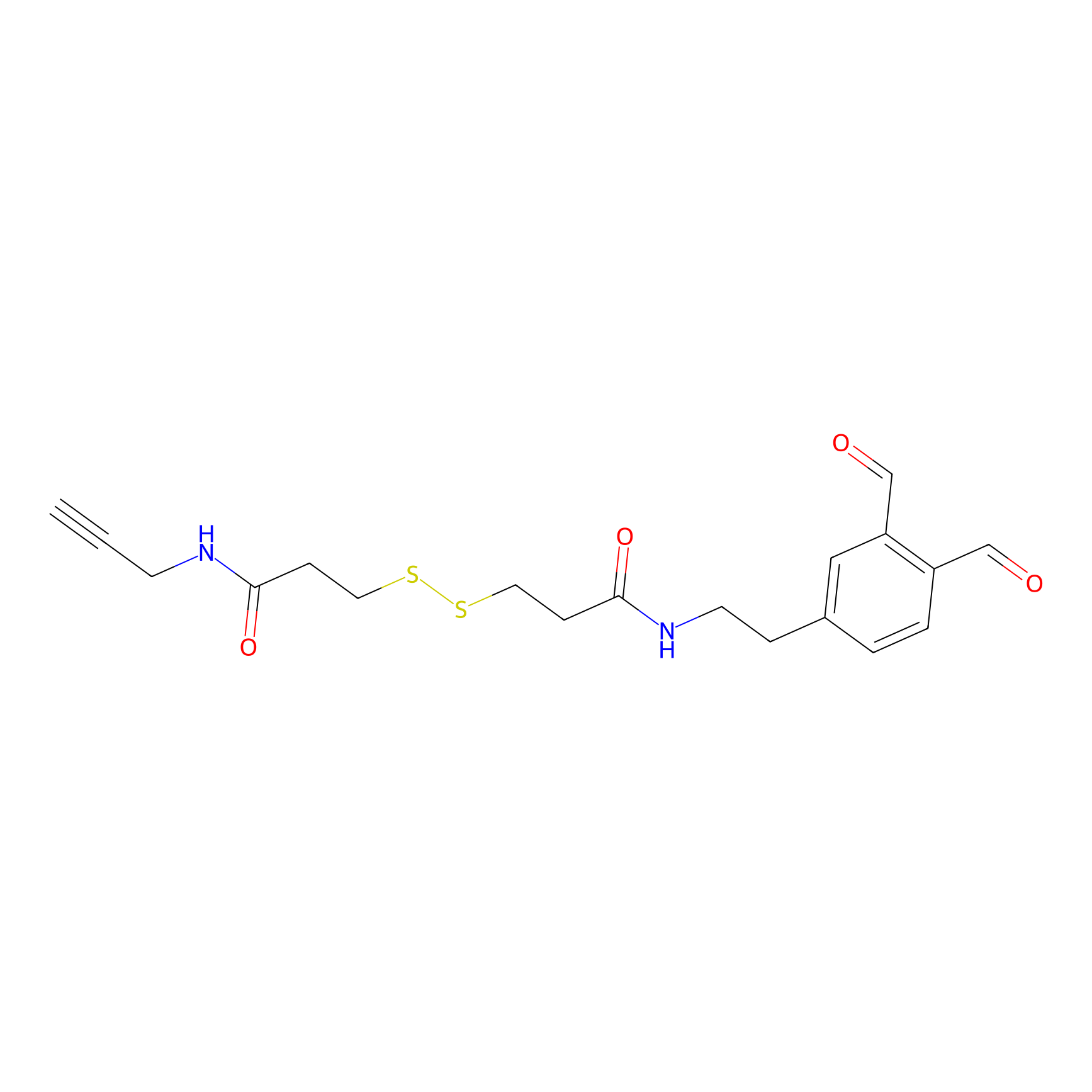 |
K346(0.56); K241(3.23); K74(6.22) | LDD3494 | [1] | |
|
DBIA Probe Info |
 |
C64(1.60) | LDD3414 | [2] | |
|
Acrolein Probe Info |
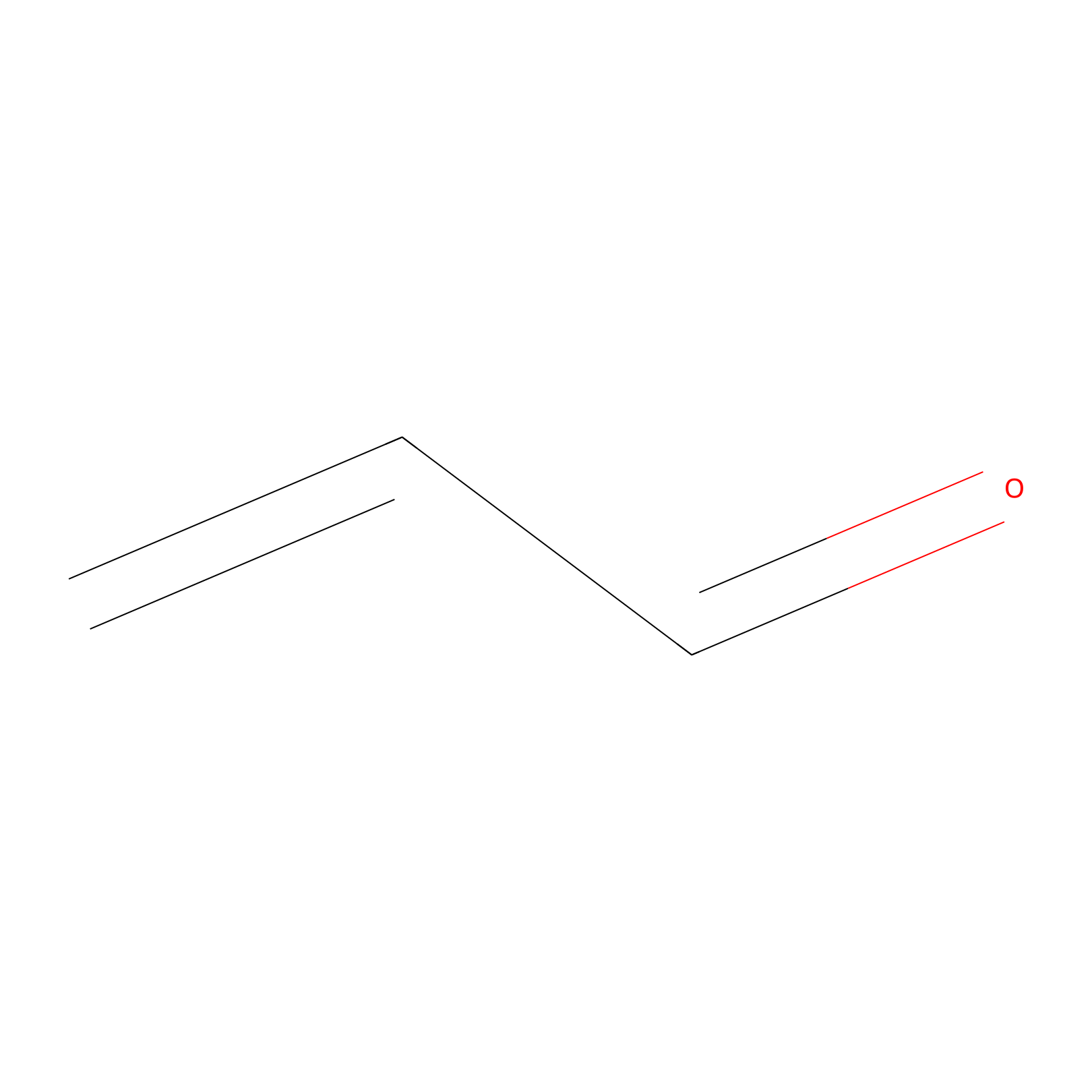 |
N.A. | LDD0217 | [3] | |
Competitor(s) Related to This Target
| Competitor ID | Name | Cell line | Binding Site(Ratio) | Interaction ID | Ref |
|---|---|---|---|---|---|
| LDCM0108 | Chloroacetamide | HeLa | H78(0.00); H79(0.00) | LDD0222 | [3] |
| LDCM0107 | IAA | HeLa | N.A. | LDD0221 | [3] |
| LDCM0022 | KB02 | CAL-72 | C64(1.59) | LDD2290 | [2] |
| LDCM0023 | KB03 | CAL-72 | C64(2.27) | LDD2707 | [2] |
| LDCM0024 | KB05 | SAOS-2 | C64(1.60) | LDD3414 | [2] |
| LDCM0109 | NEM | HeLa | N.A. | LDD0223 | [3] |
The Interaction Atlas With This Target
References
