Details of the Target
General Information of Target
| Target ID | LDTP00628 | |||||
|---|---|---|---|---|---|---|
| Target Name | Transmembrane protease serine 2 (TMPRSS2) | |||||
| Gene Name | TMPRSS2 | |||||
| Gene ID | 7113 | |||||
| Synonyms |
PRSS10; Transmembrane protease serine 2; EC 3.4.21.122; Serine protease 10) [Cleaved into: Transmembrane protease serine 2 non-catalytic chain; Transmembrane protease serine 2 catalytic chain] |
|||||
| 3D Structure | ||||||
| Sequence |
MALNSGSPPAIGPYYENHGYQPENPYPAQPTVVPTVYEVHPAQYYPSPVPQYAPRVLTQA
SNPVVCTQPKSPSGTVCTSKTKKALCITLTLGTFLVGAALAAGLLWKFMGSKCSNSGIEC DSSGTCINPSNWCDGVSHCPGGEDENRCVRLYGPNFILQVYSSQRKSWHPVCQDDWNENY GRAACRDMGYKNNFYSSQGIVDDSGSTSFMKLNTSAGNVDIYKKLYHSDACSSKAVVSLR CIACGVNLNSSRQSRIVGGESALPGAWPWQVSLHVQNVHVCGGSIITPEWIVTAAHCVEK PLNNPWHWTAFAGILRQSFMFYGAGYQVEKVISHPNYDSKTKNNDIALMKLQKPLTFNDL VKPVCLPNPGMMLQPEQLCWISGWGATEEKGKTSEVLNAAKVLLIETQRCNSRYVYDNLI TPAMICAGFLQGNVDSCQGDSGGPLVTSKNNIWWLIGDTSWGSGCAKAYRPGVYGNVMVF TDWIYRQMRADG |
|||||
| Target Type |
Literature-reported
|
|||||
| Target Bioclass |
Enzyme
|
|||||
| Family |
Peptidase S1 family
|
|||||
| Subcellular location |
Secreted; Cell membrane
|
|||||
| Function |
Plasma membrane-anchored serine protease that cleaves at arginine residues. Participates in proteolytic cascades of relevance for the normal physiologic function of the prostate. Androgen-induced TMPRSS2 activates several substrates that include pro-hepatocyte growth factor/HGF, the protease activated receptor-2/F2RL1 or matriptase/ST14 leading to extracellular matrix disruption and metastasis of prostate cancer cells. In addition, activates trigeminal neurons and contribute to both spontaneous pain and mechanical allodynia.; (Microbial infection) Facilitates human coronaviruses SARS-CoV and SARS-CoV-2 infections via two independent mechanisms, proteolytic cleavage of ACE2 receptor which promotes viral uptake, and cleavage of coronavirus spike glycoproteins which activates the glycoprotein for host cell entry. The cleavage of SARS-COV2 spike glycoprotein occurs between the S2 and S2' site. Upon SARS-CoV-2 infection, increases syncytia formation by accelerating the fusion process. Proteolytically cleaves and activates the spike glycoproteins of human coronavirus 229E (HCoV-229E) and human coronavirus EMC (HCoV-EMC) and the fusion glycoproteins F0 of Sendai virus (SeV), human metapneumovirus (HMPV), human parainfluenza 1, 2, 3, 4a and 4b viruses (HPIV). Essential for spread and pathogenesis of influenza A virus (strains H1N1, H3N2 and H7N9); involved in proteolytic cleavage and activation of hemagglutinin (HA) protein which is essential for viral infectivity.
|
|||||
| TTD ID | ||||||
| Uniprot ID | ||||||
| DrugMap ID | ||||||
| Ensemble ID | ||||||
| HGNC ID | ||||||
| ChEMBL ID | ||||||
Target Site Mutations in Different Cell Lines
| Cell line | Mutation details | Probe for labeling this protein in this cell | |||
|---|---|---|---|---|---|
| MCC13 | SNV: p.E23K | DBIA Probe Info | |||
| NB1 | SNV: p.G142V | . | |||
| NCIH446 | Deletion: p.V64SfsTer26 | DBIA Probe Info | |||
| NCIH716 | Substitution: p.I291_V292delinsMM | . | |||
| REH | SNV: p.Q68Ter | . | |||
| SKNSH | SNV: p.W267C | . | |||
Probe(s) Labeling This Target
ABPP Probe
| Probe name | Structure | Binding Site(Ratio) | Interaction ID | Ref | |
|---|---|---|---|---|---|
|
DBIA Probe Info |
 |
C31(1.81) | LDD3310 | [1] | |
|
NAIA_5 Probe Info |
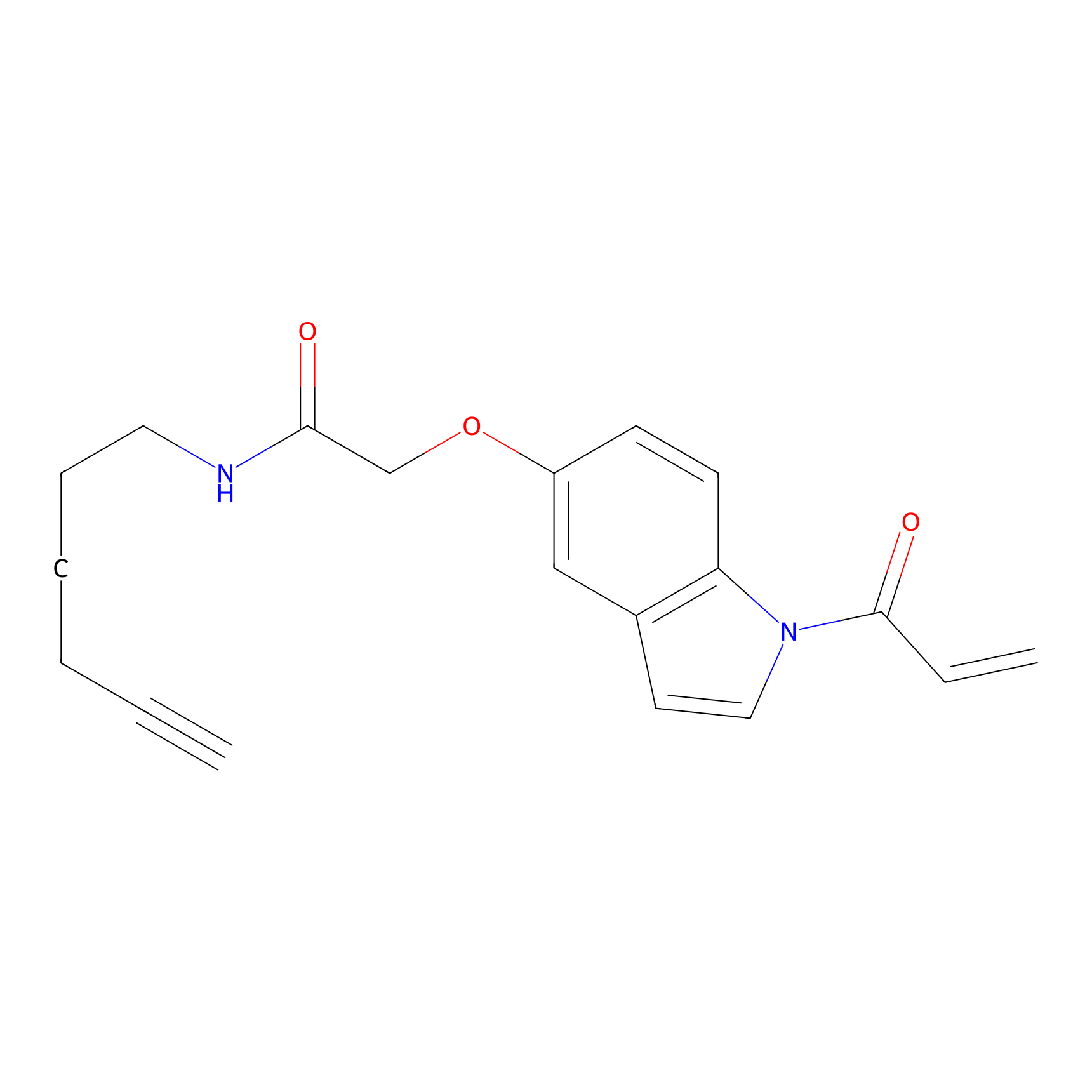 |
N.A. | LDD2225 | [2] | |
Competitor(s) Related to This Target
References
